��Ŀ����
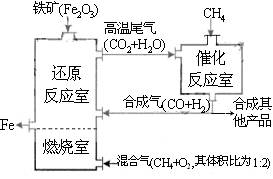
��¯ұ������������ͼ��ʹ��Ȼ�������������������������ȼ������ʹ��������Ȼ��ȼ��CH4(g)+2O2(g)=CO2(g)+2H2O(g)������Ӧ�ҷ����ķ�ӦΪ��CH4(g)+H2O(g) ![]() CO(g)+3H2(g)����H1=+216kJ/mol��CH4(g)+ CO2(g)
CO(g)+3H2(g)����H1=+216kJ/mol��CH4(g)+ CO2(g)![]() 2CO(g) + 2H2(g)�� ��H2=+260kJ/mol������������ƽ��Ĵ��ڣ�������˵����ȷ����
2CO(g) + 2H2(g)�� ��H2=+260kJ/mol������������ƽ��Ĵ��ڣ�������˵����ȷ����
A���������Ӧ�ҵ�ѹǿ�������ת���ʼ�С
B��������ά����550��750�棬Ŀ�Ľ������CH4ת��������
C������ȼ���ҵ���ҪĿ���Dz���CO2��ˮ������ԭ��������鷴Ӧ
D��������Ӧ���У��ﵽƽ��ʱ��������n(CH4)=amol,n(CO)=bmol
n(H2)=cmol,��ͨ�����Ӧ�ҵ�CH4�����ʵ���Ϊa+(b+c)/4
AD
����:
���⿼�黯ѧƽ�⡣�������֪������Ӧ�ҷ����������淴Ӧ������ѹǿ��ƽ�������ƶ��������ת���ʼ�С��Aѡ����ȷ���¶ȵ�Ҫ��Ҫ���Ǵ����Ļ��ԣ�Bѡ���������ȼ���ҵ�Ŀ�ij����ṩԭ����֮�⣬���ṩ��������Cѡ�����
CH4(g)+H2O(g) ![]() CO(g)+3H2(g) CH4(g)+ CO2(g)
CO(g)+3H2(g) CH4(g)+ CO2(g)![]() 2CO(g) + 2H2(g)
2CO(g) + 2H2(g)
��ʼ�� m 0 0 n 0 0
ת���� x x x 3x y y 2y 2y
ƽ�⣺ m-x x 3x n-y 2y 2y
��x+2y=b��3x+2y=c �ó�x+y=(b+c)/4����ƽ��ʱn(CH4)=m-x+n-y=amol����ͨ�����Ӧ�ҵ�CH4�����ʵ���Ϊm+n=a+x+y=a+(b+c)/4mol��Dѡ����ȷ��

 ��2011?������ģ����¯ұ������������ͼ��ʾ��ȼ��������Ҫ��ӦΪ��
��2011?������ģ����¯ұ������������ͼ��ʾ��ȼ��������Ҫ��ӦΪ�� ��¯ұ������������ͼ��ʹ��Ȼ�������������������������ȼ������ʹ��������Ȼ��ȼ��CH4��g��+2O2��g��=CO2��g��+2H2O��g��������Ӧ�ҷ����ķ�ӦΪ��CH4��g��+H2O��g��?CO��g��+3H2��g����H1=+216kJ/mol��CH4��g��+CO2��g��?2CO��g��+2H2��g����H2=+260kJ/mol������������ƽ��Ĵ��ڣ�������˵����ȷ���ǣ�������
��¯ұ������������ͼ��ʹ��Ȼ�������������������������ȼ������ʹ��������Ȼ��ȼ��CH4��g��+2O2��g��=CO2��g��+2H2O��g��������Ӧ�ҷ����ķ�ӦΪ��CH4��g��+H2O��g��?CO��g��+3H2��g����H1=+216kJ/mol��CH4��g��+CO2��g��?2CO��g��+2H2��g����H2=+260kJ/mol������������ƽ��Ĵ��ڣ�������˵����ȷ���ǣ�������

 CO(g)+3H2(g) ∆H1=+216
kJ/mol��
CO(g)+3H2(g) ∆H1=+216
kJ/mol��