��Ŀ����
Ϊ֤��ij������һԪ�ᣨHX�������ᣬ������ʵ�鷽�������в��������ǣ�������
| A�������£���0.1mol/L HX��Һ��pH����pH��1��֤��HX������ |
| B�������£���1mol/L NaX��Һ��pH����pH��7��֤��HX������ |
| C������Ũ�ȡ��������HCl��NaX��Һ��ϣ��������ҺpH��7��֤��HX������ |
| D����ͬ�����£���0.1 mol/L��HCl��0.1 mol/L��HX���е�����ʵ�飬����HX��Һ�മ���ĵ��ݽϰ���֤��HXΪ���� |
���㣺���������ˮ��Һ�еĵ���ƽ��
ר�⣺����ƽ������Һ��pHר��
������ֻҪ֤��HX��ˮ��Һ�ﲿ�ֵ������֤��HX��������ʣ����õķ����У�һ��Ũ��HX��Һ��pH������ͬŨ�ȵ�һԪǿ��Ƚ���Һ������ǿ��������NaX��Һ��������жϵȣ��ݴ˷������
���
�⣺A�������£���0.1mol/L HX��Һ��pH����pH��1��˵��HX��ˮ��Һ�ﲿ�ֵ��룬��֤��HX�����ᣬ��A��ȷ��
B�����£���1mol/L NaX��Һ��pH����pH��7����NaX��ǿ�������Σ�����Һ�ʼ��ԣ���֤��HX�����ᣬ��B��ȷ��
C������Ũ�ȡ��������HCl��NaX��Һ��ϣ���Һ�е�������NaCl��HX������HX��ǿ�ỹ�����ᣬHCl������������Ӷ�ʹ��Һ�����ԣ������������ҺpH��7������֤��HX�����ᣬ��C����
D����ͬ�����£���0.1 mol/L��HCl��0.1 mol/L��HX���е�����ʵ�飬����HX��Һ�മ���ĵ��ݽϰ���˵��HX��Һ������Ũ��С��HCl����֤��HX���ֵ��룬����HXΪ���ᣬ��D��ȷ��
��ѡC��
B�����£���1mol/L NaX��Һ��pH����pH��7����NaX��ǿ�������Σ�����Һ�ʼ��ԣ���֤��HX�����ᣬ��B��ȷ��
C������Ũ�ȡ��������HCl��NaX��Һ��ϣ���Һ�е�������NaCl��HX������HX��ǿ�ỹ�����ᣬHCl������������Ӷ�ʹ��Һ�����ԣ������������ҺpH��7������֤��HX�����ᣬ��C����
D����ͬ�����£���0.1 mol/L��HCl��0.1 mol/L��HX���е�����ʵ�飬����HX��Һ�മ���ĵ��ݽϰ���˵��HX��Һ������Ũ��С��HCl����֤��HX���ֵ��룬����HXΪ���ᣬ��D��ȷ��
��ѡC��
���������⿼���˵����ǿ���жϣ����ݵ���ʵ���̶�ȷ�������ǿ�������ܸ�����Һ������ǿ���жϣ�֪���ж�������ʵij��÷�������Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
���ж��л����������ʶ��ȷ���ǣ�������
| A���л������ﶼ�ǹ��ۻ����� |
| B���л������ﶼ����̼����Ԫ�� |
| C���л������ﶼ����ͬ���칹���� |
| D���л������������̼ԭ�ӵijɼ����������� |
����Ԫ���в����ڵ�������Ԫ�ص��ǣ�������
| A���� | B���� | C���� | D���� |
���л�ѧ����ʽ�У�����ˮ�ⷴӦ���ǣ�������
| A��H2O+H2O?H3O++OH- |
| B��HCO3-+OH-?H2O+CO32- |
| C��CO2+H2O?H2CO3 |
| D��CO+H2O?HCO3-+OH- |
������Ԫ��A��B��C��D��ԭ�����������������ǵ�ԭ������֮��Ϊ36����ԭ������������֮��Ϊ14��A��Cԭ�ӵ�����������֮�͵���Bԭ�ӵĴ�����������A��C��B��D��Ϊͬ����Ԫ�أ�����������ȷ����
��������
��������
| A���ڵؿ��У�BԪ�صĺ���λ�ڵڶ�λ |
| B��A��B��D����Ԫ���γɵĻ�����һ����ǿ�� |
| C��AԪ����CԪ���γɵĻ�������ˮ��Ӧ����Һ�ʼ��� |
| D��BԪ����CԪ���γɵĻ�����C2B��C2B2�����������Ӹ����Ȳ�ͬ |
��NA��ʾ�����ӵ�������ֵ������˵����ȷ���ǣ�������
| A��������Cl2�ڹ����·�Ӧ��ÿ��NA����ԭ�ӱ�������������ı�״����Cl2 11.2L |
| B��1mol���к���3NA��̼̼˫�� |
| C��14g��ϩ�ͱ�ϩ�Ļ�����к���3NA�Թ��õ��Ӷ� |
| D����״���£�22.4L�����е�̼ԭ����Ϊ6NA |
Fe��OH��3�����ȶ����ڵ���Ҫԭ���ǣ�������
| A��Fe��OH��3�н���ֱ��С��1nm |
| B�����Է������������ |
| C��Fe��OH��3��ͨ����ֽ |
| D��Fe��OH��3����������� |
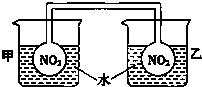 ����NO2ƽ������������ɫ���������ɫ��dz��ָʾ���ȹ��̺����ȹ��̣�
����NO2ƽ������������ɫ���������ɫ��dz��ָʾ���ȹ��̺����ȹ��̣�