��Ŀ����
8�������仯�����ڿƼ���������������Ź㷺��Ӧ�ã��밴Ҫ��ش��������⣮��1����֪��N2��g��+2O2��g���T2NO2��g����H=-67.7kJ/mol
N2H4��g��+O2��g���TN2��g��+2H2O��g����H=-534.0kJ/mol
2NO2��g��?N2O4��g����H=-52.7kJ/mol
�������N2O4�����������£�N2H4����ȼ�ϣ���д����̬������̬N2O4��ȼ�����ɵ�������̬ˮ���Ȼ�ѧ����ʽ��2N2H4��g��+N2O4��g��=3N2��g��+4H2O��g����H=-947.6kJ•mol-1��
��2��������������ƽ��ת����ͼ1��ʾ��A�Ǻ��º��ݵ�������B����ʴ�����ȡ��ݻ��ɱ���ܱ������ң��ر�K2������l mol NO2ͨ��Kl��K3�ֱ����A��B�У���Ӧ��ʼʱA��B�������ͬ��ΪaL��
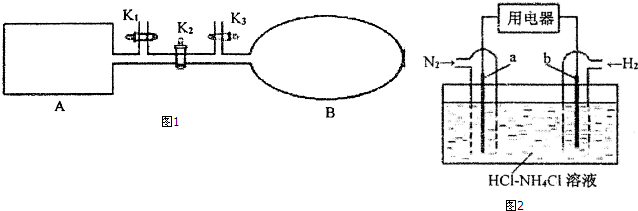
�ٿ������ж�B�п��淴Ӧ2NO2��g��?N2O4��g���Ѿ��ﵽƽ��ı�־�ǣ�д��һ����Ҳ��ٱ�С����ɫ���ٱ仯��
������A�е���ƽ������ʱ��3s���ﵽƽ���������ѹǿΪ��ʼѹǿ��0.8������ƽ����ѧ��Ӧ����V��N2O4��=$\frac{1}{15a}$mol/��L��s����ƽ�ⳣ����ֵK=$\frac{5a}{9}$��
����ʱ��K2��ƽ���B�������������0.3a L�����K2֮ǰ������B���Ϊ0.65aL��
��ԭƽ�����A�������ٳ���0.6mol N2O4�������µ���ƽ���ƽ��������NO2�������������д���������С�����䡱����С��
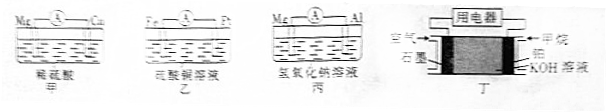
��3����N2��H2Ϊ�缫��Ӧ�HCl-NH4ClΪ�������Һ��ʹ�����͵缫���ϣ��������һ�ּ����ṩ���ܣ�����ʵ�ֵ��̶�������ȼ�ϵ�أ�ԭ����ͼ2��ʾ��
�ٸ�ȼ�ϵ�صĸ���Ϊ���a����b����b��
�ڵ��������ӦʽΪN2+8H++6e-=2NH4+��
���� ��1���ɢ�N2��g��+2O2��g��=2NO2��g����H=-67.7kJ•mol-1
��N2H4��g��+O2��g��=N2��g��+2H2O��g����H=-534.0kJ•mol-1
��2NO2��g��?N2O4��g����H=-52.7kJ•mol-1
���ݸ�˹���ɿ�֪�ڡ�2-��-�۵�2N2H4��g��+N2O4��g��=3N2��g��+4H2O��g������H=��-534.0kJ•mol-1����2-��-67.7kJ•mol-1��-��-52.7kJ•mol-1��=-947.6 kJ•mol-1���ɴ˷������
��2���ٷ�Ӧ�ﵽƽ��״̬ʱ�����淴Ӧ������ȣ�ƽ��ʱ�������ʵ����ʵ�����Ũ�ȵȲ��ٷ����仯��
��ѹǿ֮�ȵ������ʵ���֮�ȣ�ƽ��ʱ����������ʵ���Ϊ1mol��0.8=0.8mol����
2NO2��g��?N2O4��g��
��ʼ����mol����1 0
ת������mol����x 0.5x
ƽ������mol����1-x 0.5x
����1-x+0.5x=0.8�����x=0.4��
��v��N2O4��=$\frac{\frac{0.2mol}{aL}}{3s}$=$\frac{1}{15a}$mol/��L��s����K=$\frac{\frac{0.2}{a}}{��\frac{0.6}{a}��^{2}}$=$\frac{5a}{9}$�����ݵ�Чƽ���������ƽ�����A���ٳ���0.6molN2O4����ЧΪ��ԭƽ�����������ѹǿ��ƽ�������ƶ���
��3����������0�۱��+1�ۣ����ϼ����߷���������Ӧ��b�Ǹ�����
�ڵ�����������õ��ӣ���������������NH4+���缫��ӦʽΪ��N2+8H++6e-=2NH4+��
��� �⣺��1���ɢ�N2��g��+2O2��g��=2NO2��g����H=-67.7kJ•mol-1
��N2H4��g��+O2��g��=N2��g��+2H2O��g����H=-534.0kJ•mol-1
��2NO2��g��?N2O4��g����H=-52.7kJ•mol-1
���ݸ�˹���ɿ�֪�ڡ�2-��-�۵�2N2H4��g��+N2O4��g��=3N2��g��+4H2O��g����
��H=��-534.0kJ•mol-1����2-��-67.7kJ•mol-1��-��-52.7kJ•mol-1��=-947.6 kJ•mol-1��
���Ȼ�ѧ����ʽΪ2N2H4��g��+N2O4��g��=3N2��g��+4H2O��g����H=-947.6 kJ•mol-1��
�ʴ�Ϊ��2N2H4��g��+N2O4��g��=3N2��g��+4H2O��g����H=-947.6 kJ•mol-1��
��2����B��һ������ѹǿ��������������Ե���Ӧ�ﵽƽ��״̬ʱ������������ɫ������������ٸı䣬
�ʴ�Ϊ�����Ҳ��ٱ�С����ɫ���ٱ仯��
��ѹǿ֮�ȵ������ʵ���֮�ȣ�ƽ��ʱ����������ʵ���Ϊ1mol��0.8=0.8mol����
2NO2��g��?N2O4��g��
��ʼ����mol����1 0
ת������mol����x 0.5x
ƽ������mol����1-x 0.5x
����1-x+0.5x=0.8�����x=0.4��
��v��N2O4��=$\frac{\frac{0.2mol}{aL}}{3s}$=$\frac{1}{15a}$mol/��L��s����K=$\frac{\frac{0.2}{a}}{��\frac{0.6}{a}��^{2}}$=$\frac{5a}{9}$����K2�����൱�����ڵ��µ�ѹʱ��ƽ�⣬���ƽ��ʱ��Ч�ģ����ڴ�ʱ��Ӧ������ʵ�����B�еĶ��������Դ�K2֮ǰ������B���Ϊ��aL+0.3aL����2=0.65aL����ƽ�����A���ٳ���0.6molN2O4����ЧΪ��ԭƽ�����������ѹǿ��ƽ�������ƶ������µ���ƽ���ƽ��������NO2�����������С��
�ʴ�Ϊ��$\frac{1}{15a}$mol/��L��s����$\frac{5a}{9}$��0.65a�� ��С��
��3����������0�۱��+1�ۣ����ϼ����߷���������Ӧ���Ǹ������ʴ�Ϊ��b��
�ڵ�����������õ��ӣ���������������NH4+���缫��ӦʽΪ��N2+8H++6e-=2NH4+���ʴ�Ϊ��N2+8H++6e-=2NH4+��
���� ���⿼�黯ѧƽ����㣬�漰��Чƽ�����⣬�ϺõĿ���ѧ��������������������ؼ��ǵ�Чƽ��;���Ľ������Ѷ��еȣ�

 ��˼ά������ҵϵ�д�
��˼ά������ҵϵ�д�| A�� | �٢� | B�� | �٢� | C�� | �ڢ� | D�� | ȫ����ȷ |
| A�� | ��CaO����ˮ�У�CaO�TCa2++O2- | |
| B�� | ����NaHSO4���ۻ���NaHSO4�TNa++H++SO42- | |
| C�� | ��Al2��SO4��3����ˮ�У�Al2��SO4��3�TAl3++SO42- | |
| D�� | ��NaCl����ˮ�У�NaCl�TNa++Cl- |
| A�� | Na+ 1s22s22p6 | B�� | N3+ 1s22s22p6 | C�� | F?1s22s22p6 | D�� | O2?1s22s22p6 |
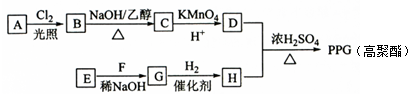
��1������ԭ���ԭ����д�±���
| װ����� | ���� | ���������� ����缫�������ƣ� |
| �� | ||
| �� | �� |
��3������������ط��������������Ƿ�һ���μӵ缫��Ӧ����һ������ǡ������ǡ���һ��������
��4��������صķŵ�����У����е������Һ�ļ��Լ��������ǿ�������������䡱����ͬ�������е������Һ�ļ��Լ�����


 ��
��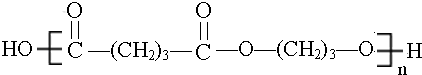 ��
�� ��
�� ��
��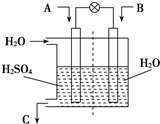 ����������ͼ��ʾװ�ã����õ绯ѧԭ����CO2��SO2ת��Ϊ��Ҫ����ԭ�ϣ�
����������ͼ��ʾװ�ã����õ绯ѧԭ����CO2��SO2ת��Ϊ��Ҫ����ԭ�ϣ�