��Ŀ����
��ÿ��2�֣�
���㣺
��12�֣���1��ijһ��Ӧ��ϵ�е������У�HCl��SnCl2��H2SnCl6��As��H3AsO3��H2O����֪��HCl�Ƿ�Ӧ��֮һ��
��д������ƽ�÷�Ӧ�Ļ�ѧ����ʽ��
���ڸ÷�Ӧ�У��õ��ӵ������� ����������Ԫ���� ��
���ڷ�Ӧ�У�ÿת��1 mol���ӣ�����HCl mol��
��2����2.5g̼���ơ�̼�����ƺ��������ƵĹ���������ȫ�ܽ���ˮ���Ƴ�ϡ��Һ��Ȼ�������Һ����μ���1mol��L��1�����ᣬ�������������������CO2���������״������ϵ����ͼ��ʾ��
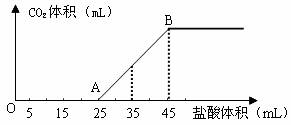
��д��OA����������Ӧ�����ӷ���ʽ______________________________________��
�ڵ�����35mL����ʱ������CO2�����Ϊ___________mL����״������
�ο��𰸣�1����12HCl + 3SnCl2 + 2H3AsO3 �� 3H2SnCl6 + 2As + 6H2O��2�֣�
��H3AsO3��1�֣� +2�۵�Sn��1�֣� ��2 ��2�֣�
��2����H+ + OH�� = H2O�� CO32�� + H+ = HCO3�� ��2��2��=4�֣� ��224 ��2�֣�

��ϰ��ϵ�д�
�����Ŀ
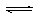 C��g��+D��g�����ڲ�ͬ�¶��£�D�����ʵ���n��D����ʱ��t�Ĺ�ϵ��ͼ���Իش��������⣺
C��g��+D��g�����ڲ�ͬ�¶��£�D�����ʵ���n��D����ʱ��t�Ĺ�ϵ��ͼ���Իش��������⣺