��Ŀ����
16����0.1mol•L-1 NaOH��Һ�ֱ�ζ������Ϊ20mLŨ�Ⱦ�Ϊ0.1mol•L-1 HCl��Һ��HX��Һ����Һ��pH�����NaOH��Һ����仯������ͼ��ʾ�������й�˵������ȷ���ǣ�������
| A�� | HXΪ���ᣬ�ҵ���ƽ�ⳣ��Ka��1��10-10 | |
| B�� | ˮ�ĵ���̶ȣ�M�����N�� | |
| C�� | P��c��Cl-��=0.05mol•L-1�� | |
| D�� | M��c��HX��-c��X-����c��OH-��-c��H+�� |
���� 0.1mol•L-1 HCl��Һ����ǿ����Һ��ȫ���룬������Һ��PH=1����0.1mol•L-1 HX��Һ��PH=5˵�������Ჿ�ֵ��룻
A������Ka=$\frac{c��{X}^{-}��•c��{H}^{+}��}{c��HX��}$���㣻
B��N��ΪNaX��Һ����M��Ϊ��Ũ��NaX��HX�����Һ��
C��P��c��Cl-��=$\frac{20��0.1}{20+10}$=0.067mol•L-1��
D��M��Ϊ��Ũ��NaX��HX�����Һ������Һ�ʼ��ԣ�˵��X-��ˮ��̶ȴ���HX�ĵ���̶ȣ��������غ��֪��2c��Na+��=c��X-��+c��HX������ϵ���غ�c��Na+��+c��H+��=c��X-��+c��OH-���жϣ�
��� �⣺A������Ka=$\frac{c��{X}^{-}��•c��{H}^{+}��}{c��HX��}$=$\frac{1{0}^{-5}��1{0}^{-5}}{0.1-1{0}^{-5}}$��1��10-9����A����
B��N��ΪNaX��Һ����ˮ�ĵ�����ٽ�����M��Ϊ��Ũ��NaX��HX�����Һ����ˮ�ĵ��������ƣ�����ˮ�ĵ���̶ȣ�M��С��N�㣬��B����
C��P��c��Cl-��=$\frac{20��0.1}{20+10}$=0.067mol•L-1����C����
D��M��Ϊ��Ũ��NaX��HX�����Һ������Һ�ʼ��ԣ�˵��X-��ˮ��̶ȴ���HX�ĵ���̶ȣ��������غ��֪��2c��Na+��=c��X-��+c��HX������ϵ���غ�c��Na+��+c��H+��=c��X-��+c��OH-���������ɵã�c��X-��+c��HX��+2c��H+��=2c��X-��+2c��OH-���������ã�c��HX��-c��X-��=2c��OH-��-2c��H+����c��OH-��-c��H+������D��ȷ��
��ѡD��
���� ���⿼��ԭ��ˮ�⡢������ʵ��롢����Ũ�ȴ�С�жϣ��ؼ��Ǹ���M���ж���ͬŨ����X-��ˮ��̶ȴ���HX�ĵ���̶ȣ��ѶȽϴ�

| A�� | 0.25mol | B�� | 0.2mol | C�� | 0.45mol | D�� | 0.65mol |
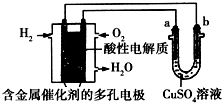
| A�� | ��װ�������������ͭʱ��aΪ�� | |
| B�� | ��װ�����ڵ��ͭʱ������ͭ��Һ��Ũ�Ȳ��� | |
| C�� | ȼ�ϵ����������ӦΪ$\frac{1}{2}$O2+2e-+2H+�TH2O | |
| D�� | ���ø�װ�ý��д�ͭ�ľ���������1 molˮ����ʱ���ɵõ�64 g��ͭ |
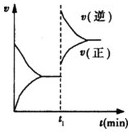
| A�� | t1ʱ��һ������С����������� | |
| B�� | t1ʱ����������������ͨ����һ������C���� | |
| C�� | t1ʱ�ı���������÷�Ӧ��ƽ�ⳣ�����ܼ�С��Ҳ���ܲ��� | |
| D�� | t1ʱ���������������м����˴��� |
| A�� | ��ˮ��Ư������ | B�� | ��ˮ�ʻ���ɫ | ||
| C�� | ��ˮ��NaOH��Ӧ | D�� | ��ˮ��п��Ӧ����H2 |
 ijѧ��������֪���ʵ���Ũ�ȵ��������ζ�δ֪���ʵ���Ũ�ȵ�����������Һʱ��ѡ���̪��ָʾ��������д���пհף�
ijѧ��������֪���ʵ���Ũ�ȵ��������ζ�δ֪���ʵ���Ũ�ȵ�����������Һʱ��ѡ���̪��ָʾ��������д���пհף���1���ñ���������Һ�ζ����������������Һʱ�����ְ�����ʽ�ζ��ܵĻ���������ҡ����ƿ���۾�ע����ƿ����Һ��ɫ�ı仯��ֱ�������һ�α�Һ��ʹ��Һ�ɺ�ɫ��Ϊ��ɫ����30s�ڲ��ָ���
�� 2������˵������ȷ����A
A���ζ�����Ҫ������ȷ�طų�һ���������Һ
B��������Һ��ǿ��������ҺӦ��ѡ���ʽ�ζ���
C���ζ�����ϴ�ķ����Ǵ��Ͽڽ���Ҫʢװ����Һװ�����ڴ��¿ڽ�Һ�����Ԥ�õ��ձ���
D���ų���ʽ�ζ��ܼ��첿�����ݵķ����Ǵ������ٷ�Һ�Ը�������
E����ʵ���д��������������Һ��Ҫ����Ͳ����ȡ
��3�����в����п���ʹ��������������Һ��Ũ����ֵƫ�͵���D
A����ʽ�ζ���δ�ñ�������Һ��ϴ��ֱ��ע���������Һ
B���ζ�ǰʢ������������Һ����ƿ������ˮϴ����û�и���
C����ʽ�ζ����ڵζ�ǰ�����ݣ��ζ���������ʧ
D����ȡ�������ʱ����ʼ���Ӷ������ζ�����ʱ���Ӷ���
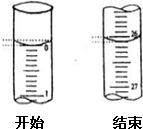
��4�����ζ���ʼ�ͽ���ʱ����ʽ�ζ����е�Һ����ͼ��ʾ���뽫���������������Ŀհ״���
| �ζ����� | ��������������Һ�����/mL | 0.1000mol/L����������mL��] | ||
| �ζ�ǰ�̶� | �ζ���̶� | ��Һ���/mL | ||
| ��һ�� | 25.00 | |||
| �ڶ��� | 25.00 | 1.56 | 28.08 | 26.52 |
| ������ | 25.00 | 0.22 | 26.34 | 26.12 |
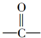 ��HCN��Ӧ���룺
��HCN��Ӧ���룺 $��_{һ������}^{HCN}$
$��_{һ������}^{HCN}$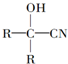
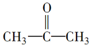 $��_{һ��������}^{HCN}$A$\underset{\stackrel{{H}_{2}O��{H}^{+}}{��}}{��}$B$��_{��}^{ŨH_{2}SO_{4}����}$C$��_{������}^{�ۺ�}$D
$��_{һ��������}^{HCN}$A$\underset{\stackrel{{H}_{2}O��{H}^{+}}{��}}{��}$B$��_{��}^{ŨH_{2}SO_{4}����}$C$��_{������}^{�ۺ�}$D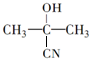 ��D
��D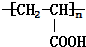 ��
��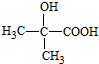 $��_{��}^{Ũ����}$CH2=CHCOOH+H2O��
$��_{��}^{Ũ����}$CH2=CHCOOH+H2O��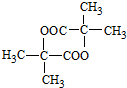 ��
��