��Ŀ����
��һ����CO��CO2��H2��ˮ��������������ɵĻ�����壬Ҫ֤�����к���CO��H2����ѡ�õ�������ҩƷ���£�
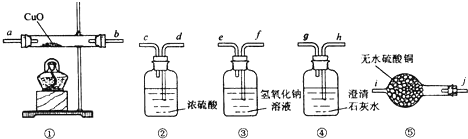
��1��װ�â��е�NaOH��Һ��Ũ��Ϊ2mol/L�����Ϊ300mL��Ҫ���Ƹ���Һ��Ӧѡ������ƿ�Ĺ��Ϊ ��ѡ��100mL��250mL��500mL��1000mL������ҪNaOH���������Ϊ g��
��2��ʵ��ʱ������װ�õ�˳���� �� ���١� �� ������ţ���
��3��װ�â��ڿ�ʼ����ʱ����ȷ������ ������ĸ����ͬ����ʵ�����ʱ����ȷ������ ��
A���ȼ��ȣ���ͨ����
B����ͨ���壬�����
C����ֹͣ���ȣ���ֹͣͨ����
D����ֹͣͨ���壬��ֹͣ����
��4��֤����������к���CO��ʵ�������� ��֤����������к���H2��ʵ�������� ��
��5��װ�â��з�����Ӧ�Ļ�ѧ����ʽΪ ��д������һ������

��1��װ�â��е�NaOH��Һ��Ũ��Ϊ2mol/L�����Ϊ300mL��Ҫ���Ƹ���Һ��Ӧѡ������ƿ�Ĺ��Ϊ
��2��ʵ��ʱ������װ�õ�˳����
��3��װ�â��ڿ�ʼ����ʱ����ȷ������
A���ȼ��ȣ���ͨ����
B����ͨ���壬�����
C����ֹͣ���ȣ���ֹͣͨ����
D����ֹͣͨ���壬��ֹͣ����
��4��֤����������к���CO��ʵ��������
��5��װ�â��з�����Ӧ�Ļ�ѧ����ʽΪ
���㣺��������ļ���
ר�⣺
��������1������ƿֻ��һ���̶��ߣ�ֻ���������������Ӧ����Һ�����������m=CVM�����㣻
��2��֤������һ����̼����������Ҫ�ȳ�ȥ������̼��ˮ��������ȥ������̼���Լ�������������Һ����ȥˮ�������Լ���Ũ������һ����̼�������ļ�����Ҫ���ݷ�Ӧ����������飬������ͨ�����ȵ�����ͭ��Ӧ���ɶ�����̼��ˮ��������ͨ����ˮ����ͭ����ˮ���������ɣ���ͨ�����ʯ��ˮ���������̼�Ĵ��ڣ�
��3����ʼʱ��Ҫ�ӷ�ֹ����ͻ���ﱬը�ĽǶ�������������ʱ��Ҫ��ֹ�����е��������룬�����ɵ�ͭ�ָ������ĽǶ������ǣ�
��4���������������CO����ͨ������ͭ������ɶ�����̼���������������������������ˮ��
��5��
��2��֤������һ����̼����������Ҫ�ȳ�ȥ������̼��ˮ��������ȥ������̼���Լ�������������Һ����ȥˮ�������Լ���Ũ������һ����̼�������ļ�����Ҫ���ݷ�Ӧ����������飬������ͨ�����ȵ�����ͭ��Ӧ���ɶ�����̼��ˮ��������ͨ����ˮ����ͭ����ˮ���������ɣ���ͨ�����ʯ��ˮ���������̼�Ĵ��ڣ�
��3����ʼʱ��Ҫ�ӷ�ֹ����ͻ���ﱬը�ĽǶ�������������ʱ��Ҫ��ֹ�����е��������룬�����ɵ�ͭ�ָ������ĽǶ������ǣ�
��4���������������CO����ͨ������ͭ������ɶ�����̼���������������������������ˮ��
��5��
���
�⣺��1������ƿֻ��һ���̶��ߣ�ֻ���������������Ӧ����Һ�������������300mL����ƿ��������300mL��Һ�����ݴ������ԭ��Ӧѡ��500mL����ƿ������ѡ��500mL����ƿ�������Ƴ�500mL��Һ������Ҫ��NaOH������m=CVM=2mol/L��0.5L��40g/mol=40g���ʴ�Ϊ��500mL��40��
��2��ʵ��ʱ�����ܽӿڵ�˳������������������Һ��ȥ������̼����Ũ�����ȥˮ����������һ����̼������������ͭ��Ӧ���ɵIJ��������̼��ˮ��������һ����̼�������Ĵ��ڣ������ø��������ˮ����ͭ����ˮ�Ĵ��ڣ�Ȼ�����ó���ʯ��ˮ���������̼�Ĵ��ڣ��ʴ�Ϊ���ۢڢݢܣ�
��3����ʼʱ��Ҫ�ӷ�ֹ����ͻ���ﱬը����Ӧ��ͨ���壬��װ���еĿ����ų���Ȼ���ټ��ȣ���ѡB������ʱ��Ҫ��ֹ�����е��������룬�����ɵ�ͭ�ָ���������Ӧ��Ϩ�ƣ���ͭ��ȴ����ֹͣͨ�����壬��C���ʴ�Ϊ��B��C��
��4��֤����������к���CO��ʵ�������Ǣ��г��ֻ��ǣ�֤����������к���������ʵ�������Ǣ��е���ˮ����ͭ�������ʴ�Ϊ�����г��ֻ��ǣ����е���ˮ����ͭ������
��5��װ�â��ڷ�����Ӧ�Ļ�ѧ����ʽ��һ����̼������ͭ��Ӧ���ɶ�����̼��ͭ������������ͭ��Ӧ����ͭ��ˮ��������Ӧ�Ļ�ѧ����ʽΪ��
CuO+CO�TCu+CO2 ��CuO+H2�TCu+H2O���ʴ�Ϊ��CuO+CO�TCu+CO2 ��CuO+H2�TCu+H2O��
��2��ʵ��ʱ�����ܽӿڵ�˳������������������Һ��ȥ������̼����Ũ�����ȥˮ����������һ����̼������������ͭ��Ӧ���ɵIJ��������̼��ˮ��������һ����̼�������Ĵ��ڣ������ø��������ˮ����ͭ����ˮ�Ĵ��ڣ�Ȼ�����ó���ʯ��ˮ���������̼�Ĵ��ڣ��ʴ�Ϊ���ۢڢݢܣ�
��3����ʼʱ��Ҫ�ӷ�ֹ����ͻ���ﱬը����Ӧ��ͨ���壬��װ���еĿ����ų���Ȼ���ټ��ȣ���ѡB������ʱ��Ҫ��ֹ�����е��������룬�����ɵ�ͭ�ָ���������Ӧ��Ϩ�ƣ���ͭ��ȴ����ֹͣͨ�����壬��C���ʴ�Ϊ��B��C��
��4��֤����������к���CO��ʵ�������Ǣ��г��ֻ��ǣ�֤����������к���������ʵ�������Ǣ��е���ˮ����ͭ�������ʴ�Ϊ�����г��ֻ��ǣ����е���ˮ����ͭ������
��5��װ�â��ڷ�����Ӧ�Ļ�ѧ����ʽ��һ����̼������ͭ��Ӧ���ɶ�����̼��ͭ������������ͭ��Ӧ����ͭ��ˮ��������Ӧ�Ļ�ѧ����ʽΪ��
CuO+CO�TCu+CO2 ��CuO+H2�TCu+H2O���ʴ�Ϊ��CuO+CO�TCu+CO2 ��CuO+H2�TCu+H2O��
���������⿼���˳�������ļ���ͳ��ӣ���Ҫ�ǻ���������ʱ��Ҫ��ȥ�������壬��������Ҫ����������������ʵ����̣�����������������жϣ�

��ϰ��ϵ�д�
�����Ŀ
��������Һ�м���Ba��OH��2��Һ�����ɳ��������ʵ���������Ba��OH��2�����ʵ����Ĺ�ϵ��ͼ��ʾ��������ȷ���ǣ�������
A��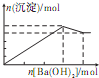 |
B��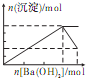 |
C��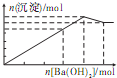 |
D��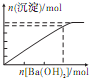 |
�����й�ʵ���������ȷ���ǣ�������
| A������AlCl3��Һ�ɵô�������ˮ�Ȼ��� |
| B���ü�ʽ�ζ�����ȡ20.00mL���Ը��������Һ |
| C����pH��ֽ�ⶨ��Һ��pHʱԤ��Ҫ������ˮʪ�� |
| D��ʵ��������SnCl2 ��Һʱ����Ҫ�������������� |
ijͬѧ���������ʵ��̽��ijЩ�ǽ���Ԫ�ؼ��仯��������ʣ������ܴﵽĿ���ǣ�������
| A���ⶨ��ͬŨ��HX��X=F��Cl��Br��I����Һ��pH�Ƚ�±��Ԫ�صķǽ����� |
| B����������CuSO4?5H2O���������Ũ�����У���֤Ũ�������ˮ�� |
| C����CO2ͨ��ˮ�����У��Ƚ�̼������������ǿ�� |
| D������Ƭ�����ȵ�Ũ�����У���֤����Ũ�����з����ۻ� |