��Ŀ����
5����ij��ѧ�Ļ�ѧʵ����ҩƷ���У���һ��װ��Na2O2�Լ������ޣ�ʹ�ö��꣬�⼣�߰ߣ�ƿ���а�ɫ���壬�����н�飮��1��Χ��������ʵ�������Ļ�ѧʵ�����飬���һ���м�ֵ��̽�����⣨�����⣩̽���Լ��Ƿ���ʣ�
��2��ij��ѧ��ȤС���ƿ�ڹ������ɫ�����жϣ����Լ��Ѿ����ʣ�������ƿ���а�ɫ���壬��Na2O2Ϊ����ɫ���壮
��3����������������жϺ���ȤС��������������ַ������ж����о�������
 ����Ʒ��Na2O2�ĺ�����
����Ʒ��Na2O2�ĺ���������1����������
ͨ������Na2O2��ˮ��Ӧ�ų�O2�������������Ʒ��Na2O2�ĺ�����
��������Ϊ1��������1��ע���� �������װж���������������ã���ʵ������ͼ���£�

�������㡱��ָ����ע�����ݻ�ȷ��m��Ʒ�����ֵ�����Թ�����Ʒ�������
���������ʵ������ͼ��д�� A��C��ʵ�����������B�IJ������裺
A��������B����ˮ���ý���Ѹ�ٶ�ס��ͷ��C��������
����2--������
������Ʒ�к���Ԫ�ص�����ֻ��Na2O2��Na2CO3��ʵ�������ͼ��
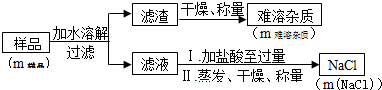
������ʵ�鲽���У���Ҫ�õ��������IJ������ܽ⡢���˻������������ֲ�����������
������Һ�м�ϡ��������������Ŀ����ʹ��Һ�е�NaOH��Na2CO3���ѧʽ����ȫת��ΪNaCl��
��ʵ������������������еμ�ϡ���ᣬ�������ܽ⣬������Һ�ʻ�ɫ��˵����Ʒ�е���������ΪFe2O3��
�ܸ�С֯ʵ�����ݼ�¼�����
| m��Ʒ | m�������� | m��NaCl�� |
| 8.00g | 0.42g | 10.53g |
��4���������ֲⶨNa2O2�����ķ����У�����1���1����2�����IJ��������ȷ��
���� ��1�������ṩ��ҩƷ�������ѧ��ѧ��ѧ���ݣ�����̽�������ڵ��Լ��Ƿ���ʣ�
��2���������ʵ���ɫ�ɳ����ƶ����ʵ����ࣻ
��3������1������������ԭ����֪����Ҫȷ��ȡһ��������Ʒ������ע�����У�����ע����������������ˮ�������ý�����ס��ͷ����ֹ�����������ݳ���ʹ����������ˮ��Ӧ��ƽ��ע������ʹע��������ѹǿһ�£�ȷ����������ע�����ϵĶ�����֪������������ݴ˽��м��㣻
����2������Ʒ�ܽ⡢���˵���Һ������������Ϊ���������ʣ��������ʵ�������֪��Ʒ��Na2O2��Na2CO3����������Һ��Ϊ�������ƺ�̼���ƣ������������ᷴӦ���Ȼ��ƣ������������������Ȼ��Ƶ����������������Ȼ��Ƶ���������Ʒ��Na2O2��Na2CO3�������з��������ù������Ƶ�����������һ�������ܽ⡢���ˡ��������õ���������
��4������2����������Ʒ�к���Ԫ�ص�����ֻ��Na2O2��Na2CO3������ʵ�ϲ�һ������������ɣ�Ҳ�����������ƻ�̼�����Ƶȣ��������Щ���ʣ�����������з���������������Ƶ������Ͳ�ȷ�ˣ��ݴ˴��⣮
��� �⣺��1�������ṩ��ҩƷ�������ѧ��ѧ��ѧ���ݣ�����̽�������ڵ��Լ��Ƿ���ʣ��ʴ�Ϊ��̽���Լ��Ƿ���ʣ�
��2����������Ϊ����ɫ��ĩ�����ƿ�ڹ������ɫΪ��ɫ����ɳ���˵�����������Ѿ����ʣ��ʴ�Ϊ�����ƣ�
��3������1������������ԭ����֪����Ҫȷ��ȡһ��������Ʒ������ע�����У�����ע����������������ˮ�������ý�����ס��ͷ����ֹ�����������ݳ���ʹ����������ˮ��Ӧ��ƽ��ע������ʹע��������ѹǿһ�£�ȷ����������ע�����ϵĶ�����֪������������ݴ˽��м��㣬����A����Ϊ������B����Ϊ�ý���Ѹ�ٶ�ס��ͷ��C����Ϊ������
�ʴ�Ϊ��������������������
����2������Ʒ�ܽ⡢���˵���Һ������������Ϊ���������ʣ��������ʵ�������֪��Ʒ��Na2O2��Na2CO3����������Һ��Ϊ�������ƺ�̼���ƣ������������ᷴӦ���Ȼ��ƣ������������������Ȼ��Ƶ����������������Ȼ��Ƶ���������Ʒ��Na2O2��Na2CO3�������з��������ù������Ƶ�������
������һ�������ܽ⡢���ˡ��������õ���������
�ʴ�Ϊ���ܽ⣻���ˡ�������
����Һ��Ϊ�������ƺ�̼���ƣ�����Һ�м�ϡ��������������Ŀ����ʹ��Һ�е�NaOH��Na2CO3��ȫת��ΪNaCl���ʴ�Ϊ��NaOH��Na2CO3��
�������������еμ�ϡ���ᣬ�������ܽ⣬������Һ�ʻ�ɫ��˵����Ʒ�е���������ΪFe2O3���ʴ�Ϊ��Fe2O3��
�ܸ��ݱ��е����ݿ�֪��Ʒ��Na2O2��Na2CO3������Ϊ8.00g-0.42g=7.58g������Ʒ��Na2O2�����ʵ���Ϊxmol��Na2CO3�����ʵ���Ϊymol����$\left\{\begin{array}{l}{2��x+y����58.5=10.53}\\{78x+106y=7.58}\end{array}\right.$�����x=0.068��y=0.021������Na2O2����������Ϊ$\frac{78��0.068}{8}$��100%=66.3%��
�ʴ�Ϊ��66.3%��
��4������2����������Ʒ�к���Ԫ�ص�����ֻ��Na2O2��Na2CO3������ʵ�ϲ�һ������������ɣ�Ҳ�����������ƻ�̼�����Ƶȣ��������Щ���ʣ�����������з���������������Ƶ������Ͳ�ȷ�ˣ�������1��ֻ�й���������ˮ��Ӧ�������������Է���1�IJ��������ȷ��
�ʴ�Ϊ��1��
���� ���⿼����������ɵĶ���ʵ�飬ʵ����̵���Ʒ���������ʵ����֤����Ʒ����������жϣ����ջ����ǹؼ�����Ŀ�Ѷ��еȣ�

| A�� | AlCl3��Һ�м��������ˮ��Al3++4NH3•H2O�TAlO${\;}_{2}^{-}$+4NH${\;}_{4}^{+}$+2H2O | |
| B�� | �ں����ҵĽ���Һ������I-���еμ�H2O2�õ�I2��2I-+H2O2+2H+�TI2+O2��+2H2O | |
| C�� | ������������Һ�еμ����������Һ��PH=7��Ba2++2OH-+2H++SO42-�TBaSO4��+2H2O | |
| D�� | ��̼������Һ���ݹ�¯ˮ����Ca2++CO32-�TCaCO3�� |
| A�� | �������ڶ�ͯ���ݵ����� | |
| B�� | �������ڹ����������ͷ��� | |
| C�� | �������ڻ�����Ca10��PO4��6��OH��2������ | |
| D�� | ��ʹ��ͯ������ |
��2NO2?N2O4 ������ӦΪ���ȷ�Ӧ��
��C��s��+CO2?2CO ������ӦΪ���ȷ�Ӧ��
��N2+3H2?2NH3 ������ӦΪ���ȷ�Ӧ��
��H2S?H2+S��s��������ӦΪ���ȷ�Ӧ��
| A�� | �٢� | B�� | �� | C�� | �٢� | D�� | �ڢ� |
��1��β���е�CO��Ҫ���������͵IJ���ȫȼ�գ�
���������밴���з�Ӧ��ȥCO��2CO��g���T2C��s��+O2��g����H=+221kJ•mol-1�������������ܷ�ʵ�ַ���ǡ��������ǣ��÷�Ӧ���������ؼ��ķ�Ӧ���κ��¶��¾������Է����У�
���о���������ӦCO��g��+H2O��g��?H2��g��+CO2��g��ƽ�ⳣ�����¶ȵı仯���±���ʾ��
| �¶�/�� | 400 | 500 | 800 |
| ƽ�ⳣ��K | 9.94 | 9 | 1 |
��2����CO��ȼ�ϵ�ص��NaCl��Һ��FeCl3��FeCl2���Һ��ʾ��ͼ��ͼ1��ʾ������A��B��D��Ϊʯī�缫��CΪͭ�缫��ת��0.4mol e-�Ͽ�K��
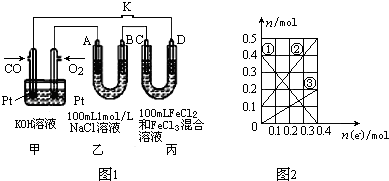
�����в����������ڱ�״���µ����Ϊ7.28L��
�ڱ�װ����Һ�н��������ӵ����ʵ�����ת�Ƶ��ӵ����ʵ����仯��ϵ��ͼ2��ʾ����Ӧ������Ҫʹ��װ���н���������ǡ����ȫ��������Ҫ280mL 5.0mol•L-1 NaOH��Һ��
��3����ⷨ����������������Ŀǰ������Ⱦ������һ����˼·��ԭ���ǽ�NOx�ڵ����зֽ������Ⱦ��N2��O2��ȥ����ͼ2��ʾ�����缫���ǹ������������ʣ���һ�������¿����ɴ���O2-������������ӦΪ2NOx+4xe-=N2+2xO2-��
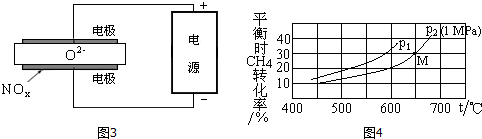
��4��β���е�̼�⻯�������飬���������Ʊ��������䷴Ӧ����ʽΪ��
CH4��g��+H2O��g��?CO��g��+3H2��g����H=+206.2kJ/mol
[����Ͷ�ϱ�n��CH4����n��H2O��=1��1]��
�������෴Ӧ����ij��֣�B����ƽ��ѹǿ��PB���������ʵ���Ũ�ȣ�cB��Ҳ�ɱ�ʾƽ�ⳣ��������KP����ѹ=��ѹ�����ʵ�����������ƽ��ʱCH4��ת�������¶ȡ�ѹǿ����ѹ���Ĺ�ϵ��ͼ4��ʾ��
��p1�� p2 ���������������p2ʱM���ƽ�ⳣ��KP=0.066��С�������3λ����
| A�� | X2+��XO4-�Ļ�ԭ���� | |
| B�� | ��Һ�пɷ�����Z2+2A2+�T2A3++2Z- | |
| C�� | ������ǿ����˳��Ϊ��XO4-��B2��Z2��A3+ | |
| D�� | Z2�ڢ������������������������ |