��Ŀ����
19��������³����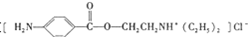 ����һ�־ֲ����������������ýϿ졢��ǿ�����Խϵͣ���ϳ�·�����£�
����һ�־ֲ����������������ýϿ졢��ǿ�����Խϵͣ���ϳ�·�����£�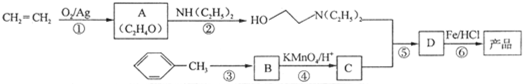
��֪����
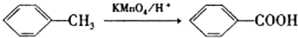
��
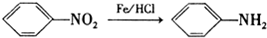
��
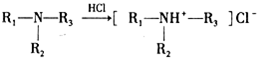
��ش��������⣺
��1��A�ĺ˴Ź�������ֻ��һ���壬��A�Ľṹ��ʽΪ
 ��
����2��C�Ľṹ��ʽΪ
 ��C�к��еĺ��������ŵ�����Ϊ�������Ȼ���
��C�к��еĺ��������ŵ�����Ϊ�������Ȼ�����3����Ӧ�ݵĻ�ѧ��Ӧ����ʽΪ
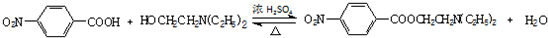 ��
����4��B��ij��ͬϵ��E����Է���������B��28������-NO2�뱽��ֱ����������E�Ľṹ��ʽΪ
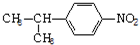 ����дһ�֣���
����дһ�֣�����5����������������E��ͬ���칹�����Ŀ��30�֣�
�ٽṹ�к���һ����-NH2����һ����-COOH��
�ڱ�����������������ͬ��ȡ������
���� ��ϩ����������Ӧ����A��A�ĺ˴Ź�������ֻ��һ���壬��A�Ľṹ��ʽΪ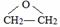 ��D��Fe/HCl�����·�����Ӧ���ɲ�Ʒ��D�ṹ��ʽΪ
��D��Fe/HCl�����·�����Ӧ���ɲ�Ʒ��D�ṹ��ʽΪ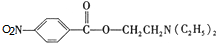 �����ݷ�Ӧ��֪��C�Ľṹ��ʽΪ
�����ݷ�Ӧ��֪��C�Ľṹ��ʽΪ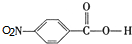 ��B����������C����������Ϣ֪��B�ṹ��ʽΪ
��B����������C����������Ϣ֪��B�ṹ��ʽΪ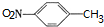 ���ױ�������Ӧ���ɶԼ�����������Ӧ��Ϊȡ����Ӧ���ݴ˷������
���ױ�������Ӧ���ɶԼ�����������Ӧ��Ϊȡ����Ӧ���ݴ˷������
��� �⣺��ϩ����������Ӧ����A��A�ĺ˴Ź�������ֻ��һ���壬��A�Ľṹ��ʽΪ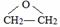 ��D��Fe/HCl�����·�����Ӧ���ɲ�Ʒ��D�ṹ��ʽΪ
��D��Fe/HCl�����·�����Ӧ���ɲ�Ʒ��D�ṹ��ʽΪ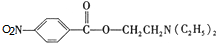 �����ݷ�Ӧ��֪��C�Ľṹ��ʽΪ
�����ݷ�Ӧ��֪��C�Ľṹ��ʽΪ ��B����������C����������Ϣ֪��B�ṹ��ʽΪ
��B����������C����������Ϣ֪��B�ṹ��ʽΪ ���ױ�������Ӧ���ɶԼ�����������Ӧ��Ϊȡ����Ӧ��
���ױ�������Ӧ���ɶԼ�����������Ӧ��Ϊȡ����Ӧ��
��1��A�ĺ˴Ź�������ֻ��һ���壬��A�Ľṹ��ʽΪ ��
��
�ʴ�Ϊ�� ��
��
��2��C�Ľṹ��ʽΪ ��C�к��еĺ��������ŵ�����Ϊ�������Ȼ���
��C�к��еĺ��������ŵ�����Ϊ�������Ȼ���
�ʴ�Ϊ�� ���������Ȼ���
���������Ȼ���
��3����Ӧ�ݵĻ�ѧ��Ӧ����ʽΪ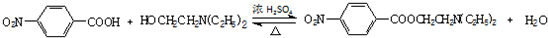 ���ʴ�Ϊ��
���ʴ�Ϊ��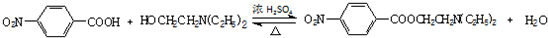 ��
��
��4��B��ij��ͬϵ��E����Է���������B��28��˵��E��B��2��̼ԭ�ӣ�����-NO2�뱽��ֱ����������E�Ľṹ��ʽΪ ����дһ�֣���
����дһ�֣���
�ʴ�Ϊ�� ��
��
��5��E��ͬ���칹����������������ٽṹ�к���һ����-NH2 ����һ����-COOH�����ڱ�����������������ͬ��ȡ�������������-NH2��-COOH��-CH2CH3�����-NH2��-COOH���ڣ���������ͬ���칹�壬���-NH2��-COOH��䣬��������ͬ���칹�壬���-NH2��-COOH���������ͬ���칹�壬����10��ͬ���칹�壻
�������-NH2��-CH2COOH��-CH3�����-NH2��-CH2COOH����������ͬ���칹�壬���-NH2��-CH2COOH���������ͬ���칹�壬���-NH2��-CH2COOH�����2��ͬ���칹�壬����10�֣�
�������-CH2NH2��-COOH��-CH3�����-CH2NH2��-COOH����������ͬ���칹�壬���-CH2NH2��-COOH���������ͬ���칹�壬���-CH2NH2��-COOH���������ͬ���칹�壬���Թ���10�֣�
����������Ĺ���30��ͬ���칹�壻
�ʴ�Ϊ��30��
���� ���⿼���л����ƶϣ����ؿ���ѧ�������жϡ�֪ʶǨ�ơ���ȡ��Ϣ������Ϣ���������ݲ�Ʒ�ṹ����Ŀ��Ϣ���������ϵķ��������ƶϣ��ѵ��ǣ�5��ͬ���칹�������жϣ�Ҫ����λ�ýṹ��̼���칹���������칹��ע�ⰱ���ᷢ�����۷�Ӧ�ϼ��ͳɼ�λ�ã���Щ���Ǹ߿��ȵ���ѵ㣮

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�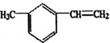 ����˵����ȷ���ǣ�������
����˵����ȷ���ǣ�������| A�� | ����������̼ԭ�ӿ��ܴ���ͬһƽ���� | |
| B�� | ������������7��̼ԭ��һ������ͬһƽ���� | |
| C�� | ����������ԭ���п��ܴ���ͬһƽ���� | |
| D�� | ������������16��ԭ�Ӵ���ͬһƽ���� |
| A�� |  ����������ζ | B�� |  ���ű���ʳ��ˮ���ռ����� | ||
| C�� |  ʢװҺ�ȵ����� | D�� |  ֤������������NaOH��Һ��Ӧ |
| �� | �� | �� | �� | �� | �� | �� | �� | |
| ԭ�Ӱ뾶��10-10m�� | 0.74 | 1.60 | 1.52 | 1.10 | 0.99 | 1.86 | 0.75 | 0.82 |
| �����ͻ��ϼ� | +2 | +1 | +5 | +7 | +1 | +5 | +3 | |
| -2 | -3 | -1 | -3 |
��1������Ԫ�����ڱ��е�λ���ǵڶ�����VIA��д��������������Ӧˮ������ݵ��⻯��ˮ��Һ��Ӧ�����ӷ���ʽ��Mg��OH��2+2H+=Mg2++2H2O��
��2��������������ˮ�����У�������ǿ�Ļ�����ķ���ʽ��HClO4��������ǿ�Ļ������������ӻ��������ӡ����ۡ�����
��3���ȽϢܺ͢ߵ��⻯����ȶ��ԣ��û�ѧʽ��ʾ��NH3��PH3��
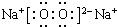 �����к��еĻ�ѧ�������Ӽ������ۼ���
�����к��еĻ�ѧ�������Ӽ������ۼ���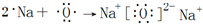 ��
��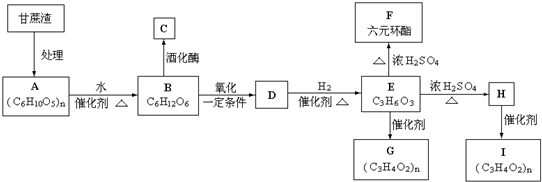
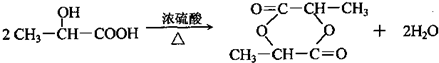


 ���� ��̼ԭ�Ӹ�����1��֧����ȡ������
���� ��̼ԭ�Ӹ�����1��֧����ȡ������ ��
��