��Ŀ����
7���������ʳ�ṹ���Ա������彡������ij������Ʒ�İ�װ��ӡ��������ϣ���ѡ���⡢���ǡ����ۡ��������ƣ����и��������ʵ��Ǿ�ѡ���⣬���ڷ����������������ƣ����������������յ�ˮ������������ǣ����ǵĻ�ѧʽ��C12H22O11��
����Ȼ��֬ˮ���Ĺ�ͬ�����Ǹ��ͣ�
���г������۵��㳦����ˮѼ��ե�ˡ������ɵ�ʳƷ����������հ�װ����հ�װ��Ŀ���Ƿ�ֹʳƷ�����������������ʣ�
���� �پ�ѡ�����и��������ʣ��������ƾ��з������ã������Ƕ���ˮ������ղ����������ǣ����ǵ���Ҫ�ɷ�Ϊ���ǣ����������и��ɷֵ���ɺ����ʺ���;�жϽ��
����Ȼ��֬ˮ��IJ���Ϊ��֬������ͣ�
�۸��ݿ����еijɷּ������������⣮
��� �⣺�����ݻ�����Ʒ�İ�װ��ӡ��������Ͽ�֪��ѡ�����и��������ʣ��������ƾ��з������ã������Ƕ��ǣ�ˮ������ղ����������ǣ����ǵ���Ҫ�ɷ�Ϊ���ǣ���ѧʽΪ��C12H22O11��
�ʴ�Ϊ����ѡ���⡢�������ơ������ǡ�C12H22O11��
����Ȼ��֬ˮ��IJ���Ϊ��֬������ͣ��ʹ�ͬ����Ϊ���ͣ�
�ʴ�Ϊ�����ͣ�
�ۿ����е��������������ԣ��������������Է�ֹʳ����������������ʣ�
�ʴ�Ϊ��������
���� ���⿼�鵰���ʡ���������ʣ���֬���Լ�������ʳƷ���Ӽ��ȣ��ѶȲ���ע�����֪ʶ�Ļ��ۣ�

��ϰ��ϵ�д�
�����Ŀ
14�������£�������Һ�и�����Ũ�ȹ�ϵ��ȷ���ǣ�������
| A�� | ������Һ��NaOH��Һ���Ϻ�������Һ�����ԣ�c��Na+����c��CH3COO-�� | |
| B�� | Ũ��Ϊ0.1 mol•L-1��̼������Һ��c��Na+��=2c��CO32-��+c��HCO3-��+c��H2CO3�� | |
| C�� | pH=12�İ�ˮ��Һ��pH=2������������ϣ�c��Cl-����c��NH4+����c��H+����c��OH-�� | |
| D�� | �����ʵ����İ�ˮ�������Ϻ����Һ��c��H+��=c��OH-��+c��NH3•H2O�� |
15�������£����и���������ָ����Һ��һ���ܴ���������ǣ�������
| A�� | $\frac{{K}_{W}}{c{��H}^{+}��}$=10-13 mol•L-1����Һ�У�NH4+��Ca2+��Cl-��NO3- | |
| B�� | ����0.1 mol•L-1 Fe3+����Һ��Na+��K+��SCN-��NO3- | |
| C�� | ��ʹ���۵⻯����ֽ����ɫ����Һ��K+��SO42-��S2-��SO32- | |
| D�� | ˮ�����c��H+��=1��10-13mol•L-1����Һ�У�K+��Na+��AlO2-��CO32- |
12����������һˮ�ϰ��ĵ���ƽ�⼸��������Ӱ����ǣ�������
| A�� | 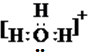 | B�� |  | C�� |  | D�� |  |
16�����������ӳɷ�Ӧ�������������ǣ�������
| A�� | ��ϩʹ������Ȼ�̼��Һ��ɫ | |
| B�� | NO2ͨ��ˮ�У�����ɫ����ȥ | |
| C�� | ����������Һ�еμ����������ɫ���� | |
| D�� | ����������ڹ����·�Ӧ������ɫ����ȥ |
 2SO2��g��+O2��g��?2SO3��g����Ӧ���̵������仯��ͼ��ʾ����֪1mol SO2��g�� �ڷ�Ӧ�б�����Ϊ1mol SO3��g�� �ġ�H=-99kJ/mol���ش��������⣺
2SO2��g��+O2��g��?2SO3��g����Ӧ���̵������仯��ͼ��ʾ����֪1mol SO2��g�� �ڷ�Ӧ�б�����Ϊ1mol SO3��g�� �ġ�H=-99kJ/mol���ش��������⣺