��Ŀ����
�����£�0.1 mol/L��ˮ��Һ��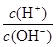 ��1��10-8�����������������
��1��10-8�����������������
A������Һ�������ӵ�Ũ�ȣ�c(H+ ) �� 1��10-9 mol/L
B��0.1 mol/L��ˮ��Һ��0.1 mol/L HCl��Һ�������Ϻ�������Һ�У�
c(NH4+ ) + c(H+ ) �� c(Cl- ) + c(OH-)
C��0.1 mol/L�İ�ˮ��Һ��0.05 mol/L H2SO4��Һ�������Ϻ�������Һ�У�
c(NH4+ ) + c(NH3) + c(NH3��H2O) �� 2c(SO42-)
D��Ũ�Ⱦ�Ϊ0.1 mol/L��NH3��H2O��NH4Cl��Һ�������Ϻ�����Һ�ʼ��ԣ���
c(NH4+ )�� c(NH3��H2O) �� c(Cl-) �� c(OH-) �� c(H+ )
AD
��������
��������������£�0.1
mol/L��ˮ��Һ��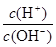 ��1��10-8������Һ��c(OH-)��0.001mol/L������Һ�������ӵ�Ũ�ȣ�c(H+ ) �� 1��10-11 mol/L��A����ȷ��B��ȷ�����ŵ���غ㣻C��ǡ����������泥�����������غ��֪ѡ��C��ȷ��D����Һ�ʼ��ԣ�˵����ˮ�ĵ���̶ȴ���NH4+��ˮ��̶ȣ�����Ӧ����c(NH4+
)�� c(Cl-)
�� c(NH3��H2O)
��c(OH-) �� c(H+ )��D����ȷ����ѡAD��
��1��10-8������Һ��c(OH-)��0.001mol/L������Һ�������ӵ�Ũ�ȣ�c(H+ ) �� 1��10-11 mol/L��A����ȷ��B��ȷ�����ŵ���غ㣻C��ǡ����������泥�����������غ��֪ѡ��C��ȷ��D����Һ�ʼ��ԣ�˵����ˮ�ĵ���̶ȴ���NH4+��ˮ��̶ȣ�����Ӧ����c(NH4+
)�� c(Cl-)
�� c(NH3��H2O)
��c(OH-) �� c(H+ )��D����ȷ����ѡAD��
���㣺����������Ũ�ȵļ��㡢��������Ũ�ȴ�С�Ƚϵ��ж�
�����������Ǹ߿��еij������ͣ������е��Ѷȵ����⡣���������߿���������ǿ����ע�ض�ѧ������֪ʶ������ѵ����ͬʱ�����ض�ѧ����������������ⷽ����ָ����ѵ����ּ�ڿ���ѧ��������û���֪ʶ���ʵ�����������������������ѧ���������������ͷ�ɢ˼ά����������Ĺؼ������úü����غ��ϵ��������غ㡢�����غ��Լ������غ㡣
