��Ŀ����
6����������Ԫ�صķ�ˮ����ˮ����� �ɵ�ˮ�帻Ӫ������ȾԽ��Խ���أ�������Ҳһֱ��Ѱ�ұ�����ʩ������ר����Ϊ�����ý�������ˮ���е�$NO_3^-$����ԭΪN2���Ӷ�������Ⱦ���䷴Ӧ�����ӷ���ʽ�ǣ�6$NO_3^-$+10Al+18H2O��3N2��+10Al��OH��3+6OH-��1������ƽ�������ӷ���ʽ6NO3-+10Al+18H2O�T3N2��+10Al��OH��3+6OH-
��2�������ø÷�Ҫ��ȥ1m3����Ԫ��0.3mol�ķ�ˮ�е�$NO_3^-$���赪Ԫ�ض���$NO_3^-$����ʽ���ڣ���ͬ������������Ҫ���Ľ�����13.5g��
��3����ҵ��ˮ�е�NaNO2�ɲ��ü�������NH4Cl�ķ�����ȥ����ʹNaNO2ת��Ϊ�����������Ⱦ��N2����Ӧ�Ļ�ѧ����ʽΪNH4Cl+NaNO2=NaCl+N2��+2H2O��
��4��������Ϊ����þ�����ܸ�������������Ⱦ���䷴Ӧԭ���ͽ�������ͬ��
��д��þ�ͺ�����ˮ��Ӧ�����ӷ���ʽ2NO3-+5Mg+6H2O�TN2��+5Mg��OH��2+2OH-��
����֪����þ���ԴӺ�ˮ����ȡ��MgCl2ͨ������Ƶã��������������þ�Ļ�ѧ����ʽΪMgCl2$\frac{\underline{\;ͨ��\;}}{\;}$Mg+Cl2����
��Ҫ��ȥ1m3����Ԫ��0.3mol�ķ�ˮ�е�$NO_3^-$����������Ҫ��0.5%������������MgCl2�ĺ�ˮ14.25kg��
���� ��1��������Ӧ��Ԫ�ػ��ϼ۱仯���ҳ����ϼ����ߵĺͻ��ϼ۽��͵�Ԫ�أ�����������ԭ��Ӧ��ʧ�����غ���ɽ��ԭ�Ӹ����غ���ƽ����ʽ��
��2����Ӧ��NO3-ȫ������ԭ�����ݷ���ʽ�����������ʵ�����������������������
��3��ʹNaNO2ת��Ϊ�����������Ⱦ��N2��NԪ�صĻ��ϼ۽��ͣ��Ȼ������ԭ����
��4���ٽ���þ�����ܸ�������������Ⱦ����Ӧԭ���ͽ�������ͬ�����Բ���Ϊ������������þ�����������ӣ����ݵ���ת���غ���Ԫ���غ㡢����غ���ƽ��
�ڵ�������Ȼ�þ����þ�����������ݹ�ϵʽ5MgCl2��5Mg��2NO3-���㣮
��� �⣺��1��NO3-�е��Ļ��ϼ���+5�۽���Ϊ0�ۣ��仯5��Al�Ļ��ϼ���0������Ϊ+3�ۣ��仯3��Ҫʹ������ԭ��Ӧ��ʧ�����غ㣬������������ϵ��Ϊ6����ϵ��Ϊ10������ԭ�Ӹ����غ㷴Ӧ����ʽΪ��6NO3-+10Al+18H2O�T3N2��+10Al��OH��3+6OH-��
�ʴ�Ϊ��6NO3-+10Al+18H2O�T3N2��+10Al��OH��3+6OH-��
��2����Ϊ����0.3mol�ķ�ˮ�е�NO3-�����ʵ���Ϊ0.3mol�����ݻ�ѧ����ʽ6NO3-+10Al+18H2O�T3N2��+10Al��OH��3+6OH-��0.3mol��������������Ľ����������ʵ���Ϊ$\frac{0.3mol��10}{6}$=0.5mol��������Ҫ�������������ǣ�0.5mol��27g/mol=13.5g��
�ʴ�Ϊ��13.5��
��3��ʹNaNO2ת��Ϊ�����������Ⱦ��N2��NԪ�صĻ��ϼ۽��ͣ���ԭ��NH4Cl �е�Ԫ�ػ��ϼ�����Ϊ0�ۣ�������ӦΪNH4Cl+NaNO2=NaCl+N2��+2H2O��
�ʴ�Ϊ��NH4Cl+NaNO2=NaCl+N2��+2H2O��
��4���ٽ���þ�����ܸ�������������Ⱦ����Ӧԭ���ͽ�������ͬ�����Բ���Ϊ������������þ�����������ӣ�
��Ӧ���ӷ���ʽΪ2NO3-+5Mg+6H2O�TN2��+5Mg��OH��2+2OH-��
�ʴ�Ϊ��2NO3-+5Mg+6H2O�TN2��+5Mg��OH��2+2OH-����
�ڵ�������Ȼ�þ�Ļ�ѧ����ʽΪ��MgCl2$\frac{\underline{\;ͨ��\;}}{\;}$Mg+Cl2����
��������Ҫ��0.5%������������MgCl2�ĺ�ˮmkg����
5MgCl2��������5Mg��������2NO3-
5��95g 2mol
m��103g��0.5% 0.3mol
����$\frac{5��95g}{m��1{0}^{3}g��0.5%}$=$\frac{2mol}{0.3mol}$�����m=14.25��
�ʴ�Ϊ��MgCl2$\frac{\underline{\;ͨ��\;}}{\;}$Mg+Cl2����14.25��
���� ���⿼��������ԭ��Ӧ�ļ��㡢���ӷ���ʽ�ȣ��Ѷ��еȣ�����������÷���ʽ����ϵʽ���У��ȵ���ת���غ����ֱ�ۣ��������ѡ��

��1��3 ��2��3 ��1��7 ��2��7 ������ȣ�
| A�� | �ڢ� | B�� | ֻ�Т� | C�� | �ڢ� | D�� | �٢� |
����������ƽ����14.31g NaCl��
����100mL��Ͳ��ȡ4.5mLϡ���ᡡ
������ɫʯ����Һ������Һ������ԡ�
����pH��ֽ�����Һ��pHΪ5.60��
| A�� | �٢� | B�� | �ڢ� | C�� | �ۢ� | D�� | �٢ڢ� |
| A�� | 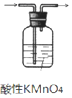 ��ͼ 1��ʾװ�ó�ȥ��ϩ������ SO2 ���� | |
| B�� |  ��ͼ װ����ȡ 20.00 mL 0.10 mol•L-1���� | |
| C�� | 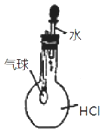 ��ͼ װ������֤ HCl ������ˮ�е��ܽ��� | |
| D�� | 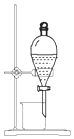 ��ͼ װ�ôӵ�� CCl4��Һ�з������ |
| A�� | CH3CH=CH2��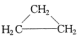 | B�� | ��Ȳ�ͱ� | C�� |  �� ��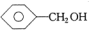 | D�� | ����ͼ������ |
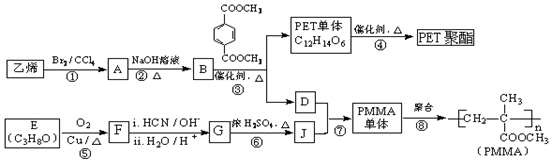
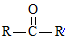 $��_{ii��H_{2}O/H+}^{i��HCN/OH-}$
$��_{ii��H_{2}O/H+}^{i��HCN/OH-}$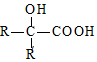 ��R��R�����������
��R��R����������� ��
�� ��
�� �������仯�����ڻ�ѧ�а�������Ҫ�Ľ�ɫ���Ը��ݸ����ʵ���������������⣺��1������ˮ��Ӧʱ����Ư����ˮ���ϣ���ͬѧ�ô���С�����佫0.46g�ư���������Ͷ��ˮ�У�ʹ�Ƴ���ˮ����ˮ����������Ӧ����Ӧ���������ɵ�����������ڣ�����ڡ���С�ڡ����ڡ��� 224mL ����״������
�������仯�����ڻ�ѧ�а�������Ҫ�Ľ�ɫ���Ը��ݸ����ʵ���������������⣺��1������ˮ��Ӧʱ����Ư����ˮ���ϣ���ͬѧ�ô���С�����佫0.46g�ư���������Ͷ��ˮ�У�ʹ�Ƴ���ˮ����ˮ����������Ӧ����Ӧ���������ɵ�����������ڣ�����ڡ���С�ڡ����ڡ��� 224mL ����״������ CuSO4+SO2��+2H2O������SO2�����ʵ���С��0.46mol��ԭ�������ŷ�Ӧ�Ľ��������Ũ����С��ͭ����ϡ���ᷴӦ�����Բ���SO2�����ʵ���С��0.46mol��
CuSO4+SO2��+2H2O������SO2�����ʵ���С��0.46mol��ԭ�������ŷ�Ӧ�Ľ��������Ũ����С��ͭ����ϡ���ᷴӦ�����Բ���SO2�����ʵ���С��0.46mol�� +CH3Br$\stackrel{����}{��}$
+CH3Br$\stackrel{����}{��}$