��Ŀ����
���������г��ù��������������ⶨ�����̵ĺ�������Ӧԭ��Ϊ��
2Mn2++5S2O82-+8H2O
2MnO4-+10SO42-+16H+
��1����̬��ԭ�ӵĺ�������Ų�ʽΪ ��
��2��������Ӧ�漰��Ԫ������ͬ����Ԫ�أ����һ�������ɴ�С��˳��Ϊ ����Ԫ�ط��ţ���
��3����֪H2S2O8�Ľṹ��ͼ1��

��H2S2O8��ԭ�ӵĹ���ӻ���ʽΪ
��������Ӧ�б���ԭ��Ԫ��Ϊ ��
��������Ӧÿ����1mol MnO4-S2O82-���ѵĹ��ۼ����ͼ�����ĿΪ ��
��4��һ�������£�ˮ���Ӽ��ͨ��OһH��O�������H2O���ӽ�ϳ���ά�Ǽܽṹ�����еĶ������Ѩ�пɰ�������С���ӣ��γ�����ˮ�ϰ����ᄃ��
��ͼ2��һ����ˮ���ӹ��ɵ���ʮ������Ǽܣ���o����ʾˮ���ӣ���������������Ϊ ��
��ʵ���ñ��������������Ϊ18.8ĿkJ?mol-1���������ۻ���Ϊ5.0kJ?mol-1����ԭ������� ��
2Mn2++5S2O82-+8H2O
| ||
��1����̬��ԭ�ӵĺ�������Ų�ʽΪ
��2��������Ӧ�漰��Ԫ������ͬ����Ԫ�أ����һ�������ɴ�С��˳��Ϊ
��3����֪H2S2O8�Ľṹ��ͼ1��

��H2S2O8��ԭ�ӵĹ���ӻ���ʽΪ
��������Ӧ�б���ԭ��Ԫ��Ϊ
��������Ӧÿ����1mol MnO4-S2O82-���ѵĹ��ۼ����ͼ�����ĿΪ
��4��һ�������£�ˮ���Ӽ��ͨ��OһH��O�������H2O���ӽ�ϳ���ά�Ǽܽṹ�����еĶ������Ѩ�пɰ�������С���ӣ��γ�����ˮ�ϰ����ᄃ��
��ͼ2��һ����ˮ���ӹ��ɵ���ʮ������Ǽܣ���o����ʾˮ���ӣ���������������Ϊ
��ʵ���ñ��������������Ϊ18.8ĿkJ?mol-1���������ۻ���Ϊ5.0kJ?mol-1����ԭ�������
���㣺Ԫ�ص����ܡ��縺�Եĺ��弰Ӧ��,���ʽṹ�еĻ�ѧ����Ŀ����,ԭ�Ӻ�������Ų�,ԭ�ӹ���ӻ���ʽ���ӻ������ж�,����Ĵ��ڶ��������ʵ�Ӱ��
ר�⣺
��������1����ԭ������Ϊ25�������������ԭ������������ԭ���ͺ��ع����ۺϿ��ǣ�д����������Ų�ʽ��
��2����Ӧ2Mn2++5S2O82-+8H2O
2MnO4-+10SO42-+16H+�У��漰��Ԫ������ͬ����Ԫ��ΪO��S��ͬ���壬���϶��µ�һ�����ܽ��ͣ�
��3���ٸ��ݼ۲���Ӷ�=�� �����Ӷ�+����ԭ���ϵŵ��Ӷԣ����S2O8 2-�ṹ�ж��ӻ����ͣ�
��������ԭ��Ӧ�У�Ԫ�صĻ��ϼ۽��ͣ�����ԭ��
�ۻ��ý����ͻ��÷ǽ���Ԫ��֮�����γ����Ӽ�����ͬ�ǽ���Ԫ��֮�����γɼ��Լ���ͬ�ַǽ���Ԫ��֮�����γɷǼ��Լ�������S2O8 2-�ṹ�������
��4�����ɴ˽ṹ��֪���˵�Ԫ�к���ˮ���ӵĸ���Ϊ��20������ÿ��ˮ��������������Ԫ�����ݴ˼��������
�ڱ��������������Ϊ18.8kJ?mol-1�������ۻ���Ϊ5.0kJ?mol-1��˵�����ۻ�ΪҺ̬ˮʱֻ���ƻ���һ�������������Һ̬ˮ�����������
��2����Ӧ2Mn2++5S2O82-+8H2O
| ||
��3���ٸ��ݼ۲���Ӷ�=�� �����Ӷ�+����ԭ���ϵŵ��Ӷԣ����S2O8 2-�ṹ�ж��ӻ����ͣ�
��������ԭ��Ӧ�У�Ԫ�صĻ��ϼ۽��ͣ�����ԭ��
�ۻ��ý����ͻ��÷ǽ���Ԫ��֮�����γ����Ӽ�����ͬ�ǽ���Ԫ��֮�����γɼ��Լ���ͬ�ַǽ���Ԫ��֮�����γɷǼ��Լ�������S2O8 2-�ṹ�������
��4�����ɴ˽ṹ��֪���˵�Ԫ�к���ˮ���ӵĸ���Ϊ��20������ÿ��ˮ��������������Ԫ�����ݴ˼��������
�ڱ��������������Ϊ18.8kJ?mol-1�������ۻ���Ϊ5.0kJ?mol-1��˵�����ۻ�ΪҺ̬ˮʱֻ���ƻ���һ�������������Һ̬ˮ�����������
���
�⣺��1����ԭ������Ϊ25��������Ϊ25��ԭ�Ӻ��������Ϊ25�������������ԭ������������ԭ���ͺ��ع���Mn�ĵ����Ų�ʽΪ[Ar]3d54s2��
�ʴ�Ϊ��[Ar]3d54s2��
��2��ͬ�������϶��µ�һ�����ܼ�С��O��Sͬ���壬���Ե�һ�������ɴ�С��˳��ΪO��S���ʴ�Ϊ��O��S��
��3����H2S2O8�У���ԭ�Ӽ۲���Ӷ���=�� �����Ӷ�+����ԭ���ϵŵ��Ӷ�=4+
��6-4��1-2��=4�����Բ�ȡsp3�ӻ����ʴ�Ϊ��sp3�ӻ���
�ڸ÷�Ӧ�У�MnԪ�صĻ��ϼ����ߣ�+2��+7����SԪ�صĻ��ϼ۽��ͣ�+7��+6�������Ա���ԭ��Ԫ��ΪS���ʴ�Ϊ��S��
���ɷ�Ӧ��֪��MnԪ�صĻ��ϼ����ߣ�+2��+7����SԪ�صĻ��ϼ۽��ͣ�+7��+6��������10molSO42-ת�Ƶ���10mol���ӣ���ÿ����1 mol MnO4-��ת�Ƶ���5mol���ӣ�S2O8 2-����2.5mol����2.5NA��O-O��Ǽ��Թ��ۼ����ʴ�Ϊ���Ǽ��Լ���2.5NA��
��4�����ɴ˽ṹ��֪���˵�Ԫ�к���ˮ���ӵĸ���Ϊ��20������ÿ��ˮ�����γɵ��������2����Ԫ������ÿ��ˮ�����γ��������Ϊ��
�����ܹ��γ������Ϊ��20��
=30���ʴ�Ϊ��30��
�ڱ��������������Ϊ18.8kJ?mol-1�������ۻ���Ϊ5.0kJ?mol-1��˵�����ۻ�ΪҺ̬ˮʱֻ���ƻ���һ�������������Һ̬ˮ������������ʴ�Ϊ��Һ̬ˮ����Ȼ���ڴ��������
�ʴ�Ϊ��[Ar]3d54s2��
��2��ͬ�������϶��µ�һ�����ܼ�С��O��Sͬ���壬���Ե�һ�������ɴ�С��˳��ΪO��S���ʴ�Ϊ��O��S��
��3����H2S2O8�У���ԭ�Ӽ۲���Ӷ���=�� �����Ӷ�+����ԭ���ϵŵ��Ӷ�=4+
| 1 |
| 2 |
�ڸ÷�Ӧ�У�MnԪ�صĻ��ϼ����ߣ�+2��+7����SԪ�صĻ��ϼ۽��ͣ�+7��+6�������Ա���ԭ��Ԫ��ΪS���ʴ�Ϊ��S��
���ɷ�Ӧ��֪��MnԪ�صĻ��ϼ����ߣ�+2��+7����SԪ�صĻ��ϼ۽��ͣ�+7��+6��������10molSO42-ת�Ƶ���10mol���ӣ���ÿ����1 mol MnO4-��ת�Ƶ���5mol���ӣ�S2O8 2-����2.5mol����2.5NA��O-O��Ǽ��Թ��ۼ����ʴ�Ϊ���Ǽ��Լ���2.5NA��
��4�����ɴ˽ṹ��֪���˵�Ԫ�к���ˮ���ӵĸ���Ϊ��20������ÿ��ˮ�����γɵ��������2����Ԫ������ÿ��ˮ�����γ��������Ϊ��
| 3 |
| 2 |
| 3 |
| 2 |
�ڱ��������������Ϊ18.8kJ?mol-1�������ۻ���Ϊ5.0kJ?mol-1��˵�����ۻ�ΪҺ̬ˮʱֻ���ƻ���һ�������������Һ̬ˮ������������ʴ�Ϊ��Һ̬ˮ����Ȼ���ڴ��������
���������⿼���������Ų���������ԭ��Ӧ����һ�����ܡ��ӻ������ۼ����͡������ļ����֪ʶ�㣬Ϊ��Ƶ���㣬������ԭ��Ӧ����շ�Ӧ��Ԫ�صĻ��ϼ۱仯������Ӧ���ӻ����ۣ����յ�һ�����ܱ仯����Ϊ���Ĺؼ�����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
�����ƵĻ������������ʻ���;��������ǣ�������
| A��Na2O2-����ɫ���壬������Ư�� |
| B��Na2O-��ɫ���壬���ʲ��ȶ� |
| C��Na2CO3-�����ȶ���������θ����� |
| D��NaHCO3-�����ֽ⣬���������ͷ� |
�ö��Ե缫���CuSO4��Һһ��ʱ���ֹͣ�����������Һ�м���0.2molCu��OH��2��ǡ�÷�Ӧʱ��ҺŨ�Ȼָ������ǰ�����ڵ�����˵������ȷ���ǣ�������
| A�����ɵ�ͭ�����ʵ�����0.2mol |
| B�����ŵ�������Һ��pH��С |
| C��ת�Ƶ��ӵ����ʵ���Ϊ0.4mol |
| D��������Ӧ��40H--4e-=2H20+O2�� |
���з���������ԭ�Ӷ����������Ϊ8���ӽṹ���ǣ�������
| A��BF3 |
| B��H2O |
| C��CH4 |
| D��CO2 |
���и������ʽ��ȷ���ǣ�������
A��N2�ĵ���ʽ�� |
| B��������̼���ӵĽṹʽΪ O-C-O |
C��NaCl�ĵ���ʽ��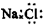 |
D��F-�Ľṹʾ��ͼ�� |
����˵����ȷ���ǣ�������
| A����п���Ͷ������ĶƲ������ǰ�߽��ױ���ʴ |
| B������ǽ�ȼ���еĻ�ѧ��ֱ��ת��Ϊ���ܵĹ��� |
| C����ú��������ȼ�տ������ú��ȼ��Ч�� |
| D����ѧ��Ӧ���Ȳ�����ͨ���ı��������ı� |
 CO2Ϊ̼Դ��ȡ��̼�л����Ϊ�����о����㣬����ΪCO2������ȡ�Ҵ��ķ�Ӧ��2CO2��g��+6H2��g��=CH3CH2OH��g��+3H2O��g����H=QkJ/mol ��Q��0�����ܱ������У���CO2��H2�����ʵ���֮��Ϊ1��3����Ͷ�ϣ���5MPa�²�ò�ͬ�¶���ƽ����ϵ�и������ʵ����������y%����ͼ��ʾ��
CO2Ϊ̼Դ��ȡ��̼�л����Ϊ�����о����㣬����ΪCO2������ȡ�Ҵ��ķ�Ӧ��2CO2��g��+6H2��g��=CH3CH2OH��g��+3H2O��g����H=QkJ/mol ��Q��0�����ܱ������У���CO2��H2�����ʵ���֮��Ϊ1��3����Ͷ�ϣ���5MPa�²�ò�ͬ�¶���ƽ����ϵ�и������ʵ����������y%����ͼ��ʾ��