��Ŀ����
��֪���ʯ��ʯī�ֱ�����������ȫȼ�յ��Ȼ�ѧ����ʽΪ��C�����ʯ��s)��O2(g)��CO2(g)����H����395.41kJ/mol��C��ʯī��s����O2(g)��CO2(g)����H����393.51kJ/mol������ʯת��ʯīʱ���Ȼ�ѧ����ʽΪ��____________________________________��
�ɴ˿������ȶ���̼��ͬ��������Ϊ��____________����ȡ���ʯ��ʯī��Ͼ��干1mol ��O2����ȫȼ�գ���������ΪQkJ������ʯ��ʯī�����ʵ���֮��Ϊ_____________���ú�Q�Ĵ���ʽ��ʾ��
C�����ʯ��s����C��ʯī��s�� ��H����1.90kJ/mol��ʯī��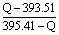
����������������ݸ�˹���ɿ�֪���٣��ڼ��õ�C�����ʯ��s����C��ʯī��s�������Ը÷�Ӧ�ķ�Ӧ�ȡ�H����395.41kJ/mol��393.51kJ/mol����1.90kJ/mol�������Ȼ�ѧ����ʽ��֪�����ʯ����������ʯī�ģ�����ʯī�Ƚ��ʯ�ȶ�������ʯ��ʯī�����ʵ����ֱ���x��y����x��y��1mol�������Ȼ�ѧ����ʽ��֪��395.41kJ/mol��x��393.51kJ/mol��y��QkJ�����x�Uy��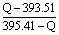 ��
��
���㣺���鷴Ӧ�ȵ��йؼ����Լ��Ȼ�ѧ����ʽ����д
�����������Ȼ�ѧ����ʽ���㷴Ӧ��ʱ��һ�㳣���ݸ�˹���ɽ��С���������һ�ʣ�Ҳ����ͨ��ʮ�ֽ��淨���С�

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д���12�֣����³�ѹ�£�����1mol�����룩������ӻ�ѧ�������յ��������γ�1mol �����룩������ӻ�ѧ�����ų���������Ϊ���ܡ��±���һЩ�������ݣ���λΪKJ��mol-1��
| ��ѧ�� | ���� | ��ѧ�� | ���� | ��ѧ�� | ���� |
| C��F | 427 | C��Cl | 330 | C��I | 218 |
| H��H | 436 | S��S | 255 | H��S | 339 |
�ش��������⣺
(1)�ɱ������ݹ���Ԥ��C��Br���ļ��ܷ�Χ��
_________ <C��Br����<__________
(2)�Ȼ�ѧ����ʽ2H2(g)��S2(g) =2H2S(g)����H= QKJ��mol��1����Q=
(3) ��֪�����Ȼ�ѧ����ʽ��
O2 (g) = O+2(g)+ e�� ��H1= +1175.7 kJ��mol-1
PtF6(g) + e��= PtF6��(g) ��H2=-771.1 kJ��mol-1
O2+PtF6��(s)= O2+(g) + PtF6��(g) ��H3= +482.2 kJ��mol-1
��ӦO2(g)+ (g)= O2+PtF6�� (s)�ġ�H=___________________ kJ��mol-1��
(4)��֪���ʯ��ʯī�ֱ�����������ȫȼ�յ��Ȼ�ѧ����ʽΪ��
C�����ʯ,s��+O2(g) ��CO2(g) ��H=��395.4 kJ•mol��1
C(ʯī,s)+O2(g)��CO2(g) ��H=��393.5 kJ•mol��1
��ʯīת��Ϊ���ʯʱ���Ȼ�ѧ����ʽΪ
�ɴ˿���̼��������ͬ���������и��ȶ���Ϊ ����
��12�֣����³�ѹ�£�����1mol�����룩������ӻ�ѧ�������յ��������γ�1mol �����룩������ӻ�ѧ�����ų���������Ϊ���ܡ��±���һЩ�������ݣ���λΪKJ��mol-1��
|
��ѧ�� |
���� |
��ѧ�� |
���� |
��ѧ�� |
���� |
|
C��F |
427 |
C��Cl |
330 |
C��I |
218 |
|
H��H |
436 |
S��S |
255 |
H��S |
339 |
�ش��������⣺
(1)�ɱ������ݹ���Ԥ��C��Br���ļ��ܷ�Χ��
_________ <C��Br����<__________
(2)�Ȼ�ѧ����ʽ2H2(g)��S2(g) =2H2S(g)����H= QKJ��mol��1����Q=
(3) ��֪�����Ȼ�ѧ����ʽ��
��֪�����Ȼ�ѧ����ʽ��
O2 (g) = O+2(g) + e�� ��H1= +1175.7 kJ��mol-1
 PtF6(g) + e��= PtF6��(g)
��H2=-771.1 kJ��mol-1
PtF6(g) + e��= PtF6��(g)
��H2=-771.1 kJ��mol-1
O2+PtF6��(s) = O2+(g) + PtF6��(g) ��H3= +482.2 kJ��mol-1
 ��ӦO2(g)
+ (g)
= O2+PtF6�� (s)�ġ�H=___________________ kJ��mol-1��
��ӦO2(g)
+ (g)
= O2+PtF6�� (s)�ġ�H=___________________ kJ��mol-1��
(4)��֪���ʯ��ʯī�ֱ�����������ȫȼ�յ��Ȼ�ѧ����ʽΪ��
C�����ʯ,s��+O2(g) ��CO2(g) ��H=��395.4 kJ•mol��1
C(ʯī,s)+O2(g)��CO2(g) ��H=��393.5 kJ•mol��1
��ʯīת��Ϊ���ʯʱ���Ȼ�ѧ����ʽΪ
�ɴ˿���̼��������ͬ���������и��ȶ���Ϊ ����