��Ŀ����
 ��Ȼ������Ҫ�ɷּ���ȼ�����ɶ�����̼��Һ̬ˮ���Ȼ�ѧ����ʽ�ǣ�
��Ȼ������Ҫ�ɷּ���ȼ�����ɶ�����̼��Һ̬ˮ���Ȼ�ѧ����ʽ�ǣ�CH4��g��+2O2��g���TCO2��g��+2H2O��l����H�T-889.6kJ/mol
��ش��������⣺
��1����Ӧ�������ܺ�
��2����1mol������ȫȼ�����ɶ�����̼��ˮ��������ų�������
��3����֪����ȼ������Һ̬ˮ���Ȼ�ѧ����ʽ�ǣ�2H2��g��+O2��g���T2H2O��l����H=-572kJ/mol������ͬ�����ļ������������ȫȼ������Һ̬ˮ�����Ƚ϶����
��4����ͼ��ʾ��װ������CH4��O2��KOH��Һ��ɵ�����ȼ�ϵ�أ����ø�װ�ÿ��Խ�
���㣺��Ӧ�Ⱥ��ʱ�,��ѧ��Դ���͵��
ר�⣺��ѧ��Ӧ�е������仯
��������1�������Ȼ�ѧ����ʽ�С�H�ķ��ŷ�����
��2��ˮ����ת��ΪҺ̬ˮҪ�ų��������ݴ˷�����
��3�����ݷ���ʽ�����ͬ�����ļ������������ȫȼ������Һ̬ˮ�ų�������������
��4��ȼ�ϵ�ؽ���ѧ��ת��Ϊ���ܣ�
��2��ˮ����ת��ΪҺ̬ˮҪ�ų��������ݴ˷�����
��3�����ݷ���ʽ�����ͬ�����ļ������������ȫȼ������Һ̬ˮ�ų�������������
��4��ȼ�ϵ�ؽ���ѧ��ת��Ϊ���ܣ�
���
�⣺��1����֪CH4��g��+2O2��g��=CO2��g��+2H2O��l����H=-889.6kJ/mol����Ӧ�Ƿ��ȷ�Ӧ�����Է�Ӧ�������ܺʹ��������������ܺͣ��ʴ�Ϊ�����ڣ�
��2��ˮ����ת��ΪҺ̬ˮҪ�ų�����������1mol������ȫȼ�����ɶ�����̼��ˮ�������ų�������С��889.6kJ���ʴ�Ϊ������
��3����֪CH4��g��+2O2��g��=CO2��g��+2H2O��l����H=-889.6kJ/mol��2H2��g��+O2��g��=2H2O��l����H=-572kJ/mol����1g������ȫȼ�շų�������Ϊ
889.6kJ��1g������ȫȼ�շų�������Ϊ
��572kJ��������ͬ�����ļ������������ȫȼ������Һ̬ˮ�������ŵ������϶࣬�ʴ�Ϊ��������
��4��ԭ����ǰѻ�ѧ��ת��Ϊ���ܵ�װ�ã�����ȼ�ϵ�ؽ���ѧ��ת��Ϊ���ܣ��ʴ�Ϊ����ѧ�ܣ����ܣ�
��2��ˮ����ת��ΪҺ̬ˮҪ�ų�����������1mol������ȫȼ�����ɶ�����̼��ˮ�������ų�������С��889.6kJ���ʴ�Ϊ������
��3����֪CH4��g��+2O2��g��=CO2��g��+2H2O��l����H=-889.6kJ/mol��2H2��g��+O2��g��=2H2O��l����H=-572kJ/mol����1g������ȫȼ�շų�������Ϊ
| 1 |
| 16 |
| 1 |
| 4 |
��4��ԭ����ǰѻ�ѧ��ת��Ϊ���ܵ�װ�ã�����ȼ�ϵ�ؽ���ѧ��ת��Ϊ���ܣ��ʴ�Ϊ����ѧ�ܣ����ܣ�
���������⿼���˻�ѧ��Ӧ�������ı仯��ԭ���ԭ��ԭ������ȷԭ��ص缫�Ϸ�����Ӧ�����ͼ��ɷ�������⣬�ѶȲ���

��ϰ��ϵ�д�
 ��ĩ100�ִ��غ�������ϵ�д�
��ĩ100�ִ��غ�������ϵ�д�
�����Ŀ
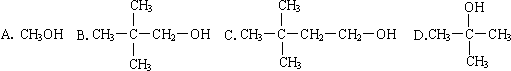
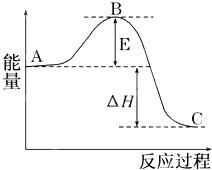 2SO2��g��+O2��g��?2SO3��g����Ӧ���̵������仯��ͼ��ʾ����֪1mol SO2��g������Ϊ1mol SO3��g���ġ�H=-99kJ/mol��
2SO2��g��+O2��g��?2SO3��g����Ӧ���̵������仯��ͼ��ʾ����֪1mol SO2��g������Ϊ1mol SO3��g���ġ�H=-99kJ/mol��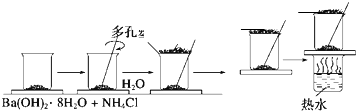 ���о���ѧ��Ӧ�е������仯ʱ������ͨ���������ʵ�飺��һ��С�ձ������20g����ĥ�ɷ�ĩ��Ba��OH��2?8H2O����С�ձ����������ѵ���3��4��ˮ�IJ���Ƭ�ϣ�Ȼ�����ձ��м���Լ10g NH4Cl���壬����ʵ�鲽�裬��д�±������ش����⣮
���о���ѧ��Ӧ�е������仯ʱ������ͨ���������ʵ�飺��һ��С�ձ������20g����ĥ�ɷ�ĩ��Ba��OH��2?8H2O����С�ձ����������ѵ���3��4��ˮ�IJ���Ƭ�ϣ�Ȼ�����ձ��м���Լ10g NH4Cl���壬����ʵ�鲽�裬��д�±������ش����⣮