��Ŀ����
��2010?������ģ����ͼװ�ñպϵ��ʱ�������Ƶ�ָ�뽫����ƫת���Իش��������⣮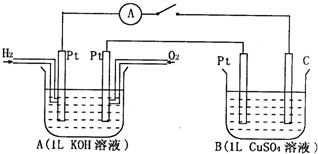
��1��д��A��H2�缫������
��2��B�ع���һ��ʱ���ҪʹB��CuSO4��Һ�ָ�������ǰ��Ũ�ȿɼ���������������
a��Cu b��CuSO4
c��CuO d��CuCO3
��3�������B���е����缫������ͭ�缫������һ��ʱ�����Һ����ɫ
��4������缫���ϲ��䣬��B��CuSO4��Һ����NaCl��Һ������һ��ʱ���C�缫����pH
��5���缫���ϲ��䣬��B��ΪlL Cu��NO3��2��KNO3�Ļ����Һ��ͨ��һ��ʱ�䣬����������11.2L����״�������壬������ͭ������Ϊ

��1��д��A��H2�缫������
����
����
���õ缫��Ӧʽ2OH-+H2-2e-=2H2O
2OH-+H2-2e-=2H2O
����2��B�ع���һ��ʱ���ҪʹB��CuSO4��Һ�ָ�������ǰ��Ũ�ȿɼ���������������
cd
cd
a��Cu b��CuSO4
c��CuO d��CuCO3
��3�������B���е����缫������ͭ�缫������һ��ʱ�����Һ����ɫ
�ޱ仯
�ޱ仯
��������������dz�����ޱ仯������4������缫���ϲ��䣬��B��CuSO4��Һ����NaCl��Һ������һ��ʱ���C�缫����pH
����
����
�����������С�����ޱ仯������B�ع���ʱ�ܵķ�Ӧ���ӷ���ʽΪ��2Cl-+2H2O
2OH-+H2��+Cl2��
| ||
2Cl-+2H2O
2OH-+H2��+Cl2��
��
| ||
��5���缫���ϲ��䣬��B��ΪlL Cu��NO3��2��KNO3�Ļ����Һ��ͨ��һ��ʱ�䣬����������11.2L����״�������壬������ͭ������Ϊ
32g
32g
����������1��A��Ϊ��������ȼ�ϵ�أ���������������Ӧ����������������ˮ������������ԭ��Ӧ����������ԭ�������������ӣ�
��2��B��Ϊ���أ�����������������������ͭ��
��3�������B���е����缫������ͭ�缫��Ϊ���װ�ã����ҺŨ�Ȳ��䣻
��4������缫���ϲ��䣬��B��CuSO4��Һ����NaCl��Һ��C�缫Ϊ������������ԭ��Ӧ�������������������ӣ���������������
��5����B��ΪlL Cu��NO3��2��KNO3�Ļ����Һ��ͨ��һ��ʱ�䣬����������11.2L����״�������壬˵�������ֱ�����ͭ������������������������ϵ缫����ʽ���㣮
��2��B��Ϊ���أ�����������������������ͭ��
��3�������B���е����缫������ͭ�缫��Ϊ���װ�ã����ҺŨ�Ȳ��䣻
��4������缫���ϲ��䣬��B��CuSO4��Һ����NaCl��Һ��C�缫Ϊ������������ԭ��Ӧ�������������������ӣ���������������
��5����B��ΪlL Cu��NO3��2��KNO3�Ļ����Һ��ͨ��һ��ʱ�䣬����������11.2L����״�������壬˵�������ֱ�����ͭ������������������������ϵ缫����ʽ���㣮
����⣺��1��A��Ϊ��������ȼ�ϵ�أ���������������Ӧ����������������ˮ������������ԭ��Ӧ����������ԭ�������������ӣ�������ӦʽΪ2OH-+H2-2e-=2H2O���ʴ�Ϊ��������2OH-+H2-2e-=2H2O��
��2��B��Ϊ���أ�����������������������ͭ��B�ع���һ��ʱ���ҪʹB��CuSO4��Һ�ָ�������ǰ��Ũ�ȿɼ�������������ͭ��̼��ͭ������ͭ����Ӧ����������ͭŨ��ƫ�ʴ�Ϊ��cd��
��3�������B���е����缫������ͭ�缫��Ϊ���װ�ã����ҺŨ�Ȳ��䣬�ʴ�Ϊ���ޱ仯��
��4������缫���ϲ��䣬��B��CuSO4��Һ����NaCl��Һ��C�缫Ϊ������������ԭ��Ӧ�������������������ӣ�pH������������������ܷ���ʽΪ2Cl-+2H2O
2OH-+H2��+Cl2�����ʴ�Ϊ������2Cl-+2H2O
2OH-+H2��+Cl2����
��5����Һ������������������ʧ�������������������ӷŵ�˳��Cu2+��H+��
�����缫��ӦΪ��4OH--4e-�T2H2O+O2���������缫��ӦΪ��Cu2++2e-�TCu��2H++2e-�TH2����
������������11.2L��״���µ��������ʵ���Ϊ0.5mol�����ݵ����غ�缫ת�Ƶ���Ϊ4��0.5mol=2mol����������ת�Ƶ���1mol��ͭ���ӵõ�����1mol����Ӧ��ͭ�������ʵ���0.5mol������ͭ������Ϊ0.5mol��64g/mol=32g���ʴ�Ϊ��32g��
��2��B��Ϊ���أ�����������������������ͭ��B�ع���һ��ʱ���ҪʹB��CuSO4��Һ�ָ�������ǰ��Ũ�ȿɼ�������������ͭ��̼��ͭ������ͭ����Ӧ����������ͭŨ��ƫ�ʴ�Ϊ��cd��
��3�������B���е����缫������ͭ�缫��Ϊ���װ�ã����ҺŨ�Ȳ��䣬�ʴ�Ϊ���ޱ仯��
��4������缫���ϲ��䣬��B��CuSO4��Һ����NaCl��Һ��C�缫Ϊ������������ԭ��Ӧ�������������������ӣ�pH������������������ܷ���ʽΪ2Cl-+2H2O
| ||
| ||
��5����Һ������������������ʧ�������������������ӷŵ�˳��Cu2+��H+��
�����缫��ӦΪ��4OH--4e-�T2H2O+O2���������缫��ӦΪ��Cu2++2e-�TCu��2H++2e-�TH2����
������������11.2L��״���µ��������ʵ���Ϊ0.5mol�����ݵ����غ�缫ת�Ƶ���Ϊ4��0.5mol=2mol����������ת�Ƶ���1mol��ͭ���ӵõ�����1mol����Ӧ��ͭ�������ʵ���0.5mol������ͭ������Ϊ0.5mol��64g/mol=32g���ʴ�Ϊ��32g��
�����������ۺϿ���ԭ��غ͵���֪ʶ��Ϊ�߿��������ͣ�������ѧ���ķ��������ͼ��������Ŀ��飬����������ѧ�������õĿ�ѧ�������ѶȲ���ע�����ԭ��غ͵��صĹ���ԭ����

��ϰ��ϵ�д�
 ӥ�ɽ̸��νӽ̲ĺӱ�����������ϵ�д�
ӥ�ɽ̸��νӽ̲ĺӱ�����������ϵ�д� ���������ν�ϵ�д�
���������ν�ϵ�д�
�����Ŀ