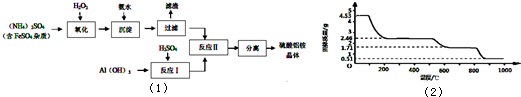
��Ŀ����
�������������մɲ��ϡ����ӹ�ҵ������ҽҩ�ȷ����й�����Ӧ��ǰ��������ͨ��������茶����ȷֽ�õ���[��֪��������茶������ѧʽΪ��NH4��Al��SO4��n?12H2O]���Ʊ�������茶����ʵ��������ͼ��1����

��1���������������С����ˡ��������Ƿ������ʵ�鷽����
��2�����������У������롱�������IJ�������Ϊ��
��3���������ˮ��Һ�����Ե�ԭ����
��4��д����������Ӧ���л��������茶���Ļ�ѧ����ʽ
��5�������������Һ�м�������������Һ��A13+�պó�����ȫ��д����Ӧ�����ӷ���ʽ
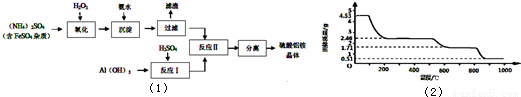
��6��ȡ4.53g������茶�����ȷֽ⣬���ȹ����У�����������ʱ��ı仯��ͼ��2����ʾ��д��400��ʱʣ�����ɷֵĻ�ѧʽ

��1���������������С����ˡ��������Ƿ������ʵ�鷽����
ȡ������Һ���Թ��У��Ӽ���KSCN��Һ������Һ�����ɫ���������ѳ���
ȡ������Һ���Թ��У��Ӽ���KSCN��Һ������Һ�����ɫ���������ѳ���
����2�����������У������롱�������IJ�������Ϊ��
����Ũ��
����Ũ��
����ȴ�ᾧ
��ȴ�ᾧ
�����ˡ�ϴ�ӡ������3���������ˮ��Һ�����Ե�ԭ����
Al3++3H2O?Al��OH��3+H+��NH4++H2O?NH3?H2O+H+
Al3++3H2O?Al��OH��3+H+��NH4++H2O?NH3?H2O+H+
�������ӷ���ʽ��ʾ������4��д����������Ӧ���л��������茶���Ļ�ѧ����ʽ
Al2��SO4��3+��NH4��2SO4+24H2O=2��NH4��Al��SO4��2?12H2O
Al2��SO4��3+��NH4��2SO4+24H2O=2��NH4��Al��SO4��2?12H2O
����5�������������Һ�м�������������Һ��A13+�պó�����ȫ��д����Ӧ�����ӷ���ʽ
2Al3++3SO42-+3Ba2++6OH-=3BaSO4��+2Al��OH��3��
2Al3++3SO42-+3Ba2++6OH-=3BaSO4��+2Al��OH��3��
����6��ȡ4.53g������茶�����ȷֽ⣬���ȹ����У�����������ʱ��ı仯��ͼ��2����ʾ��д��400��ʱʣ�����ɷֵĻ�ѧʽ
��NH4��2Al2��SO4��4?H2O
��NH4��2Al2��SO4��4?H2O
����������1���ɹ������̿�֪������������⽫��Һ��Fe2+����ΪFe3+�����백ˮ������ҺPHֵ��Fe3+ʹת��ΪFe��OH��3�����˺����Һ�п��ܺ���Fe3+����KSCN��Һ�����Ƿ���Fe3+��
��2���ɹ������̿�֪�������С����롱�Ǵ���Һ�л�þ��壬����Ϊ����Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ�����ȣ�
��3��Al3+��NH4+ˮ��ʹ��Һ�����ԣ�
��4���ɻ�ѧʽ��֪n=2����������Һ���������Һ��Ӧ���ɣ�NH4��Al��SO4��2?12H2O��
��5�������������Һ�м�������������Һ��A13+�պó�����ȫ��OH-��Al3+��Ӧ��NH4+����Ӧ����ӦΪ������������������6�����㾧����ˮ�����������ݹ�����������㾧����ʣ��ᾧˮ����������������ʣ�������n[��NH4��Al��SO4��2]��n��H2O�����ݴ���д��ѧʽ��
��2���ɹ������̿�֪�������С����롱�Ǵ���Һ�л�þ��壬����Ϊ����Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ�����ȣ�
��3��Al3+��NH4+ˮ��ʹ��Һ�����ԣ�
��4���ɻ�ѧʽ��֪n=2����������Һ���������Һ��Ӧ���ɣ�NH4��Al��SO4��2?12H2O��
��5�������������Һ�м�������������Һ��A13+�պó�����ȫ��OH-��Al3+��Ӧ��NH4+����Ӧ����ӦΪ������������������6�����㾧����ˮ�����������ݹ�����������㾧����ʣ��ᾧˮ����������������ʣ�������n[��NH4��Al��SO4��2]��n��H2O�����ݴ���д��ѧʽ��
����⣺��1���ɹ������̿�֪������������⽫��Һ��Fe2+����ΪFe3+�����백ˮ������ҺPHֵ��Fe3+ʹת��ΪFe��OH��3�����˺����Һ�п��ܺ���Fe3+��ȡ������Һ���Թ��У��Ӽ���KSCN��Һ������Һ�����ɫ���������ѳ�����
�ʴ�Ϊ��ȡ������Һ���Թ��У��Ӽ���KSCN��Һ������Һ�����ɫ���������ѳ�����
��2���ɹ������̿�֪�������С����롱�Ǵ���Һ�л�þ��壬����Ϊ����Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ�����ȣ�
�ʴ�Ϊ������Ũ������ȴ�ᾧ��
��3����Һ��Al3+ˮ��Al3++3H2O?Al��OH��3+H+��NH4+ˮ��NH4++H2O?NH3?H2O+H+��ʹ��Һ�����ԣ�
�ʴ�Ϊ��Al3++3H2O?Al��OH��3+H+��NH4++H2O?NH3?H2O+H+��
��4����������Һ���������Һ��Ӧ���ɣ�NH4��Al��SO4��2?12H2O��
��Ӧ����ʽΪAl2��SO4��3+��NH4��2SO4+24H2O=2��NH4��Al��SO4��2?12H2O��
�ʴ�Ϊ��Al2��SO4��3+��NH4��2SO4+24H2O=2��NH4��Al��SO4��2?12H2O��
��5�������������Һ�м�������������Һ��A13+�պó�����ȫ��OH-��Al3+��Ӧ��NH4+����Ӧ����ӦΪ��������������������Ӧ���ӷ���ʽΪ2Al3++3SO42-+3Ba2++6OH-=3BaSO4��+2Al��OH��3����
�ʴ�Ϊ��2Al3++3SO42-+3Ba2++6OH-=3BaSO4��+2Al��OH��3����
��6��ȡ4.53g������茶�������ʵ���Ϊ
=0.01mol��
4.53g������茶�����ˮ������Ϊ0.01mol��12��18g/mol=2.16g��
����400��ʱ�����������١�m=4.53g-2.46g=2.07g��2.16g
ʣ������нᾧˮ�����ʵ���Ϊ
=0.005mol��
ʣ�������n[��NH4��Al��SO4��2]��n��H2O��=0.01mol��0.005mol=2��1��
��400��ʱʣ�����ɷֵĻ�ѧʽΪ��NH4��2Al2��SO4��4?H2O��
�ʴ�Ϊ����NH4��2Al2��SO4��4?H2O��
�ʴ�Ϊ��ȡ������Һ���Թ��У��Ӽ���KSCN��Һ������Һ�����ɫ���������ѳ�����
��2���ɹ������̿�֪�������С����롱�Ǵ���Һ�л�þ��壬����Ϊ����Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ�����ȣ�
�ʴ�Ϊ������Ũ������ȴ�ᾧ��
��3����Һ��Al3+ˮ��Al3++3H2O?Al��OH��3+H+��NH4+ˮ��NH4++H2O?NH3?H2O+H+��ʹ��Һ�����ԣ�
�ʴ�Ϊ��Al3++3H2O?Al��OH��3+H+��NH4++H2O?NH3?H2O+H+��
��4����������Һ���������Һ��Ӧ���ɣ�NH4��Al��SO4��2?12H2O��
��Ӧ����ʽΪAl2��SO4��3+��NH4��2SO4+24H2O=2��NH4��Al��SO4��2?12H2O��
�ʴ�Ϊ��Al2��SO4��3+��NH4��2SO4+24H2O=2��NH4��Al��SO4��2?12H2O��
��5�������������Һ�м�������������Һ��A13+�պó�����ȫ��OH-��Al3+��Ӧ��NH4+����Ӧ����ӦΪ��������������������Ӧ���ӷ���ʽΪ2Al3++3SO42-+3Ba2++6OH-=3BaSO4��+2Al��OH��3����
�ʴ�Ϊ��2Al3++3SO42-+3Ba2++6OH-=3BaSO4��+2Al��OH��3����
��6��ȡ4.53g������茶�������ʵ���Ϊ
| 4.53g |
| 453g/mol |
4.53g������茶�����ˮ������Ϊ0.01mol��12��18g/mol=2.16g��
����400��ʱ�����������١�m=4.53g-2.46g=2.07g��2.16g
ʣ������нᾧˮ�����ʵ���Ϊ
| 2.16g-2.07g |
| 18g/mol |
ʣ�������n[��NH4��Al��SO4��2]��n��H2O��=0.01mol��0.005mol=2��1��
��400��ʱʣ�����ɷֵĻ�ѧʽΪ��NH4��2Al2��SO4��4?H2O��
�ʴ�Ϊ����NH4��2Al2��SO4��4?H2O��
����������þ�����仯��������ʣ��Թ����������⡢���û�ѧ�������ˮ�⡢���ʵķ����ᴿ�����Ӽ��顢��ѧ����ȣ��ۺ��Խϴ��ѶȽϸߣ���Ҫѧ���߱���ʵ�Ļ���������֪ʶ��������������������5�������ӷ���ʽ��дΪ�״��㣬ע����Һ����笠����Ӻ������ӣ����������������ӷ�Ӧ��������������Ȼ������������笠����ӷ�Ӧ������ܽ�����������

��ϰ��ϵ�д�
�����Ŀ
�������������մɲ��ϡ����ӹ�ҵ������ҽҩ�ȷ����й�����Ӧ��ǰ��������ͨ��������茶����ȷֽ�õ���[��֪��������茶���Ļ�ѧʽΪ(NH4)Al (SO4)n��12H2O]
�Ʊ�������茶����ʵ���������£�
 |
��1���������������С����ˡ��������Ƿ������ʵ�鷽���� ��
��2�����������У������롱�������IJ�������Ϊ�� �� �����ˡ�ϴ�ӡ����
��3���������ˮ��Һ�����Ե�ԭ���� �������ӷ���ʽ��ʾ����
��4��д����������Ӧ���л��������茶���Ļ�ѧ����ʽ ��
��5�������������Һ�м�������������Һ��Al3+�պó�����ȫ��д����Ӧ�����ӷ���ʽ ��
��6��ȡ4.53 g������茶�����ȷֽ⣬���ȹ����У�����������ʱ��ı仯����ͼ��ʾ��

д��400��ʱʣ�����ɷֵĻ�ѧʽ ��