��Ŀ����
��10�֣�
���º����£���2mol A�����2mol B����ͨ�����Ϊ2L���ܱ������з������·�Ӧ��2A��g����B��g�� xC��g����2D��s����2minʱ��Ӧ�ﵽƽ��״̬����ʱʣ��1.2mol B�������C��Ũ��Ϊ1
xC��g����2D��s����2minʱ��Ӧ�ﵽƽ��״̬����ʱʣ��1.2mol B�������C��Ũ��Ϊ1 ��2mol��L��1��
��2mol��L��1��
��1��x��_______________��
��2������A�Ļ�ѧ��Ӧ����Ϊ________��A��ת������B��ת����֮��Ϊ_________��
��3���˷�Ӧ�ڸ��¶�ʱ�Ļ�ѧƽ�ⳣ��Ϊ_________
��4�������¶Ⱥ��ݻ����䣬��ƽ����ϵ���ٳ���2mol A��2mol B���壬�ﵽ��ƽ��ʱB�����ʵ���Ϊ_____________��
���º����£���2mol A�����2mol B����ͨ�����Ϊ2L���ܱ������з������·�Ӧ��2A��g����B��g��
 xC��g����2D��s����2minʱ��Ӧ�ﵽƽ��״̬����ʱʣ��1.2mol B�������C��Ũ��Ϊ1
xC��g����2D��s����2minʱ��Ӧ�ﵽƽ��״̬����ʱʣ��1.2mol B�������C��Ũ��Ϊ1 ��2mol��L��1��
��2mol��L��1����1��x��_______________��
��2������A�Ļ�ѧ��Ӧ����Ϊ________��A��ת������B��ת����֮��Ϊ_________��
��3���˷�Ӧ�ڸ��¶�ʱ�Ļ�ѧƽ�ⳣ��Ϊ_________
��4�������¶Ⱥ��ݻ����䣬��ƽ����ϵ���ٳ���2mol A��2mol B���壬�ﵽ��ƽ��ʱB�����ʵ���Ϊ_____________��
��10�֣�
��1��3��2�֣�
��2�� 0.2 mol��L��1��min��1��2�֣� 2��1��2�֣�
��3��72��2�֣�
��4��2.4mol��2�֣�
��1��3��2�֣�
��2�� 0.2 mol��L��1��min��1��2�֣� 2��1��2�֣�
��3��72��2�֣�
��4��2.4mol��2�֣�
��

��ϰ��ϵ�д�
�����Ŀ
 SO3(g)+NO(g���Ŀ��淴Ӧ��������˵����Ӧ�ﵽƽ��״̬����
SO3(g)+NO(g���Ŀ��淴Ӧ��������˵����Ӧ�ﵽƽ��״̬���� pC��g�����ﵽƽ��ʱ��ά���¶Ȳ��䣬�����������С��ԭ����1/2�����ﵽ�µ�ƽ��ʱ������C��Ũ�ȱ�Ϊԭƽ��ʱ��1.9����������˵����ȷ���ǣ� ��
pC��g�����ﵽƽ��ʱ��ά���¶Ȳ��䣬�����������С��ԭ����1/2�����ﵽ�µ�ƽ��ʱ������C��Ũ�ȱ�Ϊԭƽ��ʱ��1.9����������˵����ȷ���ǣ� �� Z(g)(δ��ƽ)���¶�ΪT0ʱ�����ݻ��̶��������з�����Ӧ�����ʵ�Ũ����ʱ��仯�Ĺ�ϵ��ͼa��ʾ������������ͬ���¶ȷֱ�ΪT1��T2ʱ������Ӧ��Z��Ũ����ʱ��仯�Ĺ�ϵ��ͼb��ʾ������������ȷ���ǣ� ��
Z(g)(δ��ƽ)���¶�ΪT0ʱ�����ݻ��̶��������з�����Ӧ�����ʵ�Ũ����ʱ��仯�Ĺ�ϵ��ͼa��ʾ������������ͬ���¶ȷֱ�ΪT1��T2ʱ������Ӧ��Z��Ũ����ʱ��仯�Ĺ�ϵ��ͼb��ʾ������������ȷ���ǣ� ��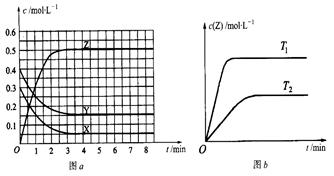
 C(g)
C(g) mol.
mol. 2C��g�����ﵽƽ��ʱ��A��B�����ʵ���֮����C�����ʵ�����ȣ����ʱA��ת����Ϊ
2C��g�����ﵽƽ��ʱ��A��B�����ʵ���֮����C�����ʵ�����ȣ����ʱA��ת����Ϊ nB��g����nC��g���ﵽƽ�⡣�����¶Ȳ��䣬�������ݻ�ѹ����ԭ����һ�룬���ﵽ�µ�ƽ��ʱ��B��C��Ũ�Ⱦ���ԭ����1.8������
nB��g����nC��g���ﵽƽ�⡣�����¶Ȳ��䣬�������ݻ�ѹ����ԭ����һ�룬���ﵽ�µ�ƽ��ʱ��B��C��Ũ�Ⱦ���ԭ����1.8������ CO��g����H2O��g����
CO��g����H2O��g���� ��C12(g)��S(g)������Ӧ(����ֻ��һ��)���������ʵ����ʵ�����ʱ��仯��������ͼ��ʾ����ش��������⣺
��C12(g)��S(g)������Ӧ(����ֻ��һ��)���������ʵ����ʵ�����ʱ��仯��������ͼ��ʾ����ش��������⣺