��Ŀ����
��һ���¶��£�������Һ���ڵ���ƽ�⣺CH3COOH?CH3COO-+H+
��1��ij�¶�ʱ��0.1mol/L������Һ�е�c��H+����0.01mol/L������Һ�е� c��H+���ı�ֵ ������ڡ�����С�ڡ����ڡ���10
��2����֪��25��ʱ���õ���ƽ���ƽ�ⳣ��Ϊ1.75��10-5��
������¶�ʱ��amol/L�Ĵ�����Һ��c1��H+��= mol/L ���ú�a�Ĵ���ʽ��ʾ����[��ʾ����ʱa�Ƚ�С�����м��㣬ƽ��ʱc��CH3COOH�����ó�ʼŨ�ȴ��棬ˮ�������c��H+����c��OH-�����Բ��ƣ���ͬ]
�������¶�ʱ�����Һ�м���һ������CH3COONH4��������Һ������䣩��ʹ��Һ��c��CH3COO-��Ϊbmol/L�����ʱc2��H+��= mol/L���ú�a��b�Ĵ���ʽ��ʾ����
��c1��H+�� c2��H+��������ڡ�����С�ڡ����ڡ���
��1��ij�¶�ʱ��0.1mol/L������Һ�е�c��H+����0.01mol/L������Һ�е� c��H+���ı�ֵ
��2����֪��25��ʱ���õ���ƽ���ƽ�ⳣ��Ϊ1.75��10-5��
������¶�ʱ��amol/L�Ĵ�����Һ��c1��H+��=
�������¶�ʱ�����Һ�м���һ������CH3COONH4��������Һ������䣩��ʹ��Һ��c��CH3COO-��Ϊbmol/L�����ʱc2��H+��=
��c1��H+��
���㣺���������ˮ��Һ�еĵ���ƽ��
ר�⣺����ƽ������Һ��pHר��
��������1��������Һ��Ũ��ԽС����̶�Խ��
��2����c��H+��=
�����㣻
�ڸ���c��H+��=
���������Һ��������Ũ�ȣ�
�۴ӵ���ƽ���ƶ��ĽǶȷ������ǣ�
��2����c��H+��=
| Ka?c(CH3COOH) |
�ڸ���c��H+��=
| Kac(CH3COOH) |
| c(CH3COO-) |
�۴ӵ���ƽ���ƶ��ĽǶȷ������ǣ�
���
�⣺��1��������Һ��Ũ��ԽС����̶�Խ�����0.1 mol/L��CH3COOH��Һ��0.01 mol/L��CH3COOH��Һ�е�c��H+��֮��С��10���ʴ�Ϊ��С�ڣ�
��2����c��H+��=
=
mol/L���ʴ�Ϊ��
��
��c��H+��=
=
���ʴ�Ϊ��
��
�۴ӵ���ƽ���ƶ����м���һ������CH3COONH4��������Һ������䣩����������Ũ�����������ƽ�������ƶ���������Ũ�ȼ�С������c1��H+����c2��H+�����ʴ�Ϊ�����ڣ�
��2����c��H+��=
| Ka?c(CH3COOH) |
| a��1.75��10-5 |
| a��1.75��10-5 |
��c��H+��=
| Kac(CH3COOH) |
| c(CH3COO-) |
| 1.75��10-5��a |
| b |
| 1.75��10-5��a |
| b |
�۴ӵ���ƽ���ƶ����м���һ������CH3COONH4��������Һ������䣩����������Ũ�����������ƽ�������ƶ���������Ũ�ȼ�С������c1��H+����c2��H+�����ʴ�Ϊ�����ڣ�
���������⿼�����ƽ�ⳣ�������ã���Ŀ�Ѷ��еȣ�ע��������ص���ƽ�ⳣ�����㹫ʽ�����ã�

��ϰ��ϵ�д�
�����Ŀ
����ͷ���������������ı����϶���һ�����ʴ������������ʳ��ʱ������Ĺ�ͷ�����⣬���������ķ�Ӧ��ȷ���ǣ�������
| A������Sn-2e��Sn2+ |
| B������Fe-2e��Fe2+ |
| C������2H2O+O2-2e��4OH- |
| D������Fe-2e��Fe2+ |
���л�ѧ����ʽ�����ӷ���ʽ��ȷ���ǣ�������
| A���ô���ȥ��ˮ����CaCO3+2H+�TCa2++H2O+CO2�� | ||
B����ȩ����������Cu��OH��2��Һ��Ӧ��HCHO+2Cu��OH��2
| ||
C��ʵ������Һ��ͱ��ڴ������������屽�� | ||
D����CO2ͨ�뱽������Һ�� |
���л�ѧʽֻ�ܱ�ʾһ�ִ�������ǣ�������
| A��CF2Cl2 |
| B��C3H6 |
| C��C6H12O6 |
| D��C2H4O2 |
�����й�����������У���ȷ���ǣ�������
| A��ɳ�����к���CaCO3�������к����� |
| B��������ָ���Խ�ˮ����ˮ��pH��7 |
| C��δ����Ⱦ���������н��µ���ˮһ����������ˮ? |
| D���ļ��������¶Ƚϸߣ�����SO2��NO��ת��Ϊ���������������ף����ļ���ˮ���Ի��ǿ |
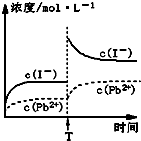 ȡһ������PbI2����������ˮ��ɱ�����Һ����ȡ25.00mLPbI2������Һ���ִ�������R-H��s���������·�Ӧ��Pb2+��aq��+2R-H��s��=R2Pb��s��+2H+��aq������ַ�Ӧ���ˣ�����ƿʢװ��Һ������������ˮϴ�ӳ�������ϴ��Һһ��������ƿ�У�����ָʾ������0.0025mol/LNaOH��Һ�ζ������ﵽ�ζ��յ�ʱ����ȥ����������Һ20.00mL���������ʵ�����ݣ������й�˵����ȷ���ǣ�������
ȡһ������PbI2����������ˮ��ɱ�����Һ����ȡ25.00mLPbI2������Һ���ִ�������R-H��s���������·�Ӧ��Pb2+��aq��+2R-H��s��=R2Pb��s��+2H+��aq������ַ�Ӧ���ˣ�����ƿʢװ��Һ������������ˮϴ�ӳ�������ϴ��Һһ��������ƿ�У�����ָʾ������0.0025mol/LNaOH��Һ�ζ������ﵽ�ζ��յ�ʱ����ȥ����������Һ20.00mL���������ʵ�����ݣ������й�˵����ȷ���ǣ�������| A���Է�̪Ϊָʾ��ʱ���ζ����յ�ʱ��Һ�ɺ�ɫ��Ϊ��ɫ |
| B�����¶Ȳ���ʱ����PbI2������Һ�м�����������ǦŨ��Һ��PbI2��Ksp��С |
| C���¶Ȳ��䣬Tʱ�̣���PbI2������Һ�м�������KIŨ��Һ������Ũ�ȱ仯����ͼ��ʾ |
| D������ʵ���õ�t��PbI2��Ksp=4��10-9 |
ij�����ʽ��NaHY��ˮ��Һ�У�HY-�ĵ���̶�С��HY-��ˮ��̶ȣ��йص�������ȷ���ǣ�������
| A��H2Y�ĵ��뷽��ʽΪ��H2Y=2H++Y2- |
| B���ڸ���ʽ����Һ�У�[Na+]��[Y2-]��[HY-]��[OH-]��[H+] |
| C��HY-��ˮ�ⷽ��ʽ��HY-+H2O?H3O++Y2- |
| D���ڸ���ʽ����Һ�У�[Na+]��[HY-]��[OH-]��[H+]��[Y2-] |
