��Ŀ����
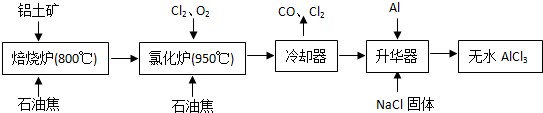
��ˮAlCl3���������������л��ϳɵĴ����ȣ���ҵ����������A12O3��Fe2O3��Ϊԭ���Ʊ���ˮAlCl3�Ĺ����������£�
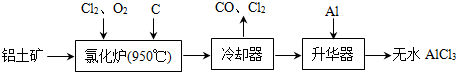
��1���Ȼ�¯��Al2O3��C12��C��Ӧ�Ļ�ѧ����ʽΪ
��2����Na2SO3��Һ�ɳ�ȥ��ȴ���ų�β���е�Cl2���˷�Ӧ�����ӷ���ʽΪ��
��3������������Ҫ����AlCl3��FeCl3�����������Al����������
��4��Ϊ�ⶨ�Ƶõ���ˮAlCl3��Ʒ��������FeCl3���Ĵ��ȣ���ȡ16.25g��ˮAlCl3��Ʒ�����ڹ�����NaOH��Һ�����˳�����������ᆳϴ�ӡ����ա���ȴ�����أ�������������Ϊ0.32g��
��д�����ӹ������漰�����ӷ���ʽ��
��AlCl3��Ʒ�Ĵ���Ϊ
��5����ҵ����һ��������Ϊԭ���Ʊ���ˮAlCl3�����У����һ������AlCl3?6H2O��ˮ�Ʊ���ˮAlCl3��ʵ����һ���ķ�����

��1���Ȼ�¯��Al2O3��C12��C��Ӧ�Ļ�ѧ����ʽΪ
A12O3+3C12+3C
2A1C13+3CO
| ||
A12O3+3C12+3C
2A1C13+3CO
��
| ||
��2����Na2SO3��Һ�ɳ�ȥ��ȴ���ų�β���е�Cl2���˷�Ӧ�����ӷ���ʽΪ��
SO32-+C12+H2O�TSO42-+2C1-+2H+
SO32-+C12+H2O�TSO42-+2C1-+2H+
����3������������Ҫ����AlCl3��FeCl3�����������Al����������
��ȥFeCl3
��ȥFeCl3
����4��Ϊ�ⶨ�Ƶõ���ˮAlCl3��Ʒ��������FeCl3���Ĵ��ȣ���ȡ16.25g��ˮAlCl3��Ʒ�����ڹ�����NaOH��Һ�����˳�����������ᆳϴ�ӡ����ա���ȴ�����أ�������������Ϊ0.32g��
��д�����ӹ������漰�����ӷ���ʽ��
Fe3++3OH-=Fe��OH��3��
Fe3++3OH-=Fe��OH��3��
��Al3++4OH-=AlO2-+2H2O
Al3++4OH-=AlO2-+2H2O
����AlCl3��Ʒ�Ĵ���Ϊ
96%
96%
����5����ҵ����һ��������Ϊԭ���Ʊ���ˮAlCl3�����У����һ������AlCl3?6H2O��ˮ�Ʊ���ˮAlCl3��ʵ����һ���ķ�����
��HCl�����м����ѽᾧˮ
��HCl�����м����ѽᾧˮ
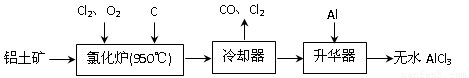
����������1�����ݹ������̿�֪�������к���AlCl3�ȣ�����Al2O3��C12��C��Ӧ������A1C13����������β����֪��������CO��
��2��Cl2��ǿ�����ԣ���SO32-����ΪSO42-����������ԭΪ2C1-��
��3������������Ҫ����AlCl3��FeCl3��FeCl3�۵㡢�е�ϵͣ������Ʊ���ˮAlCl3��Ӧ��ȥFeCl3��
��4���Ʊ���ˮAlCl3��������FeCl3��������������Ϊ0.32gΪFe2O3��������ԭ���غ����FeCl3���������������AlCl3���������ٸ��ݲ�Ʒ�Ĵ��ȶ�����㣮
��5���Ȼ�����Һ����ˮ�⣬��AlCl3?6H2O��ˮ�Ʊ���ˮ�Ȼ�������HCl�����м����ѽᾧˮ��
��2��Cl2��ǿ�����ԣ���SO32-����ΪSO42-����������ԭΪ2C1-��
��3������������Ҫ����AlCl3��FeCl3��FeCl3�۵㡢�е�ϵͣ������Ʊ���ˮAlCl3��Ӧ��ȥFeCl3��
��4���Ʊ���ˮAlCl3��������FeCl3��������������Ϊ0.32gΪFe2O3��������ԭ���غ����FeCl3���������������AlCl3���������ٸ��ݲ�Ʒ�Ĵ��ȶ�����㣮
��5���Ȼ�����Һ����ˮ�⣬��AlCl3?6H2O��ˮ�Ʊ���ˮ�Ȼ�������HCl�����м����ѽᾧˮ��
����⣺��1�����ݹ������̿�֪�Ȼ�¯�IJ������ȴ���������Ʊ���ˮAlCl3��˵���Ȼ�¯�IJ����к���A1C13��������β������CO������Al2O3��C12��C��Ӧ������A1C13��CO����Ӧ����ʽΪA12O3+3C12+3C
2A1C13+3CO��
�ʴ�Ϊ��A12O3+3C12+3C
2A1C13+3CO��
��2��Cl2��ǿ�����ԣ���SO32-����ΪSO42-����������ԭΪ2C1-����Ӧ���ӷ���ʽΪSO32-+C12+H2O�TSO42-+2C1-+2H+��
�ʴ�Ϊ��SO32-+C12+H2O�TSO42-+2C1-+2H+��
��3������������Ҫ����AlCl3��FeCl3��FeCl3�۵㡢�е�ϵͣ������Ʊ���ˮAlCl3��Ӧ��FeCl3ȥ�����Լ�������AlĿ���dz�ȥFeCl3��
�ʴ�Ϊ����ȥFeCl3��
��4���ٳ��ӹ����������������������ӽ�ϳ��������������������������������ӽ�ϳ�ƫ�������ˮ�����ӷ���ʽΪ��Fe3++3OH-=Fe��OH��3����Al3++4OH-=AlO2-+2H2O��
�ʴ�Ϊ��Fe3++3OH-=Fe��OH��3����Al3++4OH-=AlO2-+2H2O��
�ڸ�����ԭ���غ㣬�������FeCl3����Ϊm����
Fe2O3����������2FeCl3
160 325
0.32g m
=
��ã�m=0.65g��
����AlCl3��Ʒ�Ĵ���Ϊ
��100%=96%��
�ʴ�Ϊ��96%��
��5���Ȼ�����Һ����ˮ�⣬��AlCl3?6H2O��ˮ�Ʊ���ˮ�Ȼ�������HCl�����м����ѽᾧˮ��
�ʴ�Ϊ����HCl�����м����ѽᾧˮ��
| ||
�ʴ�Ϊ��A12O3+3C12+3C
| ||
��2��Cl2��ǿ�����ԣ���SO32-����ΪSO42-����������ԭΪ2C1-����Ӧ���ӷ���ʽΪSO32-+C12+H2O�TSO42-+2C1-+2H+��
�ʴ�Ϊ��SO32-+C12+H2O�TSO42-+2C1-+2H+��
��3������������Ҫ����AlCl3��FeCl3��FeCl3�۵㡢�е�ϵͣ������Ʊ���ˮAlCl3��Ӧ��FeCl3ȥ�����Լ�������AlĿ���dz�ȥFeCl3��
�ʴ�Ϊ����ȥFeCl3��
��4���ٳ��ӹ����������������������ӽ�ϳ��������������������������������ӽ�ϳ�ƫ�������ˮ�����ӷ���ʽΪ��Fe3++3OH-=Fe��OH��3����Al3++4OH-=AlO2-+2H2O��
�ʴ�Ϊ��Fe3++3OH-=Fe��OH��3����Al3++4OH-=AlO2-+2H2O��
�ڸ�����ԭ���غ㣬�������FeCl3����Ϊm����
Fe2O3����������2FeCl3
160 325
0.32g m
| 160 |
| 0.32g |
| 325 |
| m |
��ã�m=0.65g��
����AlCl3��Ʒ�Ĵ���Ϊ
| 16.25g-0.65g |
| 16.25g |
�ʴ�Ϊ��96%��
��5���Ȼ�����Һ����ˮ�⣬��AlCl3?6H2O��ˮ�Ʊ���ˮ�Ȼ�������HCl�����м����ѽᾧˮ��
�ʴ�Ϊ����HCl�����м����ѽᾧˮ��
���������⿼��ѧ�����ڹ�������ԭ�������⡢�Բ�����ʵ���������Ƶ�����ȣ��漰���û�ѧ������д����ѧ���㡢�����ᴿ�ȣ���Ҫѧ���߱���ʵ�Ļ������ۺ������������Ѷ��еȣ�

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ