��Ŀ����
12��ijͬѧ��ʵ�����г�ȡ20g NaOH���壬����500mL 1mol•L-1������������Һ����1����ͬѧ����ʵ����Ʒ�У�������ƽ�������룩��ҩ�ס���Ͳ���������ȣ���ȱ�ٵIJ���������500 mL����ƿ���ձ�����ͷ�ιܣ�
��2�������й�ʵ���������ȷ����BD��
A�������������ƹ���ʱ��Ҫ����ƽ���������������Ϸֱ����������ͬ����С�ձ�
B��ijͬѧ���������������ƹ����ܽⲢ��ȴ�����£�Ȼ��ת�Ƶ�����ƿ��
C��ijͬѧ�ڶ���ʱ����С�ļ�ˮ�����̶��ߣ��ֽ������Һ������ֱ���̶���
D��ijͬѧ���ܽ⡢ת��������������Һ���ܽ��õ�����ϴ��2��3�Σ�����ϴ��Һת�Ƶ�����ƿ��
��3���������ڸ�ʵ���е�������
�ٽ��裬�����������ƹ�����ܽ⣻
������Һ�壮
��4����ʵ�����������������������������Һ�����ʵ���Ũ���к�Ӱ�죨�ƫ�ߡ�����ƫ�͡�����Ӱ�족����
�ٳ�����������ʱ��ֱ�ӷŵ�������ƽ�ϳ���ƫ�ͣ�
�ڶ���ҡ�Ⱥ���������Һ������������Һ��Ũ��ƫ�ͣ�
���� ��1����������һ�����ʵ���Ũ����Һһ�㲽��ѡ����Ҫ������
��2������ʵ������ķ���������
��3�����������ܽ����ʱ����Ϊ���裬���ٹ����ܽ⣻����Һʱ��������Һ�壻
��4���������������ʵ����ʵ�������Һ�����Ӱ�죬����C=$\frac{n}{V}$������������
��� �⣺��1������һ�����ʵ���Ũ����Һһ�㲽�裺���㡢�������ܽ⡢��Һ��ϴ�ӡ����ݡ�ҡ�ȣ��õ���������������ƽ�������룩��ҩ�ס���Ͳ�����������ձ�������ƿ����ͷ�ιܣ�����500mL 1mol•L-1������������Һ��Ӧѡ��500mL����ƿ�����Ի�ȱ�ٵ�������500 mL����ƿ���ձ�����ͷ�ιܣ�
�ʴ�Ϊ��500 mL����ƿ���ձ�����ͷ�ιܣ�
��2��A����������Ϊ�׳���Ĺ��壬Ӧ���ڲ��������г�����������Ҫ����һ���Ϸ����ձ�����A����
B�����������ƹ����ܽ�ˮ���ȣ�Ӧ����ȴ�����£�Ȼ��ת�Ƶ�����ƿ�У��������������B��ȷ��
C��ijͬѧ�ڶ���ʱ����С�ļ�ˮ�����̶��ߣ��ֽ������Һ������ֱ���̶��ߣ����²������ʱ��������������Ƶ�Ũ��ƫ�ͣ���C����
D��ijͬѧ���ܽ⡢ת��������������Һ���ܽ��õ�����ϴ��2��3�Σ�����ϴ��Һת�Ƶ�����ƿ�У�����ȷ������ȫ��ת�Ƶ�����ƿ�У���D��ȷ��
��ѡ��BD��
��3�����������ܽ����ʱ����Ϊ���裬���ٹ����ܽ⣻����Һʱ��������Һ�壬�ʴ�Ϊ�����裬�����������ƹ�����ܽ⣻����Һ�壻
��4���ٳ�����������ʱ��ֱ�ӷŵ�������ƽ�ϳ����������������տ����е�ˮ�Ͷ�����̼�����³�ȡ��������������ƫС�����ʵ����ʵ���ƫС����ҺŨ��ƫ�ͣ�
�ʴ�Ϊ��ƫ�ͣ�
�ڶ���ҡ�Ⱥ���������Һ�������������ʵ����ʵ���ƫС����ҺŨ��ƫ�ͣ�
�ʴ�Ϊ��ƫ�ͣ�
���� ���⿼����һ�����ʵ���Ũ����Һ�����ƣ���ȷ����ԭ�������������ǽ���ؼ���ע���������ķ�������Ŀ�ѶȲ���

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д���1��SiO2+2C $\frac{\underline{\;����\;}}{\;}$Si���ֹ裩+2CO
��2��Si���֣�+2Cl2$\frac{\underline{\;����\;}}{\;}$SiCl4
��3��SiCl4+2H2$\frac{\underline{\;����\;}}{\;}$ Si ������+4HCl
������������Ӧ�����У�����ȷ���ǣ�������
| A�� | ��1����3��Ϊ�û���Ӧ | B�� | ��1����2����3����Ϊ������ԭ��Ӧ | ||
| C�� | ��2��Ϊ���Ϸ�Ӧ | D�� | ��Ӧ���й�Ԫ�ؾ�����ԭ |
| A�� | NH4Cl�ĵ���ʽ��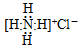 | |
| B�� | �������ױ���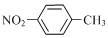 | |
| C�� | ̼��ĵ��뷽��ʽ��H2CO3?2H++CO${\;}_{3}^{2-}$ | |
| D�� | �����ӵĽṹʾ��ͼ��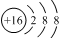 |
| A�� | �����ӵĽṹʾ��ͼ��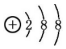 | B�� | ��ϩ�Ľṹ��ʽΪCH2CH2 | ||
| C�� | ��������ĵ���ʽ��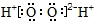 | D�� | H2O���ӵı���ģ�� |
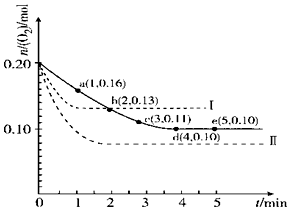 һ���¶��£���2L���ܱ������г���0.4mol SO2��0.2mol O2��������Ӧ��2SO2��g��+O2��g��?2SO3��g����H��0��n��O2����ʱ��ı仯��ϵ��ͼʵ����ʾ��
һ���¶��£���2L���ܱ������г���0.4mol SO2��0.2mol O2��������Ӧ��2SO2��g��+O2��g��?2SO3��g����H��0��n��O2����ʱ��ı仯��ϵ��ͼʵ����ʾ��
 ��ͼ��ʾ�����ʼ��ת����ϵ����֪A��HΪ���ӻ����H���������������ӵĸ�����Ϊ2��1���������������D��EΪ���ʣ�
��ͼ��ʾ�����ʼ��ת����ϵ����֪A��HΪ���ӻ����H���������������ӵĸ�����Ϊ2��1���������������D��EΪ���ʣ�