��Ŀ����
��֪��A��B��C��DΪ���ڱ�1��36���е�Ԫ�أ����ǵ�ԭ������������A�Ļ�̬ԭ����3����ͬ���ܼ������ܼ��е�������ȣ�C�Ļ�̬ԭ��2p�ܼ��ϵ�δ�ɶԵ�������Aԭ����ͬ��D�Ļ�̬ԭ�ӵ�M���Ӳ�����4��δ�ɶԵ��ӣ�
��ش��������⣺
��1��D ��Ԫ�����ڱ��е� ���ڣ��� ���Ԫ�أ����̬ԭ�ӵ���Χ�����Ų�ʽΪ ��
��2��A��B��C��D����Ԫ���У��縺�������� ����Ԫ�ط��ţ���A��B��C��һ������������ ����Ԫ�ط��ţ���
��3��B���⻯��ķе�Զ����A���⻯�����Ҫԭ���� ��
��4��D��AC��5�����³�Һ̬���۵�Ϊ-20.5�棬�е�Ϊ103�棬�����ڷǼ����ܼ����ݴ˿��ж�D��AC��5�������� ��������ͣ���
��ش��������⣺
��1��D ��Ԫ�����ڱ��е�
��2��A��B��C��D����Ԫ���У��縺��������
��3��B���⻯��ķе�Զ����A���⻯�����Ҫԭ����
��4��D��AC��5�����³�Һ̬���۵�Ϊ-20.5�棬�е�Ϊ103�棬�����ڷǼ����ܼ����ݴ˿��ж�D��AC��5��������
���㣺λ�ýṹ���ʵ����ϵӦ��
ר�⣺Ԫ����������Ԫ�����ڱ�ר��
������A��B��C��DΪ���ڱ�1��36���е�Ԫ�أ����ǵ�ԭ������������A�Ļ�̬ԭ����3����ͬ���ܼ������ܼ��е�������ȣ���������Ų�Ϊ1s22s22p2����AΪCԪ�أ�C�Ļ�̬ԭ��2p�ܼ��ϵ�δ�ɶԵ�������Aԭ����ͬ����������Ų�Ϊ1s22s22p4����CΪOԪ�أ�BԪ��ԭ����������̼����֮�䣬��BΪNԪ�أ�D�Ļ�̬ԭ�ӵ�M���Ӳ�����4��δ�ɶԵ��ӣ���������Ų�Ϊ1s22s22p63s23p63d64s2����DΪFe���ݴ˽��
���
�⣺A��B��C��DΪ���ڱ�1��36���е�Ԫ�أ����ǵ�ԭ������������A�Ļ�̬ԭ����3����ͬ���ܼ������ܼ��е�������ȣ���������Ų�Ϊ1s22s22p2����AΪ̼Ԫ�أ�C�Ļ�̬ԭ��2p�ܼ��ϵ�δ�ɶԵ�������Aԭ����ͬ����������Ų�Ϊ1s22s22p4����CΪOԪ�أ�BԪ��ԭ����������̼����֮�䣬��BΪNԪ�أ�D�Ļ�̬ԭ�ӵ�M���Ӳ�����4��δ�ɶԵ��ӣ���������Ų�Ϊ1s22s22p63s23p63d64s2����DΪFe��
��1��DΪFeԪ�أ����ڵ������ڵڢ����壬�������26�����ӣ����̬ԭ�ӵ���Χ�����Ų�ʽΪ3d64s2��
�ʴ�Ϊ���ģ�����3d64s2��
��2��ͬ����������ҵ縺�����ǽ�����Խǿ�縺��Խ������Ԫ���У��縺��������O���ǽ�����Խǿ����һ������Խ��������NԪ�صõ�2p������ڰ����״̬���ʵ�һ�����ܵ���������
�ʴ�Ϊ��O��N��
��3������NH3���Ӽ����γ��������е����CH4��
�ʴ�Ϊ��NH3���Ӽ����γ������
��4��Fe��Co��5�����³�Һ̬���۵�Ϊ-20.5�棬�е�Ϊ103�棬�����ڷǼ����ܼ����ݴ˿��ж�fE��CO��5�������ڷ��Ӿ��壬
�ʴ�Ϊ�����Ӿ��壮
��1��DΪFeԪ�أ����ڵ������ڵڢ����壬�������26�����ӣ����̬ԭ�ӵ���Χ�����Ų�ʽΪ3d64s2��
�ʴ�Ϊ���ģ�����3d64s2��
��2��ͬ����������ҵ縺�����ǽ�����Խǿ�縺��Խ������Ԫ���У��縺��������O���ǽ�����Խǿ����һ������Խ��������NԪ�صõ�2p������ڰ����״̬���ʵ�һ�����ܵ���������
�ʴ�Ϊ��O��N��
��3������NH3���Ӽ����γ��������е����CH4��
�ʴ�Ϊ��NH3���Ӽ����γ������
��4��Fe��Co��5�����³�Һ̬���۵�Ϊ-20.5�棬�е�Ϊ103�棬�����ڷǼ����ܼ����ݴ˿��ж�fE��CO��5�������ڷ��Ӿ��壬
�ʴ�Ϊ�����Ӿ��壮
�����������Ƕ����ʽṹ�Ŀ��飬�漰��������Ų����縺�ԡ��������͵��жϵȣ���Ŀ�ѵ��еȣ�������Ԫ�����ʼ�ԭ�ӽṹ���ۺϿ��飬��ȷԪ��ԭ�ӵĵ����Ų��ص��ǽ���Ĺؼ���

��ϰ��ϵ�д�
 ͬ����ϰǿ����չϵ�д�
ͬ����ϰǿ����չϵ�д�
�����Ŀ
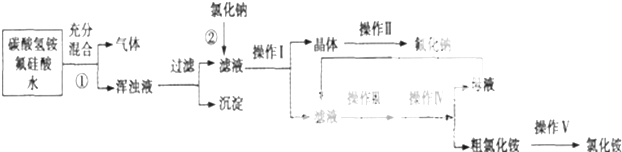
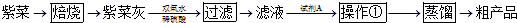
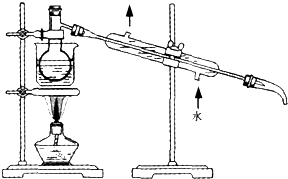
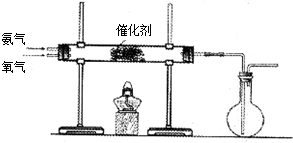
 ������Դ�뻷������Խ��Խ�����ǹ�ע��̼һ��ѧ��C1��ѧ����Ϊ�о����ȵ㣮��̼һ��ѧ�����Ե���̼��CO��CO2��CH4��CH3OH�Ⱥ�һ��̼ԭ�ӵ�����Ϊԭ�Ϻϳɹ�ҵ��Ʒ�Ļ�ѧ�빤�գ�
������Դ�뻷������Խ��Խ�����ǹ�ע��̼һ��ѧ��C1��ѧ����Ϊ�о����ȵ㣮��̼һ��ѧ�����Ե���̼��CO��CO2��CH4��CH3OH�Ⱥ�һ��̼ԭ�ӵ�����Ϊԭ�Ϻϳɹ�ҵ��Ʒ�Ļ�ѧ�빤�գ�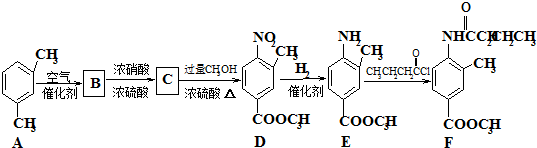
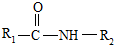 ��һ�������¿�ˮ��Ϊ
��һ�������¿�ˮ��Ϊ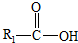 ��R2-NH2����F��ǿ��ͳ�ʱ����������·���ˮ�ⷴӦ�Ļ�ѧ����ʽ��
��R2-NH2����F��ǿ��ͳ�ʱ����������·���ˮ�ⷴӦ�Ļ�ѧ����ʽ��