��Ŀ����
�����仯����Ӧ�ù㷺�����磬�þ������ữ����������������CrO3���轺����˾�����������壬���ݹ轺��ɫ�ı仯�����ж�˾���Ƿ�Ƽݣ�

��1����̬��ԭ�ӵĵ����Ų�ʽΪ ��
��2����ͼ1�Dz�������Ԫ�ص�һ�������ݶ�ͼ��ͼ�У�a���Ӧ��Ԫ��Ϊ�⣬b��c�����Ӧ��Ԫ�طֱ�Ϊ �� ����Ԫ�ط��ţ���
��3�����Թ����У��Ҵ�������Ϊ��ȩ��CH3CHO�����Ҵ�����ȩ����Է������������Ҵ��ķе㣨78.5�棩ȴ����ȩ�ķе㣨20.8�棩�߳����࣬��ԭ���� ��
��4���Ȼ�������CrO2Cl2��������Ⱦ���ܼ����۵�Ϊ-96.5�棬�е�Ϊ117�棬�������Ȼ�̼���л��ܼ����ܣ��Ȼ������������� ��������ͣ���
��5����CrCl3?6H2O�ܽ�������ˮ�еõ�����ɫ��Һ����Һ��Cr3+��[Cr��H2O��5Cl]2+?��ʽ���ڣ�
��������Һ�У������ڵ������������� �����ţ���
A�����Ӽ� B�����ۼ� C�������� D����λ�� E�����»���
��[Cr��H2O��5Cl]2+��Cr3+�Ĺ���ӻ���ʽ����sp3�������� ��
��6����������Һ�У�H2O2�����ظ���������������ɫ��CrO5�����ӷ���ʽΪ��
4H2O2+2H++Cr2O72-�T2CrO5+5H2O
����ͼ2��ʾCr2O72-��CrO5�Ľṹ�жϣ�������Ӧ ����ǡ����ǡ���������ԭ��Ӧ��

��1����̬��ԭ�ӵĵ����Ų�ʽΪ
��2����ͼ1�Dz�������Ԫ�ص�һ�������ݶ�ͼ��ͼ�У�a���Ӧ��Ԫ��Ϊ�⣬b��c�����Ӧ��Ԫ�طֱ�Ϊ
��3�����Թ����У��Ҵ�������Ϊ��ȩ��CH3CHO�����Ҵ�����ȩ����Է������������Ҵ��ķе㣨78.5�棩ȴ����ȩ�ķе㣨20.8�棩�߳����࣬��ԭ����
��4���Ȼ�������CrO2Cl2��������Ⱦ���ܼ����۵�Ϊ-96.5�棬�е�Ϊ117�棬�������Ȼ�̼���л��ܼ����ܣ��Ȼ�������������
��5����CrCl3?6H2O�ܽ�������ˮ�еõ�����ɫ��Һ����Һ��Cr3+��[Cr��H2O��5Cl]2+?��ʽ���ڣ�
��������Һ�У������ڵ�������������
A�����Ӽ� B�����ۼ� C�������� D����λ�� E�����»���
��[Cr��H2O��5Cl]2+��Cr3+�Ĺ���ӻ���ʽ����sp3��������
��6����������Һ�У�H2O2�����ظ���������������ɫ��CrO5�����ӷ���ʽΪ��
4H2O2+2H++Cr2O72-�T2CrO5+5H2O
����ͼ2��ʾCr2O72-��CrO5�Ľṹ�жϣ�������Ӧ
���㣺ԭ�Ӻ�������Ų�,Ԫ�ص����ܡ��縺�Եĺ��弰Ӧ��,�����ijɼ����,ԭ�ӹ���ӻ���ʽ���ӻ������ж�,��ͬ����Ľṹ��������������������
ר�⣺��ѧ���뾧��ṹ
��������1��Crԭ�Ӻ�����24�����ӣ����ݹ���ԭ����д���������Ų�ʽ��
��2��ͬһ����Ԫ���У�Ԫ�ص�һ����������ԭ����������������������ƣ�����IIA�塢��VA��Ԫ�ص�һ�����ܴ�������Ԫ�أ�ͬһ����Ԫ���У���һ����������ԭ�������������С��
��3������Ĵ��ڵ������ʵ��۷е����ߣ�
��4�����Ӿ�����۷е�ϵͣ�
��5������Һ�зǽ���Ԫ��֮����ڹ��ۼ������йµ��ӶԺͺ��пչ����ԭ��֮�������λ��������֮����ڷ��»�����
�����Cr3+��ȡsp3�ӻ�����ֻ��4���ɼ���������γ�6����λ����
��6������Crԭ���γɵĻ�ѧ���жϣ�
��2��ͬһ����Ԫ���У�Ԫ�ص�һ����������ԭ����������������������ƣ�����IIA�塢��VA��Ԫ�ص�һ�����ܴ�������Ԫ�أ�ͬһ����Ԫ���У���һ����������ԭ�������������С��
��3������Ĵ��ڵ������ʵ��۷е����ߣ�
��4�����Ӿ�����۷е�ϵͣ�
��5������Һ�зǽ���Ԫ��֮����ڹ��ۼ������йµ��ӶԺͺ��пչ����ԭ��֮�������λ��������֮����ڷ��»�����
�����Cr3+��ȡsp3�ӻ�����ֻ��4���ɼ���������γ�6����λ����
��6������Crԭ���γɵĻ�ѧ���жϣ�
���
�⣺��1��Crԭ�Ӻ�����24�����ӣ����ݹ���ԭ��֪���������Ų�ʽΪ��[Ar]3d54s1��1s22s22p63s23p63d54s1���ʴ�Ϊ��[Ar]3d54s1��1s22s22p63s23p63d54s1��
��2��ͬһ����Ԫ���У�Ԫ�ص�һ����������ԭ����������������������ƣ�����IIA�塢��VA��Ԫ�ص�һ�����ܴ�������Ԫ�أ�ͬһ����Ԫ���У���һ����������ԭ�������������С������b��OԪ�ء�c��SiԪ�أ��ʴ�Ϊ��O��Si��
��3���Ҵ�����֮�����γ����������ȩ����֮�䲻���γ����������Ĵ��ڵ����Ҵ��۷е������ȩ���ʴ�Ϊ���Ҵ����Ӽ�������������ȩ���Ӽ䲻���������
��4�����Ӿ�����۷е�ϵͣ��Ȼ�������CrO2Cl2�����۷е�ϵͣ�����Ϊ���Ӿ��壬�ʴ�Ϊ�����Ӿ��壻
��5����A���������ˮ��Һ�������������ӣ����Ը������в��������Ӽ�����ѡ��
B��ˮ���ӻ�[Cr��H2O��5Cl]2+�У��ǽ���Ԫ��֮�䶼���ڹ��ۼ����ʲ�ѡ��
C������Һ�в����ڽ���������ѡ��
D��[Cr��H2O��5Cl]2+��Crԭ�Ӻ�ˮ�����е�Oԭ��֮�������λ�����ʲ�ѡ��
E����Һ��ˮ����֮����ڷ��»������ʲ�ѡ��
��ѡA��C��
��[Cr��H2O��5Cl]2+��Cr3+����λ����6�����Cr3+��ȡsp3�ӻ�����ֻ��4���ɼ���������γ�6����λ��������Cr3+�Ĺ���ӻ���ʽ����sp3��
�ʴ�Ϊ�����Cr3+��ȡsp3�ӻ�����ֻ��4���ɼ���������γ�6����λ����
��6������CrO5�Ľṹ�ɿ�����CrΪ+6�ۣ���4��OΪ-1�ۣ�һ��OΪ-2�ۣ���Ӧǰ��Ļ��ϼ�û�иı䣬���Բ���������ԭ��Ӧ���ʴ�Ϊ�����ǣ�
��2��ͬһ����Ԫ���У�Ԫ�ص�һ����������ԭ����������������������ƣ�����IIA�塢��VA��Ԫ�ص�һ�����ܴ�������Ԫ�أ�ͬһ����Ԫ���У���һ����������ԭ�������������С������b��OԪ�ء�c��SiԪ�أ��ʴ�Ϊ��O��Si��
��3���Ҵ�����֮�����γ����������ȩ����֮�䲻���γ����������Ĵ��ڵ����Ҵ��۷е������ȩ���ʴ�Ϊ���Ҵ����Ӽ�������������ȩ���Ӽ䲻���������
��4�����Ӿ�����۷е�ϵͣ��Ȼ�������CrO2Cl2�����۷е�ϵͣ�����Ϊ���Ӿ��壬�ʴ�Ϊ�����Ӿ��壻
��5����A���������ˮ��Һ�������������ӣ����Ը������в��������Ӽ�����ѡ��
B��ˮ���ӻ�[Cr��H2O��5Cl]2+�У��ǽ���Ԫ��֮�䶼���ڹ��ۼ����ʲ�ѡ��
C������Һ�в����ڽ���������ѡ��
D��[Cr��H2O��5Cl]2+��Crԭ�Ӻ�ˮ�����е�Oԭ��֮�������λ�����ʲ�ѡ��
E����Һ��ˮ����֮����ڷ��»������ʲ�ѡ��
��ѡA��C��
��[Cr��H2O��5Cl]2+��Cr3+����λ����6�����Cr3+��ȡsp3�ӻ�����ֻ��4���ɼ���������γ�6����λ��������Cr3+�Ĺ���ӻ���ʽ����sp3��
�ʴ�Ϊ�����Cr3+��ȡsp3�ӻ�����ֻ��4���ɼ���������γ�6����λ����
��6������CrO5�Ľṹ�ɿ�����CrΪ+6�ۣ���4��OΪ-1�ۣ�һ��OΪ-2�ۣ���Ӧǰ��Ļ��ϼ�û�иı䣬���Բ���������ԭ��Ӧ���ʴ�Ϊ�����ǣ�
���������⿼����ԭ�Ӻ�������Ų�ʽ����д��ԭ���ӻ���ʽ���жϡ������֪ʶ�㣬���ݹ���ԭ�����۲���ӶԻ������ۡ�������������ʵ�Ӱ���֪ʶ�����������ͬʱ����ѧ����֪ʶ���ճ̶ȣ��ѵ��ǣ�6��������ݳɼ��жϻ��ϼۣ��ܶ�ͬѧ����ֻ���ݻ��ϼ۵Ĵ�����Ϊ0ȷ�����ϼۣ�û�дӽṹ�Ϸ��������´���Ϊ�״��㣮

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
15.100mL 6mol/L H2SO4������п�۷�Ӧ����һ���¶��£�Ϊ�˼�����Ӧ���е����ʣ����ֲ�Ӱ����������������������Ӧ���м��������ģ�������
| A��̼���ƹ��� |
| B�������ƹ��� |
| C����������Һ |
| D���������Һ |
ijһ���л���ĽṹΪ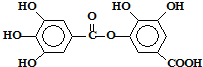 �������������ʵ������в���ȷ���ǣ�������
�������������ʵ������в���ȷ���ǣ�������
 �������������ʵ������в���ȷ���ǣ�������
�������������ʵ������в���ȷ���ǣ�������| A��������ˮ�⣬ˮ�����ֻ��һ�� |
| B��1mol���л�������ܺ�8molNaOH��Ӧ |
| C��1mol���л�������ܺ�4molBr2������Ӧ |
| D��1mol���л�������ܺ�8molH2�����ӳɷ�Ӧ |
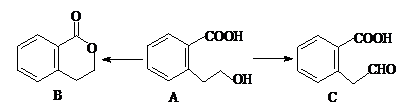
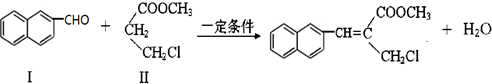
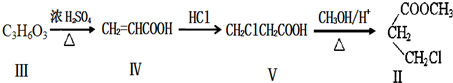
 ��������ȵ���Ƭ��ͭƬ�õ�����������500mL����ͭ��Һ�й�����ͼ��װ�ã������¾����跴Ӧ��������Һ������䣩��
��������ȵ���Ƭ��ͭƬ�õ�����������500mL����ͭ��Һ�й�����ͼ��װ�ã������¾����跴Ӧ��������Һ������䣩��