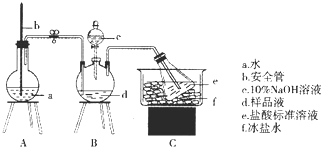
��Ŀ����
18���±�ΪԪ�����ڱ���һ���֣���ش��������⣺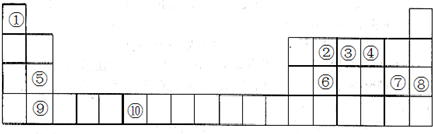
��1������Ԫ���У�����s������H��Mg��Ca����Ԫ�ط��ţ���
��2��д��Ԫ�آ�Ļ�̬ԭ�ӵĵ����Ų�ʽ[Ar]3d54s1��
��3��Ԫ�ص�һ������Ϊ�ݴ��ڢᣨ����ڡ���С�ڡ�����
��4��Ԫ�آ���̬�⻯�������ԭ����1�Ի��Ե��ӣ���VSEPRģ��Ϊ�����壻����ԭ�ӵ��ӻ�����Ϊsp3���÷���Ϊ���Է��ӣ�����ԡ��Ǽ��ԡ�������AgNO3��Һ����μ�����ˮ��Һ���ɹ۲쵽������Ϊ�Ȳ�����ɫ���������ܽ�����ܽ⣮
���� ����Ԫ�������ڱ��е�λ��֪���١���ֱ��Ǣ�H����C����N����O����Mg����Si����Cl����Ar����Ca����CrԪ�أ��ɴ˷������
��1���������Ų������������ӵ��ܼ�Ϊs�ܼ���Ϊs������Ԫ�����ڱ��а����ڢ�A�塢�ڢ�A�壻
��2��Ԫ�آ���CrԪ�أ����̬ԭ�Ӽ۵���Ϊ3d��4s�ܼ��ϵ��ӣ�3d��4s�ܼ��Ϸֱ���5��1�����ӣ�
��3��ͬ�������϶��µ�һ�����ܽ��ͣ�
��4��Ԫ�آ���̬�⻯��ΪNH3��Nԭ�Ӽ۲���Ӷ�Ϊ4���ӻ������Ϊ4��
�ж�ABn�ͷ��Ӽ��Ծ�����ɣ�������ԭ��A�Ļ��ϼ۵ľ���ֵ���ڸ�Ԫ�����ڵ�������������Ϊ�Ǽ��Է��ӣ������ȣ���Ϊ���Է��ӣ���AgNO3��Һ����μ�����ˮ��Һ����AgNO3��Һ����μ��백ˮ�������ӺͰ�ˮ��Ӧ���ɰ�ɫ����������������笠����ӣ����ӷ�Ӧ����ʽΪ��Ag++NH3•H2O�TAgOH��+NH4+���������백ˮ��ɫ�����ܽ⣬���������Ͱ�ˮ��Ӧ����������Һ��ˮ�����ӷ�Ӧ����ʽΪ��AgOH+2NH3•H2O�T[Ag��NH3��2]OH+2H2O���ߵα���ֱ�ε������ܽ⣮
��� �⣺��1����ͼ��֪�٢ݢᴦ��s�����ɢ�λ�ڵ�һ���ڵڢ�A�壬����Ԫ�أ���λ�ڵ������ڵڢ�A�壬��þԪ�أ���λ�ڵ������ڵڢ�A�壬�Ǹ�Ԫ�أ��ʴ�Ϊ��H��Mg��Ca��
��2��Ԫ�آ���CrԪ�أ����̬ԭ�Ӽ۵���Ϊ3d��4s�ܼ��ϵ��ӣ�3d��4s�ܼ��Ϸֱ���5��1�����ӣ�������۵����Ų�ʽΪ[Ar]3d54s1���ʴ�Ϊ��[Ar]3d54s1����3��Ԫ�آ���ᴦ��ͬһ���壬Ԫ�آ�˵���������Ը�ǿ����һ������С���ʴ�Ϊ�����ڣ�
��4��Ԫ�آ�ΪNԪ�أ�����̬�⻯��ΪNH3������ԭ����1�Թ¶Ե��ӣ�Nԭ�Ӽ۲���Ӷ�Ϊ4����VSEPRģ��Ϊ�������壻
Nԭ�ӵ��ӻ������Ϊ4������Nԭ�Ӳ�ȡsp3�ӻ���
NH3�ռ�ṹ���Գƣ�NԪ�ػ��ϼ۾���ֵ����3����NH3�Ǽ��Է��ӣ���AgNO3��Һ����μ��백ˮ�������ӺͰ�ˮ��Ӧ���ɰ�ɫ����������������笠����ӣ����ӷ�Ӧ����ʽΪ��Ag++NH3•H2O�TAgOH��+NH4+���������백ˮ��ɫ�����ܽ⣬���������Ͱ�ˮ��Ӧ����������Һ��ˮ�����ӷ�Ӧ����ʽΪ��AgOH+2NH3•H2O�T[Ag��NH3��2]OH+2H2O���ߵα���ֱ�ε������ܽ⣬�ʴ�Ϊ��1�������壻sp3�����ԣ��Ȳ�����ɫ���������ܽ�����ܽ⣮
���� ������Ҫ����Ԫ�����ڱ��ṹ����������Ų�ʽ�������ܡ��ӻ���������Ӽ��Եȣ������漰��֪ʶ��϶࣬�ۺ��Խϴ��ѶȽϴ�ע���˶����ʽṹ�г���������ۺϣ�

| A�� | 7.8 g������̼̼˫������ĿΪ0.3NA | |
| B�� | ��״���£�������Fe��2.24 L Cl2��Ӧת�Ƶ��ӵ���ĿΪ0.3NA | |
| C�� | 1.6 g CH4���й��õ��ӶԵ���ĿΪ0.5NA | |
| D�� | 100mLpH=2��������ˮ�У���N��OH-��+2N��ClO-��+N��HClO��=0.001NA |
| A�� | ������A��ʾ�ķ�Ӧ��ƽ������Ϊ0.6 mol•��L•s��-1 | |
| B�� | 2 sʱ����B��Ũ��Ϊ1.4 mol•L-1 | |
| C�� | 2 sʱ����A��ת����Ϊ70% | |
| D�� | 2 sʱ����A���������Ϊ$\frac{14}{27}$ |
| A�� | ���ڼ��� | B�� | ����̼���� | C�� | �������� | D�� | ������ʽ�� |
| A�� | CH2=CH2��CH2=CH-CH=CH2 | B�� | C4H8��C6H12 | ||
| C�� | 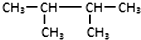 �ͣ�CH3��2CHCH��CH3��2 �ͣ�CH3��2CHCH��CH3��2 | D�� | C2H6��C4H10 |
�ٸ�����ӦΪO2+2H2O+4e-=4OH-
�ڸ�����ӦΪ2H2+4OH--4e-=4H2O
�۵�ع���ʱ������pH���ߣ�������pH�½���
�ܵ�ع���ʱ��Һ�е�����������������
| A�� | �٢ۢ� | B�� | �ڢ� | C�� | �ڢ� | D�� | �٢� |
| A�� | �Ȳ��ǵ����Ҳ���Ƿǵ���� | |
| B�� | ����ȼ�յIJ���ɵ������� | |
| C�� | ���������ȼ�ϵ�ص�ȼ�� | |
| D�� | �������IJ�ͬ������ȫȼ�գ�����ĺ��������� |

 ��
��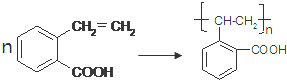 ��
��