��Ŀ����
ͭ���ʼ��仯�����ںܶ������ж�����Ҫ����;����ش��������⣺
��1����ϸͭ�ۿ�����������ϡ������ȣ����Ʊ��������£�

��NH4CuSO3�н��������ӵĺ�������Ų�ʽΪ ��N��O��S����Ԫ�صĵ�һ�����ܴ�С˳��Ϊ ����Ԫ�ط��ţ���
����CuSO4��Һ�м��������ˮ��������[Cu��NH3��4]SO4������˵����ȷ����
A��������������ˮ��ԭ��֮һ��NH3���Ӻ�H2O����֮���γ������Ե��
B��NH3���Ӻ�H2O���ӣ����ӿռ乹�Ͳ�ͬ���������ӵļ���С��ˮ���ӵļ���
C��[Cu��NH3��4]SO4��Һ�м����Ҵ�������������ɫ�ľ���
D����֪3.4g��������������ȫȼ����������Ⱦ�����壬���ų�aKJ��������NH3��ȼ���ȷ���ʽΪ��NH3��g��+
O2��g��=
N2��g��+
H2O��g����H=-5aKJ/mol
��2��ͭ�������CuMn2O4�����ڳ����´����������е�������Ϊ��������SO2��Ϊ�ȵ����壩�����ݵȵ�����ԭ����O3���ӵĿռ乹��Ϊ ��
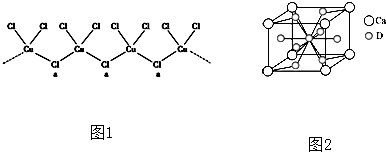
��3���Ⱥͼ��벻ͬ��̬��ͭ���������ֻ�����������Ӿ�Ϊ�������ṹ����ͼ1��ʾ����aλ����Clԭ�ӣ�����һ����λ�������ӻ��������Ϊ ��
��4����ͼ2�ǽ���Ca��D���γɵ�ij�ֺϽ�ľ����ṹʾ��ͼ����֪�����Ͻ�������Ca-D�Ͻ�����ͬ���͵ľ����ṹXYn�������к�ǿ�Ĵ�����������֪�����Ͻ�LaNin�������Ϊ9.0��10-23 cm3��������γ�LaNinH4.5�Ͻ�����뾧����϶��������䣩����LaNin��n= ������ֵ�������ںϽ��е��ܶ�Ϊ ��������λ��Ч���֣�
��1����ϸͭ�ۿ�����������ϡ������ȣ����Ʊ��������£�

��NH4CuSO3�н��������ӵĺ�������Ų�ʽΪ
����CuSO4��Һ�м��������ˮ��������[Cu��NH3��4]SO4������˵����ȷ����
A��������������ˮ��ԭ��֮һ��NH3���Ӻ�H2O����֮���γ������Ե��
B��NH3���Ӻ�H2O���ӣ����ӿռ乹�Ͳ�ͬ���������ӵļ���С��ˮ���ӵļ���
C��[Cu��NH3��4]SO4��Һ�м����Ҵ�������������ɫ�ľ���
D����֪3.4g��������������ȫȼ����������Ⱦ�����壬���ų�aKJ��������NH3��ȼ���ȷ���ʽΪ��NH3��g��+
| 3 |
| 4 |
| 1 |
| 2 |
| 3 |
| 2 |
��2��ͭ�������CuMn2O4�����ڳ����´����������е�������Ϊ��������SO2��Ϊ�ȵ����壩�����ݵȵ�����ԭ����O3���ӵĿռ乹��Ϊ
��3���Ⱥͼ��벻ͬ��̬��ͭ���������ֻ�����������Ӿ�Ϊ�������ṹ����ͼ1��ʾ����aλ����Clԭ�ӣ�����һ����λ�������ӻ��������Ϊ
��4����ͼ2�ǽ���Ca��D���γɵ�ij�ֺϽ�ľ����ṹʾ��ͼ����֪�����Ͻ�������Ca-D�Ͻ�����ͬ���͵ľ����ṹXYn�������к�ǿ�Ĵ�����������֪�����Ͻ�LaNin�������Ϊ9.0��10-23 cm3��������γ�LaNinH4.5�Ͻ�����뾧����϶��������䣩����LaNin��n=

���㣺ԭ�Ӻ�������Ų�,�жϼ��ӻ����ӵĹ���,�����ļ���,ԭ�ӹ���ӻ���ʽ���ӻ������ж�
ר�⣺ԭ�������ṹר��,��ѧ���뾧��ṹ
��������1�������жϽ������ӵĻ��ϼۣ��ٸ��ݸ��ݺ�������Ų�ʽ����д������д��ע��3d�ܼ�����������4s�ܼ���������ʧ����ʱ����ʧȥ������ϵĵ��ӣ����ݵ�һ�����ܵı仯���ɱȽ����С��
��A������������ˮ����֮��������������Ĵ���ʹ���ʵ��ܽ�����������
B���ݷ��ӵĿռ�ṹ�жϣ�
C��������������ԭ���жϣ�
D��ȼ���ȷ���ʽ��д�ڳ����½��У�H2OΪҺ̬��
��2���������ӻ������жϣ�
��3��aλ����Clԭ�ӳ�2������������2�Թ¶Ե��ӣ��ӻ������Ϊ4���ݴ��жϣ�
��4�������侧���Ķѻ������жϼ��㣮
��A������������ˮ����֮��������������Ĵ���ʹ���ʵ��ܽ�����������
B���ݷ��ӵĿռ�ṹ�жϣ�
C��������������ԭ���жϣ�
D��ȼ���ȷ���ʽ��д�ڳ����½��У�H2OΪҺ̬��
��2���������ӻ������жϣ�
��3��aλ����Clԭ�ӳ�2������������2�Թ¶Ե��ӣ��ӻ������Ϊ4���ݴ��жϣ�
��4�������侧���Ķѻ������жϼ��㣮
���
�⣺��1����ͭ��29��Ԫ�أ������������ԭ����̬ԭ�ӵĵ����Ų�ʽΪ1s22s22p63s23p63d104s1��[Ar]3d104s1����ԭ��ʧȥһ������ʱ��������ͭ���ӣ�ʧȥ�ĵ������������ӣ�������ͭ���ӵĻ�̬�ĵ����Ų�ʽΪ��1S22S22P63S23P63d10��[Ar]3d10�� Ԫ���������У�ͬһ����Ԫ�صĵ�һ����������ԭ������������������ڢ�A�͵ڢ�A���ڢ�A�͵ڢ�A������ͬһ����Ԫ�صĵ�һ����������ԭ���������������С���������һ�����ܴ�СΪN��O��S��
�ʴ�Ϊ��1s22s22p63s23p63d10��[Ar]3d10��N��O��S��
��A������������ˮ����֮��������������Ĵ���ʹ���ʵ��ܽ�����������A��ȷ��
B���������ӵļ���С��ˮ���ӵļ��ǣ���B����
C�������������ܷ������Ҵ����Ӽ�������ˮ�ļ��ԣ����Ҵ����ܽ�Ƚ��ͣ���C��ȷ��
D��ȼ���ȷ���ʽ��д�ڳ����½��У�H2OΪҺ̬����D����
�ʴ�Ϊ��AC��
��2��SO2�۵��Ӷ���=
=3���۵��Ӷ���=��λԭ����+�µ��Ӷ�������λԭ����Ϊ2������һ�Թ¶Ե��ӣ�ԭ�ӹ���ռ乹��Ϊƽ���ͣ����ӹ���ΪV�Σ�������SO2��Ϊ�ȵ����壬�ʴ�Ϊ��V�Σ�
��3��aλ����Clԭ�ӳ�2������������2�Թ¶Ե��ӣ��ӻ������Ϊ4���ӻ��������Ϊ��sp3���ʴ�Ϊ��sp3��
��4���ɾ����ṹ��֪��Caԭ�Ӵ��ڶ��㣬�����к���Caԭ����ĿΪ8��
=1��Cuԭ�Ӵ��ھ����ڲ������ϡ����ģ�������Cu��ĿΪ1+4��
+4��
=5���ʸúϽ���Ca��Cu��ԭ�Ӹ�����Ϊ1��5����n=5��������ӵ��4.5��Hԭ�ӣ����ںϽ��е��ܶ�Ϊ��=��4.5/6.02��1023���£�9.0��10-23��=0.083g/cm3��
�ʴ�Ϊ��5��0.083 g?cm-3��
�ʴ�Ϊ��1s22s22p63s23p63d10��[Ar]3d10��N��O��S��
��A������������ˮ����֮��������������Ĵ���ʹ���ʵ��ܽ�����������A��ȷ��
B���������ӵļ���С��ˮ���ӵļ��ǣ���B����
C�������������ܷ������Ҵ����Ӽ�������ˮ�ļ��ԣ����Ҵ����ܽ�Ƚ��ͣ���C��ȷ��
D��ȼ���ȷ���ʽ��д�ڳ����½��У�H2OΪҺ̬����D����
�ʴ�Ϊ��AC��
��2��SO2�۵��Ӷ���=
| 6 |
| 2 |
��3��aλ����Clԭ�ӳ�2������������2�Թ¶Ե��ӣ��ӻ������Ϊ4���ӻ��������Ϊ��sp3���ʴ�Ϊ��sp3��
��4���ɾ����ṹ��֪��Caԭ�Ӵ��ڶ��㣬�����к���Caԭ����ĿΪ8��
| 1 |
| 8 |
| 1 |
| 2 |
| 1 |
| 2 |
�ʴ�Ϊ��5��0.083 g?cm-3��
������������Ԫ�ص��ƶ�Ϊ���壬�����˵����ܡ��ӻ����ۣ����ӽṹ����������Ų����ɡ����������֪ʶ�㣬�Ƕ����ʽṹ֪ʶ���ۺϿ��飬���ض�֪ʶǨ�Ƶ����á��������������������飬ע��ͬһ����Ԫ�صĵ�һ����������ԭ�����������������������ƣ�����IIA�͵�IIIA�塢��VA��͵�VIA��֮���쳣��

��ϰ��ϵ�д�
 ���ɶ��ȫ���ƿؾ�ϵ�д�
���ɶ��ȫ���ƿؾ�ϵ�д�
�����Ŀ
���������ӡ��⡱ʳ�ν϶�ʹ���˵���أ�KIO3�ڹ�ҵ�Ͽ��õ�ⷨ��ȡ����ʯī�Ͳ����Ϊ�缫����KI��Һ��������K2CrO4��Ϊ���Һ��һ�������µ�⣬��Ӧ����ʽΪKI+3H2O
KIO3+3H2���������й�˵����ȷ���ǣ�������
| ��� |
| A�����ʱ��ʯī������������������� |
| B�����Һ������ǿ���ԱȽϺ��� |
| C���������У�I-�������ƶ�������IO3- |
| D�������ÿת��0.6mol���ӣ������Ͽɵõ�21.4g��ˮKIO3���� |
�������й㷺����;�������к�������Fe��Zn��Cu��Pt�����ʣ����õ�ⷨ�Ʊ��ߴ��ȵ�����������������ȷ���ǣ���֪��������Fe2+��Ni2+��Cu2+����������
| A������������ԭ��Ӧ����缫��ӦʽΪ��Ni2++2e-�TNi |
| B���������У����������ļ������������������Ӳ���ȣ�������������ʧ������Ŀ��� |
| C��������Һ�д��ڵĽ�����������Ni2+��Fe2+��Zn2+ |
| D�������۵ײ�������������Cu��Pt |
�����й�ԭ�ӽṹ��Ԫ�������������в���ȷ���ǣ�������
| A����IA������Ԫ������������ˮ����ļ�����ԭ��������������ǿ |
| B��ͬ����Ԫ�أ���0��Ԫ���⣩�����ң�ԭ�Ӱ뾶�����Ӱ뾶����С |
| C���ڢ�AԪ�ش��ϵ��£����⻯����ȶ������� |
| D���ڢ�A��Ԫ����ͬ�����зǽ�������ǿ��Ԫ�� |
���и���˵���в���ȷ���ǣ�������
| A��SO2��NO2��CO���̳����Ǵ�����Ⱦ�� |
| B����֧�ֱ�պ��Ũ�����Ũ��ˮ�IJ������������������ |
| C��ֻ����ˮ����������Ҵ������ᡢ��������ɫҺ�� |
| D��һ�����ļ��������������·�����Ӧ�������������� |
���з�Ӧ���ٷ���ͼ�������ص㣬�ڶ�����з�������������ȷ�ģ������ѡ��Ϊ��������
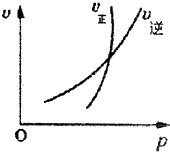

| A��2NO2��g��?N2O4��g����ƽ�������ѹǿ��������ɫ�ȱ�����dz |
| B��N2��g��+3H2��g��?2NH3��g����������Һ������������ڼӿ�����Ӧ���� |
| C��4NH3��g��+5O2��g��?4NO��g��+6H2O��g�����Ӵ�����Ũ�ȣ�����߰���ƽ��ת���� |
| D��2HI��g��?H2��g��+I2��g����ƽ����ֳ���һ����HI���ٴ�ƽ��ʱHI��ת���ʲ��� |
��ѧʵ���г�����Һ���Լ������ữ�������ữ�����Ĵ�ʩ��ȷ���ǣ�������
| A��������Һ���Ƿ���SO32-ʱ����HNO3�ữ���ټ�BaCl2��Һ |
| B��Ϊ���KMnO4��Һ�������������������ữKMnO4��Һ |
| C������ij��Һ�Ƿ���Fe3+ʱ����HNO3�ữ��KSCN��Һ |
| D��������Һ���Ƿ���SO42-ʱ����HCl�ữ��BaCl2��Һ |
���淴Ӧ H2 ��g��+I2 ��g��?2HI��g�����¶�һ������1L���ܱ������м���2molH2��g����1molI2��g����ʼ��Ӧ��2minĩ��֪������������1.2mol HI��g��������˵����ȷ���ǣ�������
| A��H2��g��������������I2��g������������ʼ����� |
| B����Ӧ�����У�H2��g����I2��g����ת����ʼ����� |
| C��ƽ��ǰ�������淴Ӧ���ʵı�ֵ�㶨 |
| D����Ӧ��ʼ��2minĩ��v��H2��=0.3mol?L-1?min-1 |
