��Ŀ����
�����Ȼ�ѧ����ʽ�����ӷ���ʽ�У���ȷ���ǣ�������
| A������ı�ȼ����Ϊ-890.3kJ?mol-1�������ȼ�յ��Ȼ�ѧ����ʽ�ɱ�ʾΪ��CH4��g��+2O2��g���TCO2��g��+2H2O��g����H=-890.3 kJ?mol-1 | ||||
B��500�桢30 MPa�£���0.5 mol N2��1.5 mol H2�����ܱ������г�ַ�Ӧ����NH3��g��������19.3 kJ�����Ȼ�ѧ����ʽΪ��N2��g��+3H2��g��
| ||||
| C���Ȼ�þ��Һ�백ˮ��Ӧ��Mg2++2OH-�TMg��OH��2�� | ||||
| D������������NaOH��Һ��Al2O3+2OH-+3H2O�T2Al��OH��4- |
���㣺���ӷ���ʽ����д,�Ȼ�ѧ����ʽ
ר�⣺
������A��0.5mol N2��1.5mol H2�����ܱյ������г�ַ�Ӧ����������ȫ��Ӧ������1mol N2��3mol H2�����ܱյ���������ȫ��Ӧ�ų�����������19.3kJ��2��
B������ı�ȼ���������ɵ�ˮӦ��ΪҺ�壻
C��һˮ�ϰ�Ϊ������ӷ���ʽ��һˮ�ϰ����ܲ�
D��������Ϊ����������ܹ�������������Һ��Ӧ����Al��OH��4-��
B������ı�ȼ���������ɵ�ˮӦ��ΪҺ�壻
C��һˮ�ϰ�Ϊ������ӷ���ʽ��һˮ�ϰ����ܲ�
D��������Ϊ����������ܹ�������������Һ��Ӧ����Al��OH��4-��
���
�⣺A��0.5mol N2��1.5mol H2�����ܱյ������г�ַ�Ӧ����������ȫ��Ӧ�����Ȼ�ѧ����ʽ�еķ�Ӧ�ȣ���ʾ1mol N2��3mol H2�����ܱյ���������ȫ��Ӧ�ų�����������19.3kJ��2=38.6 kJ?mol-1����A����
B��Һ��ˮ����̬ˮ�ȶ�����Ӧ���ɵ�Ӧ��ΪҺ̬ˮ�����Լ����ȼ���ȵ��Ȼ�ѧ����ʽ�ɱ�ʾΪ��CH4��g��+2O2��g���TCO2��g��+2H2O��l����H=-890.3kJ?mol-1����B����
C���Ȼ�þ��Һ�백ˮ��Ӧ����������þ�������Ȼ�泥���ȷ�����ӷ���ʽΪ��Mg2++2NH3?H2O=Mg��OH��2��+2NH4+����C����
D������������NaOH��Һ�����ӷ���ʽΪ��Al2O3+2OH-+3H2O�T2Al��OH��4-����D��ȷ��
��ѡD��
B��Һ��ˮ����̬ˮ�ȶ�����Ӧ���ɵ�Ӧ��ΪҺ̬ˮ�����Լ����ȼ���ȵ��Ȼ�ѧ����ʽ�ɱ�ʾΪ��CH4��g��+2O2��g���TCO2��g��+2H2O��l����H=-890.3kJ?mol-1����B����
C���Ȼ�þ��Һ�백ˮ��Ӧ����������þ�������Ȼ�泥���ȷ�����ӷ���ʽΪ��Mg2++2NH3?H2O=Mg��OH��2��+2NH4+����C����
D������������NaOH��Һ�����ӷ���ʽΪ��Al2O3+2OH-+3H2O�T2Al��OH��4-����D��ȷ��
��ѡD��
���������⿼�������ӷ���ʽ���Ȼ�ѧ����ʽ����д�����жϣ���Ŀ�Ѷ��еȣ���ȷ���ӷ���ʽ����ѧ����ʽ����дԭ��Ϊ���ؼ�������������ѧ�����Ӧ�û���֪ʶ��������

��ϰ��ϵ�д�
�����Ŀ
��֪��ӦCO��g���TC��s��+
O2��g���ġ�H��0����S��0�����H �͡�S �����¶ȶ��䣬����˵������ȷ���ǣ�������
| 1 |
| 2 |
| A���κ��¶��¶��Ƿ��Է��仯 |
| B�����������Է����� |
| C���������Ƿ��Է��仯�����������Է��仯 |
| D�����������Է����� |
����Ԫ������Ԫ�����ڱ���λ�ã��ж�����Ԫ�ض�����Ϊ�뵼����ϵ��ǣ�������
| A��Si��K | B��C��Al |
| C��Si��Ge | D��As��Se |
���и������ʵ����ʱȽϣ���ȷ���ǣ�������
| A��ԭ�Ӱ뾶��O��S��Na��K |
| B���⻯���ȶ��ԣ�H2S��HF��H2O |
| C�����ԣ�NaOH��Mg��OH��2��Al��OH��3 |
| D����ԭ�ԣ�F-��Cl-��Br-��I- |
���л�ѧ�����ʾ��ȷ���ǣ�������
A��H2O��Na2O�ĵ���ʽ������ ��ʾ ��ʾ |
B��Cl��Br��I��+1�ۺ�����Ľṹʽ�����á� ����ʾ ����ʾ |
| C�����ᡢ���������������ǵķ���ʽ�����á�CnH2nOn����ʾ |
| D��̼�ᡢ������ĵ��뷽��ʽ�����á�H2XO3?2H++XO32-����ʾ |
���е���ʽ��д������ǣ�������
A�� |
B�� |
C��[ H]- H]- |
D��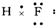 |
���и������ʣ������ʡ�����������˳�����е��ǣ�������
| A����ʯ�ҡ����ס���ʯ�� |
| B��Һ̬������ơ��ռ� |
| C�������Ȼ��⡢�ɱ� |
| D������������������ |
