��Ŀ����
15��±�����ڼ��Դ���Һ���ܷ�����ȥ��Ӧ�����磬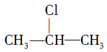 +NaOH$��_{��}^{��}$CH3-CH�TCH2+NaCl+H2O��
+NaOH$��_{��}^{��}$CH3-CH�TCH2+NaCl+H2O���÷�ӦʽҲ�ɱ�ʾΪ
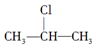 $��_{-NaCl��-H_{2}O}^{NaOH��������}$CH3-CH�TCH2�����Ǽ����л��������ת����ϵ��
$��_{-NaCl��-H_{2}O}^{NaOH��������}$CH3-CH�TCH2�����Ǽ����л��������ת����ϵ��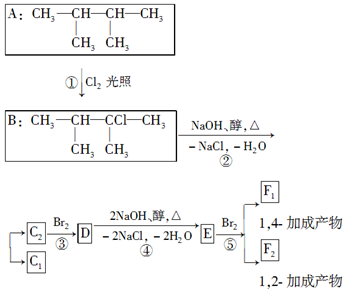
��1������ϵͳ��������������A��������2��3-�������飮
��2��������ͼ�У�����ȡ����Ӧ�����Ǽӳɷ�Ӧ��
��3��������E����Ҫ�Ĺ�ҵԭ�ϣ�д����D����E�Ļ�ѧ����ʽ��CH3CBr��CH3��CBr��CH3��2+2NaOH$��_{��}^{��}$CH2=C��CH3��C��CH3��=CH2+2NaBr+2H2O��
��4��C2�Ľṹ��ʽ�ǣ�CH3��2C=C��CH3��2��F1�Ľṹ��ʽ��BrCH2C��CH3��=C��CH3��CH2Br��F1��F2��Ϊͬ���칹�壮
���� A�ڹ�������������������ȡ����Ӧ����B��B���������ƴ���Һ�����������·�����ȥ��Ӧ����C1��C2��C2���巢���ӳɷ�Ӧ����D��D���������ƴ���Һ�����������·�����ȥ��Ӧ����E��E������Է���1��2-�ӳɷ�Ӧ����F2������1��4-�ӳɷ�Ӧ����F1����EΪCH2=C��CH3��C��CH3��=CH2��F2ΪCH2=C��CH3��CBr��CH3��CH2Br��F1ΪBrCH2C��CH3��=C��CH3��CH2Br�����ƿɵ�DΪCH3CBr��CH3��CBr��CH3��2��C2Ϊ��CH3��2C=C��CH3��2��C1Ϊ��CH3��2CHC��CH3��=CH2��
��� �⣺��1���ɻ�����A�Ľṹ��ʽ���������ǣ�2��3-�������飬�ʴ�Ϊ��2��3-�������飻
��2��������ͼ�У���Ӧ������ȡ����Ӧ����Ӧ�����ڼӳɷ�Ӧ���ʴ�Ϊ��ȡ�����ӳɣ�
��3���������Ϸ�����D����E�Ļ�ѧ����ʽ��CH3CBr��CH3��CBr��CH3��2+2NaOH$��_{��}^{��}$CH2=C��CH3��C��CH3��=CH2+2NaBr+2H2O��
�ʴ�Ϊ��CH3CBr��CH3��CBr��CH3��2+2NaOH$��_{��}^{��}$CH2=C��CH3��C��CH3��=CH2+2NaBr+2H2O��
��4���������Ϸ�����C2�Ľṹ��ʽ�ǣ���CH3��2C=C��CH3��2��F1�Ľṹ��ʽ�ǣ�BrCH2C��CH3��=C��CH3��CH2Br��F2�Ľṹ��ʽ�ǣ�CH2=C��CH3��CBr��CH3��CH2Br��F1��F2��Ϊͬ���칹��
�ʴ�Ϊ����CH3��2C=C��CH3��2��BrCH2C��CH3��=C��CH3��CH2Br��ͬ���칹�壮
���� ���⿼���л�����ƶϣ��Ѷ��еȣ��������չ����ŵ�������ת���ǹؼ�����ѧ������������һ����Ҫ��

| A�� | ϡ�������ͭƬ�ϣ�Cu+2H+�TCu2++H2�� | |
| B�� | ����þ��ϡ�����ϣ�MgO+2H+�TMg2++H2O | |
| C�� | ͭƬ������������Һ�У�Cu+Ag+�TCu2++Ag | |
| D�� | ϡ������ڴ���ʯ�ϣ�CO32-+2H+�TCO2��+H2O |
| A�� | �������ᣬ������ʹ����ʯ��ˮ����ǵ���ɫ���壬��������һ����CO32- | |
| B�� | ��AgCl�����е���ϡKI��Һ��ɫ������ƣ�˵��AgI��AgCl������ | |
| C�� | �ȼ��������������ټ���BaCl2��Һ������ɫ��������������һ����SO42- | |
| D�� | ����Һ�м���NaOH�ȣ�����ʹʪ��ĺ�ɫʯ����ֽ���������壬��һ������� |
| A�� | ��ˮ������ᡢ��ȩ������ | |
| B�� | ��ȥ��������������ϩ������������ͨ������ | |
| C�� | ��ȥ�Ҵ������������������������ʯ�ң����� | |
| D�� | ��ȼ�յķ��������Ҵ����������Ȼ�̼ |
��NH3��H2O��
��NH4+��H3O+��
��NH3��H3O+��
��O3��SO2��
��CO2��BeCl2��
| A�� | ȫ�� | B�� | �ۢܢ� | C�� | �������� | D�� | �ڢ� |
| A�� | Զ���ֵ��������п��ɱ����ִ����ܸ�ʴ | |
| B�� | ���������ڳ�ʪ���������� | |
| C�� | ��п��Ƭ�ȶ�����Ƭ����ʴ | |
| D�� | �������ڿ����в��ױ���ʴ |
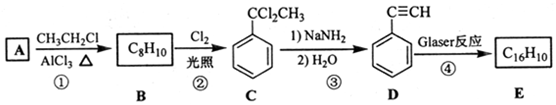
 ��D �Ļ�ѧ����Ϊ����Ȳ��
��D �Ļ�ѧ����Ϊ����Ȳ�� ����1mol E�ϳ�1��4-���������飬��������Ҫ��������4mol��
����1mol E�ϳ�1��4-���������飬��������Ҫ��������4mol��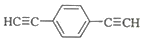 ��Ҳ�ɷ���Glaserż����Ӧ���ɾۺ���þۺϷ�Ӧ�Ļ�ѧ����ʽΪn
��Ҳ�ɷ���Glaserż����Ӧ���ɾۺ���þۺϷ�Ӧ�Ļ�ѧ����ʽΪn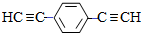 $\stackrel{����}{��}$
$\stackrel{����}{��}$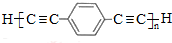 +��n-1��H2��
+��n-1��H2��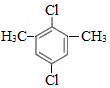 ��
�� ��
��