��Ŀ����
14������˵����ȷ���ǣ�������| A�� | ��һ���¶���AgClˮ��Һ�У�Ag+��Cl-Ũ�ȵij˻���һ������ | |
| B�� | AgCl��Ksp=1.8��10-10 mol2•L-2�����κκ�AgCl�������Һ�У�c��Ag+��=c��Cl-����Ag+��Cl-Ũ�ȵij˻�����1.8��10-10 mol2•L-2 | |
| C�� | �¶�һ��ʱ������Һ��Ag+��Cl-Ũ�ȵij˻�����Kspֵʱ������ҺΪAgCl�ı�����Һ | |
| D�� | ��2.0mLŨ�Ⱦ�Ϊ0.1mol•L-1��KCl��KI�����Һ�еμ�1��2��0.01mol•L-1 AgNO3��Һ���������ʻ�ɫ��˵��AgCl��Ksp��AgI��KspС |
���� A���粻�DZ�����Һ����Ag+��Cl-Ũ�ȵij˻����dz�����
B�������Ȼ��ƻ���������Һc��Ag+����c��Cl-������ȣ�
C��һ���¶��£��ܶȻ�Ϊ������
D��������AgI������˵��AgI��KspС��
��� �⣺A����һ���¶��µ�AgCl�ı���ˮ��Һ�У�Ksp=C��Ag+��C��Cl-����һ���������ڲ�������Һ��Ag+��Cl-Ũ�ȵij˻�����һ����������A����
B��AgCl��Ksp=1.8��10-10 mol2•L-2�����κκ�AgCl�������Һ�У�c��Ag+����һ������c��Cl-�������Ȼ�����Һ��c��Ag+����c��Cl-������Ag+��Cl-Ũ�ȵij˻�����1.8��10-10����B����
C���¶�һ��ʱ��Ũ����QC=Kspֵʱ������ҺΪ AgCl�ı�����Һ����C��ȷ��
D��KspԽСԽ�����ɳ�����������AgI������˵��AgI��KspС����D����
��ѡC��
���� ������Ҫ����������ܵ���ʵ��ܶȻ�Ksp�������Լ�Ӱ�����أ�Ϊ��Ƶ���㣬������ѧ���ķ��������Ŀ��飬ע��������ܵ���ʵ��ܽ�ƽ���Ӱ�������Լ�ƽ���ƶ��ص㣬�ѶȲ���

��ϰ��ϵ�д�
 ȫ��������ϵ�д�
ȫ��������ϵ�д� һ��һ����ʱ���ϵ�д�
һ��һ����ʱ���ϵ�д�
�����Ŀ
9����2L�������ʵ�����Ϊ0.1mol��Ca2+��Mg2+��ij��Һ�У�����һ������NaOH����ʹ��Һ��pH=ll���г������ɣ����ˣ���֪��KSP[Ca��OH��2]=4.68��10-6��KSP[Mg��OH��2]=5.61��10-12��������Ũ��С��10-5mol•L-1��Ϊ�Ѿ�����������˵����ȷ���ǣ�������
| A�� | �˳�����Mg��OH��2��Ca��OH��2�Ļ���� | |
| B�� | �˳���ֻ��Mg��OH��2 | |
| C�� | ���˺���Һ�д���Ca2+��Mg2+ | |
| D�� | ��ԭ��Һ�м���8.8 g NaOH���壬������ʹCa2+����ת��Ϊ���� |
19����0.2000mol/L�ı�NaOH��Һ�ζ�δ֪Ũ�ȵ����ᣬ������ɷ�Ϊ���¼�����
��������ˮϴ�Ӽ�ʽ�ζ��ܣ�ע��0.2000mol/L�ı�NaOH��Һ����0���̶������ϣ��ڹ̶��õζ��ܲ�ʹ�ζ��ܼ������Һ�壻�۵���Һ������0����0���̶������£������¶���������ȡ20.00mL����Һע��ྻ����ƿ�У�������3�η�̪��Һ�����ñ�Һ�ζ����յ㣬���µζ���Һ����������ظ����ϵζ�����2-3�Σ���ش�
��1�����ϲ����д�����ǣ����ţ��٣��ô�������ᵼ�²ⶨ���ƫ�� ���ƫ����ƫС������Ӱ�족����
��2��������У���ȡ20.00mL����ҺӦʹ����ʽ�ζ��ܣ����������ƣ�������ƿװҺǰ��������������ˮ���ⶨ�����Ӱ�죨�ƫ����ƫС������Ӱ�족����
��3������ݵζ�ʱ�۾�Ӧע����ƿ����Һ��ɫ�仯���жϵ���ζ��յ�������ǣ���ƿ����Һ����ɫ��Ϊdz��ɫ������Ӳ���ɫ��
��4��������ʵ�����ݼ�¼��
�ӱ��п��Կ�������1�εζ���¼��NaOH��Һ������Զ��ں����ε����������ܵ�ԭ����AB
A���ζ�ǰ�ζ��ܼ��������ݣ��ζ�����������
B����ƿ�ô���Һ��ϴ
C��NaOH��Һ����ʱ��������в��ֱ���
D���ζ�����ʱ�����Ӽ���
��5�����ݱ��м�¼���ݣ�ͨ������ɵã�������Ũ��Ϊ��0.1626mol/L��
��������ˮϴ�Ӽ�ʽ�ζ��ܣ�ע��0.2000mol/L�ı�NaOH��Һ����0���̶������ϣ��ڹ̶��õζ��ܲ�ʹ�ζ��ܼ������Һ�壻�۵���Һ������0����0���̶������£������¶���������ȡ20.00mL����Һע��ྻ����ƿ�У�������3�η�̪��Һ�����ñ�Һ�ζ����յ㣬���µζ���Һ����������ظ����ϵζ�����2-3�Σ���ش�
��1�����ϲ����д�����ǣ����ţ��٣��ô�������ᵼ�²ⶨ���ƫ�� ���ƫ����ƫС������Ӱ�족����
��2��������У���ȡ20.00mL����ҺӦʹ����ʽ�ζ��ܣ����������ƣ�������ƿװҺǰ��������������ˮ���ⶨ�����Ӱ�죨�ƫ����ƫС������Ӱ�족����
��3������ݵζ�ʱ�۾�Ӧע����ƿ����Һ��ɫ�仯���жϵ���ζ��յ�������ǣ���ƿ����Һ����ɫ��Ϊdz��ɫ������Ӳ���ɫ��
��4��������ʵ�����ݼ�¼��
�ӱ��п��Կ�������1�εζ���¼��NaOH��Һ������Զ��ں����ε����������ܵ�ԭ����AB
A���ζ�ǰ�ζ��ܼ��������ݣ��ζ�����������
| �ζ� ���� | ������� ��mL�� | NaOH��Һ���������mL�� | |
| �ζ�ǰ | �ζ��� | ||
| 1 | 20.00 | 0.00 | 18.10 |
| 2 | 20.00 | 0.00 | 16.30 |
| 3 | 20.00 | 0.00 | 16.22 |
C��NaOH��Һ����ʱ��������в��ֱ���
D���ζ�����ʱ�����Ӽ���
��5�����ݱ��м�¼���ݣ�ͨ������ɵã�������Ũ��Ϊ��0.1626mol/L��
4��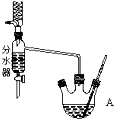 ʵ������������������Ʊ��������������й����ʵ�������������
ʵ������������������Ʊ��������������й����ʵ�������������
�������£�
����50mL������ƿ��Ͷ�뼸����ʯ����18.5mL��������13.4mL�����ᣨ��������3��4��Ũ���ᰴһ��˳����Ȼ�ϣ���װ��ˮ�������ã�ʵ������в��Ϸ����ȥ��Ӧ���ɵ�ˮ�����¶ȼƼ����������ܣ�
�ڽ���ˮ���ֳ�������ͷ�ӦҺһ�����Һ©����������ˮϴ��10% Na2CO3ϴ����ˮϴ�����ת������ƿ�����
�۽�����������������������ƿ�У���ѹ�����ռ���֣���15.1g������������
��ش��й����⣺
��1��д������һ��������ͬ���ͬ���칹��Ľṹ��ʽCH3CH2CH��OH��CH3��
��2������A�з�����Ӧ�Ļ�ѧ����ʽΪCH3COOH+CH3CH2CH2CH2OH$?_{��}^{Ũ����}$CH3COOCH2CH2CH2CH3+H2O��
��3���������������ƿ�����μ����ҩƷ�ǣ���������Ũ���ᣬ�����ᣮ
��4��������У��� 10%Na2CO3��Һϴ���л��㣬�ò�������Ŀ���dz�ȥ���л��е��������������
��5��������ڽ����������ʱ������118�濪ʼ�ռ���֣�����ƫ�ߣ�����ߡ��͡���ԭ���ǻ��ռ�������δ��Ӧ�ı��������������
��6����ʵ�����ɵ������������IJ�����65%��
 ʵ������������������Ʊ��������������й����ʵ�������������
ʵ������������������Ʊ��������������й����ʵ�������������| ������ | ��Է������� | �ܶ�/g•cm-3 | �е�/�� | �ܽ��g/l00gˮ |
| ������ | 74 | 0.80 | 118.0 | 9 |
| ������ | 60 | 1.045 | 118.1 | ���� |
| ���������� | 116 | 0.882 | 126.1 | 0.7 |
����50mL������ƿ��Ͷ�뼸����ʯ����18.5mL��������13.4mL�����ᣨ��������3��4��Ũ���ᰴһ��˳����Ȼ�ϣ���װ��ˮ�������ã�ʵ������в��Ϸ����ȥ��Ӧ���ɵ�ˮ�����¶ȼƼ����������ܣ�
�ڽ���ˮ���ֳ�������ͷ�ӦҺһ�����Һ©����������ˮϴ��10% Na2CO3ϴ����ˮϴ�����ת������ƿ�����
�۽�����������������������ƿ�У���ѹ�����ռ���֣���15.1g������������
��ش��й����⣺
��1��д������һ��������ͬ���ͬ���칹��Ľṹ��ʽCH3CH2CH��OH��CH3��
��2������A�з�����Ӧ�Ļ�ѧ����ʽΪCH3COOH+CH3CH2CH2CH2OH$?_{��}^{Ũ����}$CH3COOCH2CH2CH2CH3+H2O��
��3���������������ƿ�����μ����ҩƷ�ǣ���������Ũ���ᣬ�����ᣮ
��4��������У��� 10%Na2CO3��Һϴ���л��㣬�ò�������Ŀ���dz�ȥ���л��е��������������
��5��������ڽ����������ʱ������118�濪ʼ�ռ���֣�����ƫ�ߣ�����ߡ��͡���ԭ���ǻ��ռ�������δ��Ӧ�ı��������������
��6����ʵ�����ɵ������������IJ�����65%��
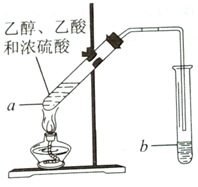 �����Ѿ��ܷⴢ��ʱ��Խ��������Խ�ã�ԭ��֮һ�Ǵ����������������ζ��������ʵ���ҿ�������ͼ��ʾ��װ����ȡ������������ش��������⣮
�����Ѿ��ܷⴢ��ʱ��Խ��������Խ�ã�ԭ��֮һ�Ǵ����������������ζ��������ʵ���ҿ�������ͼ��ʾ��װ����ȡ������������ش��������⣮