��Ŀ����
������A�����ϣ��������к�C 72.0%��H 6.67%������Ϊ��������������֪A����Է�������Ϊ150���ִ����������л�������ķ��ӽṹ���������ַ�����
����һ���˴Ź����Dz��A�ĺ˴Ź���������5���壬�����֮��Ϊ1��2��2��2��3��
����������������Dz��A���ӵĺ��������ͼ��ʾ��
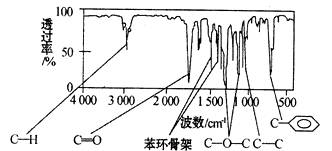
��1��A�ķ���ʽΪ________________��
��2����֪��A������ֻ��һ���������ұ�����ֻ��һ��ȡ������A����ˮ�⣬д����������������A�Ľṹ��ʽ________________��ֻдһ�֣���
��3����A������������ˮ��Ļ�ѧ����ʽΪ________________��
��1��C9H10O2
��2��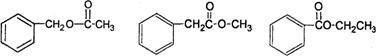 ��дһ��
��дһ��
��3����

��ϰ��ϵ�д�
�����Ŀ

 ������A�����ϣ��������к�C72.0%��H 6.67%�����ຬ����������������֪A����Է�������Ϊ150���ִ����������л�������ķ��ӽṹ���������ַ�����
������A�����ϣ��������к�C72.0%��H 6.67%�����ຬ����������������֪A����Է�������Ϊ150���ִ����������л�������ķ��ӽṹ���������ַ�����
 ����
���� ��
�� ��
��



