��Ŀ����
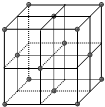 ̼�ж���ͬ�������壬�������н��ʯ��ʯī����C60������ϩ�����ӣ����ݱ���������ԱӦ�ü����ģ����ṹ����C60������N60���Ӻ�Si60���ӣ�
̼�ж���ͬ�������壬�������н��ʯ��ʯī����C60������ϩ�����ӣ����ݱ���������ԱӦ�ü����ģ����ṹ����C60������N60���Ӻ�Si60���ӣ���1��C��N��Si �ĵ�һ������ �Ӵ�С˳��
��2����֪N60������ÿ����ԭ�Ӿ���N-N���������Nԭ�Ӷ��γ�8�����ȶ��ṹ����Nԭ�ӵ��ӻ���ʽ��
��3��CH4��NH3��SiH4 ���۷е��ɵ͵��ߵ�˳����
��4��̼����Ԫ���γ�ԭ�Ӹ�����Ϊ1��3�ij��������Ʋ��������Ŀռ乹��Ϊ
��5��+1����̬��̬��������ʧȥһ�������γ�+2����̬��̬����������Ҫ��������Ϊ�ڶ�������I2�����λ���I3��I4��I5�����Ʋ�NԪ�ص� �� �� �� ͻ �� Ӧ �� �� �� ��
��6��ij�ֽ���Ԫ�غ��������29���˶�״̬������
���㣺�����ļ���,Ԫ�ص����ܡ��縺�Եĺ��弰Ӧ��,�жϼ��ӻ����ӵĹ���,ԭ�ӹ���ӻ���ʽ���ӻ������ж�
ר�⣺��ѧ���뾧��ṹ
��������1��ͬһ���ڴ����ң�Ԫ�صĵ�һ�����������VA��Ԫ�صĵ�һ�����ܴ�������Ԫ�أ�ͬһ������ϵ��£�Ԫ�صĵ�һ������С��
��2��N60����������Nԭ�Ӻ��Ҽ����Ӷ���Ϊ3�ԣ�����Nԭ�ӵĵ��Ӷ���=3+
=4��
�ṹ���Ƶķ��ӣ��������γɹ��ۼ���ԭ�Ӱ뾶ԽС������Խ����Խ�ȶ���
��3���ṹ���ƣ�Ħ������Խ��������۷е�Խ�ߣ������Ӽ�����������������Ӽ���������ʹ�۷е����ߣ�
��4��C��C�γ�ԭ�Ӹ�����Ϊ1��3�ij�������ΪCO32-������Cԭ�Ӽ۲���Ӷ�����µ��Ӷԣ�ȷ����ռ乹�ͣ�
��5����ͬ�ܲ���������������ܲ�����������ϴ�ʧȥ��ͬ�ܲ����ʱ�����ܻᷢ��ͻԾ��
��6����ԭ�Ӻ����ж��ٸ����Ӿ;��ж������˶�״̬��ij�ֽ���Ԫ�غ��������29���˶�״̬�����Ԫ��Ϊ29��Ԫ��Cu��
��2��N60����������Nԭ�Ӻ��Ҽ����Ӷ���Ϊ3�ԣ�����Nԭ�ӵĵ��Ӷ���=3+
| 5-1��3 |
| 2 |
�ṹ���Ƶķ��ӣ��������γɹ��ۼ���ԭ�Ӱ뾶ԽС������Խ����Խ�ȶ���
��3���ṹ���ƣ�Ħ������Խ��������۷е�Խ�ߣ������Ӽ�����������������Ӽ���������ʹ�۷е����ߣ�
��4��C��C�γ�ԭ�Ӹ�����Ϊ1��3�ij�������ΪCO32-������Cԭ�Ӽ۲���Ӷ�����µ��Ӷԣ�ȷ����ռ乹�ͣ�
��5����ͬ�ܲ���������������ܲ�����������ϴ�ʧȥ��ͬ�ܲ����ʱ�����ܻᷢ��ͻԾ��
��6����ԭ�Ӻ����ж��ٸ����Ӿ;��ж������˶�״̬��ij�ֽ���Ԫ�غ��������29���˶�״̬�����Ԫ��Ϊ29��Ԫ��Cu��
���
�⣺��1��ͬһ������ϵ��£�Ԫ�صĵ�һ������С�����Ե�һ�����ܴ�С��N��P��ͬһ����Ԫ���У�Ԫ�صĵ�һ����������ԭ������������������ʵ�һ�����ܴ�С��S��Si�������ڵ�VA��Ԫ��np3�����ڰ����״̬����һ�����ܴ�������Ԫ�أ��ʵ�һ�����ܴ�С��P��S�����ϵ�һ�����ܴ�С��N��S��Si��
�ʴ�Ϊ��N��S��Si��
��2��N60����������Nԭ�Ӻ��Ҽ����Ӷ���Ϊ3�ԣ�����Nԭ�ӵĵ��Ӷ���=3+
=4������Nԭ�ӵ��ӻ���ʽ��SP3��
�ṹ���Ƶķ��ӣ��������γɹ��ۼ���ԭ�Ӱ뾶ԽС������Խ����Խ�ȶ���Cԭ�Ӱ뾶С��Siԭ�Ӱ뾶��C-C�����ܴ���Si-Si�����ܣ�����C60 ���ȶ��Դ��� Si60��
�ʴ�Ϊ��SP3�����ڣ�
��3���ṹ���ƣ�Ħ������Խ��������۷е�Խ�ߣ����۷е�CH4��SiH4�������Ӽ�����������������Ӽ���������ʹ�۷е����ߣ���NH3�۷е���ߣ�
�ʴ�Ϊ��CH4��SiH4��NH3��NH3���Ӽ�������۷е���ߣ�CH4��SiH4 ���Ӽ�ֻ�з��»�������Է�������CH4��SiH4 ���»���CH4��SiH4��
��4��C��O�γ�ԭ�Ӹ�����Ϊ1��3�ij�������ΪCO32-��������Cԭ�ӵ��Ӷ���=3+
=3�����������Ŀռ乹��Ϊƽ�������Σ�
��CO32-��Ϊ�ȵ�����ķ�����SO3��BCl3 ��BF3��
�ʴ�Ϊ��ƽ�������Σ�SO3��BCl3 ��BF3��
��5����ͬ�ܲ���������������ܲ�����������ϴ�ʧȥ��ͬ�ܲ����ʱ�����ܻᷢ��ͻԾ��NԪ�ع��������ܲ㣬�ڶ��ܲ���5�����ӣ�����NԪ�صĵ�����ͻ�� Ӧ�����ڵ�ʧ����������ʱ����������������ͻ�䣻
�ʴ�Ϊ������
��6��ԭ�Ӻ����ж��ٸ����Ӿ;��ж������˶�״̬��ij�ֽ���Ԫ�غ��������29���˶�״̬�����Ԫ��Ϊ29��Ԫ��Cu�����̬ԭ�Ӻ�������Ų�ʽΪ��1s22s22p63s23p63d104s1�����Կ���ͭԭ�ӵĻ�̬ԭ�ӹ����s��p��d��������3����״��ͬ��ԭ�ӹ��������һ�����ӵ���Χ�����Ų�ʽΪ3d10������Cu������֪����ԭ�ӵ���λ��=3��8��
=12��
ÿ�������к��е�ԭ����=8��
+6��
=4����ÿ������������m=64g/mol��
=
��ÿ�����������V=��a��4��10-10��3cm3����Cu�ܶ�d=
=
����NA=
mol-1��
�ʴ�Ϊ��3�� 3d10��12��
mol-1��
�ʴ�Ϊ��N��S��Si��
��2��N60����������Nԭ�Ӻ��Ҽ����Ӷ���Ϊ3�ԣ�����Nԭ�ӵĵ��Ӷ���=3+
| 5-1��3 |
| 2 |
�ṹ���Ƶķ��ӣ��������γɹ��ۼ���ԭ�Ӱ뾶ԽС������Խ����Խ�ȶ���Cԭ�Ӱ뾶С��Siԭ�Ӱ뾶��C-C�����ܴ���Si-Si�����ܣ�����C60 ���ȶ��Դ��� Si60��
�ʴ�Ϊ��SP3�����ڣ�
��3���ṹ���ƣ�Ħ������Խ��������۷е�Խ�ߣ����۷е�CH4��SiH4�������Ӽ�����������������Ӽ���������ʹ�۷е����ߣ���NH3�۷е���ߣ�
�ʴ�Ϊ��CH4��SiH4��NH3��NH3���Ӽ�������۷е���ߣ�CH4��SiH4 ���Ӽ�ֻ�з��»�������Է�������CH4��SiH4 ���»���CH4��SiH4��
��4��C��O�γ�ԭ�Ӹ�����Ϊ1��3�ij�������ΪCO32-��������Cԭ�ӵ��Ӷ���=3+
| 4+2-2��3 |
| 2 |
��CO32-��Ϊ�ȵ�����ķ�����SO3��BCl3 ��BF3��
�ʴ�Ϊ��ƽ�������Σ�SO3��BCl3 ��BF3��
��5����ͬ�ܲ���������������ܲ�����������ϴ�ʧȥ��ͬ�ܲ����ʱ�����ܻᷢ��ͻԾ��NԪ�ع��������ܲ㣬�ڶ��ܲ���5�����ӣ�����NԪ�صĵ�����ͻ�� Ӧ�����ڵ�ʧ����������ʱ����������������ͻ�䣻
�ʴ�Ϊ������
��6��ԭ�Ӻ����ж��ٸ����Ӿ;��ж������˶�״̬��ij�ֽ���Ԫ�غ��������29���˶�״̬�����Ԫ��Ϊ29��Ԫ��Cu�����̬ԭ�Ӻ�������Ų�ʽΪ��1s22s22p63s23p63d104s1�����Կ���ͭԭ�ӵĻ�̬ԭ�ӹ����s��p��d��������3����״��ͬ��ԭ�ӹ��������һ�����ӵ���Χ�����Ų�ʽΪ3d10������Cu������֪����ԭ�ӵ���λ��=3��8��
| 1 |
| 2 |
ÿ�������к��е�ԭ����=8��
| 1 |
| 8 |
| 1 |
| 2 |
| 4 |
| NA |
| 256g/mol |
| NA |
| m |
| V |
| ||
| (a��4��10-10)3cm3 |
| 4��1030 |
| da3 |
�ʴ�Ϊ��3�� 3d10��12��
| 4��1030 |
| da3 |
���������⿼���˾����ļ��㡢��������Ų���ԭ�ӹ���ӻ���ʽ�����ݣ��ۺ��Խ�ǿ���ѶȽϴ�ע�����þ�̯������ļ��㣮

��ϰ��ϵ�д�
 ���ʿ��ÿ��ֳɳ�ϵ�д�
���ʿ��ÿ��ֳɳ�ϵ�д�
�����Ŀ
�����й�Ԫ�طǽ����ԵıȽϣ���ȷ���ǣ�������
| A������H2O��H2S���ȷֽ�ʵ�飬ȷ�����������Ԫ�صķǽ�����ǿ�� | ||
| B��HCl��Һ�����Ա�H2S��Һ������ǿ����Cl�ķǽ����Ա�Sǿ | ||
C��2C+SiO2
| ||
| D���ⶨNaCl��NaF��Һ��pH����ȷ��F��Cl��Ԫ�طǽ����Ե�ǿ�� |
����˵������ȷ���ǣ�������
| A�������½�CaO��Na2O��Na��NaOH��5�˷ֱ�Ͷ��100��ˮ�У���ֽ����������Һ�����ʵ�����������С����CaO |
| B�������顢�����顢��������۷е��ɸߵ��͵�˳��Ϊ�£��죾�� |
| C��AlO2-��HCO3-��Na+��K+�����Ӳ�����ˮ��Һ��������� |
| D����NaHCO3��Һ��Na2SO3��Һ�ֱ��������գ��õ��Ĺ��嶼����ԭ���� |
������ĵ��ӽṹ�У���һ��������С��ԭ�ӿ����ǣ�������
| A��ns2np3 |
| B��ns2np5 |
| C��ns1 |
| D��ns2np6 |
ֻ��һ���Լ����ܽ�BaCl2��NaCl��KOH������Һ�������Լ��ǣ�������
| A��Na2CO3��Һ |
| B������ |
| C������ |
| D��CuSO4��Һ |
