��Ŀ����
 ���ж��ֻ���������ᣨH3PO2���ʹ������ƣ�NaH2PO2����Ϊ���������еĻ�ԭ����
���ж��ֻ���������ᣨH3PO2���ʹ������ƣ�NaH2PO2����Ϊ���������еĻ�ԭ�������������գ�
��1��H3PO2��һԪ��ǿ�ᣬд������뷽��ʽ��
��2��������0.1mol/L��NaH2PO2��Һ��0.1mol/L��Na2CO3��Һ��pH�������
��3����ѧ������������H3PO2����Һ�е�Ag+��ԭΪ���ķ�Ӧ����֪�÷�Ӧ�л�ԭ���������������ʵ���֮��Ϊ1��4��������������
��4����ҵ�Ͽ��ð��ף�P4����Ba��OH��2��Һ��Ӧ����PH3��Ba��H2PO2��2��д������ƽ�÷�Ӧ�Ļ�ѧ����ʽ
����Ba��H2PO2��2��Һ�м��������Ʊ�H3PO2������˵Ӧ��������������ᣬ��˵�����������������ŵ㣺
��5����ͼ�����õ��ԭ���Ʊ�H3PO2��ʾ��ͼ�������ӽ���Ĥ�������ӽ���Ĥ�ֱ�ֻ��������������ͨ������֪�缫��ӦΪ��
���� 4OH--4e��O2��+H2O
���� 2H++2e��H2����
�����������ҵõ�H3PO2ԭ��
���㣺����ˮ���ԭ��,������ԭ��Ӧ,������ԭ��Ӧ����ʽ����ƽ,���ԭ��
ר�⣺
��������1������H3PO2��һԪ��ǿ���֪��H3PO2��������ʣ���Һ�в��ֵ���������ӣ��ݴ�д�����뷽��ʽ������H3PO2��һԪ��ǿ�ᣬ�����ж�NaH2PO2Ϊ���Σ���ǿ�������Σ�ˮ���Լ��ԣ�
��2��H3PO2��һԪ��ǿ�ᣬ̼�������ᣬ����Խ�����Ӧ�ε�ˮ��̶�Խ��
��3��H3PO2��AgNO3��Һ��Ӧ���л�ѧ�������˷�Ӧ���������뻹ԭ�������ʵ���֮��Ϊ4��1����ϻ��ϼ��жϲ��
��4�����ݷ�Ӧ�����������ԭ���غ�͵����غ���д����ʽ��
��5��������Ӧ������ˮ�����OH-��ʹc��H+������H2PO2-ͨ�������ӽ���Ĥ���������ң��õ���Ʒ��
��2��H3PO2��һԪ��ǿ�ᣬ̼�������ᣬ����Խ�����Ӧ�ε�ˮ��̶�Խ��
��3��H3PO2��AgNO3��Һ��Ӧ���л�ѧ�������˷�Ӧ���������뻹ԭ�������ʵ���֮��Ϊ4��1����ϻ��ϼ��жϲ��
��4�����ݷ�Ӧ�����������ԭ���غ�͵����غ���д����ʽ��
��5��������Ӧ������ˮ�����OH-��ʹc��H+������H2PO2-ͨ�������ӽ���Ĥ���������ң��õ���Ʒ��
���
�⣺��1��H3PO2��һԪ��ǿ�ᣬ��Һ�в��ֵ���������ӣ���������뷽��ʽΪH3PO2?H2PO2-+H+������H3PO2��һԪ��ǿ�ᣬ����NaH2PO2ΪһԪǿ���һԪ��ǿ���γɵ����Σ����Ը�����Һ���ڳ�H2PO2-����ˮ��ɼ��ԣ�����ʽΪH2PO2-+H2O?H3PO2+OH-��������Һ������Ũ���ɴ�С��˳��Ϊc��Na+����c��H2PO2-����c��OH-����c��H+����
�ʴ�Ϊ��H3PO2?H2PO2-+H+��c��Na+����c��H2PO2-����c��OH-����c��H+����
��2��H3PO2��һԪ��ǿ�ᣬ̼�������ᣬ����Խ�����Ӧ�ε�ˮ��̶�Խ��������ͬ���ʵ���Ũ�ȵ�Na2CO3��Һ�ļ��Ա�NaH2PO2��Һǿ��
�ʴ�Ϊ��Na2CO3��H3PO2����ǿ�ᣬ̼�������ᣬCO32-��ˮ��̶ȴ���H2PO2-��
��3���÷�Ӧ��Ag+Ϊ��������H3PO2Ϊ��ԭ�����������뻹ԭ�������ʵ���֮��Ϊ4��1���跴Ӧ������P�Ļ��ϼ�Ϊx�����ݻ��ϼ�������ȿɵã�4����1-0��=1����x-1�������x=5��������������Ϊ+5�۵�H3PO4��
�ʴ�Ϊ��H3PO4��
��4�����ף�P4����Ba��OH��2��Һ��Ӧ����PH3�����Ba��H2PO2��2����Ӧ����ʽΪ2P4+3Ba��OH��2+6H2O=3Ba��H2PO2��2+2PH3����ÿ����3molBa��H2PO2��2��6molP����������1.5molP4��������ת�Ƶ���6mol������������������ɵ����ᱵ�dz��������������H3PO2���룬
�ʴ�Ϊ��2P4+3Ba��OH��2+6H2O=3Ba��H2PO2��2+2PH3����1.5������������������ɵ����ᱵ�dz��������������H3PO2���룻
��5���ݵ�ⷴӦʽ��֪��������Ӧ������ˮ�����OH-��ʹc��H+������H2PO2-ͨ�������ӽ���Ĥ���������ң��õ���Ʒ��
�ʴ�Ϊ��������Ӧ������ˮ�����OH-��ʹc��H+������H2PO2-ͨ�������ӽ���Ĥ���������ң��õ���Ʒ��
�ʴ�Ϊ��H3PO2?H2PO2-+H+��c��Na+����c��H2PO2-����c��OH-����c��H+����
��2��H3PO2��һԪ��ǿ�ᣬ̼�������ᣬ����Խ�����Ӧ�ε�ˮ��̶�Խ��������ͬ���ʵ���Ũ�ȵ�Na2CO3��Һ�ļ��Ա�NaH2PO2��Һǿ��
�ʴ�Ϊ��Na2CO3��H3PO2����ǿ�ᣬ̼�������ᣬCO32-��ˮ��̶ȴ���H2PO2-��
��3���÷�Ӧ��Ag+Ϊ��������H3PO2Ϊ��ԭ�����������뻹ԭ�������ʵ���֮��Ϊ4��1���跴Ӧ������P�Ļ��ϼ�Ϊx�����ݻ��ϼ�������ȿɵã�4����1-0��=1����x-1�������x=5��������������Ϊ+5�۵�H3PO4��
�ʴ�Ϊ��H3PO4��
��4�����ף�P4����Ba��OH��2��Һ��Ӧ����PH3�����Ba��H2PO2��2����Ӧ����ʽΪ2P4+3Ba��OH��2+6H2O=3Ba��H2PO2��2+2PH3����ÿ����3molBa��H2PO2��2��6molP����������1.5molP4��������ת�Ƶ���6mol������������������ɵ����ᱵ�dz��������������H3PO2���룬
�ʴ�Ϊ��2P4+3Ba��OH��2+6H2O=3Ba��H2PO2��2+2PH3����1.5������������������ɵ����ᱵ�dz��������������H3PO2���룻
��5���ݵ�ⷴӦʽ��֪��������Ӧ������ˮ�����OH-��ʹc��H+������H2PO2-ͨ�������ӽ���Ĥ���������ң��õ���Ʒ��
�ʴ�Ϊ��������Ӧ������ˮ�����OH-��ʹc��H+������H2PO2-ͨ�������ӽ���Ĥ���������ң��õ���Ʒ��
���������⿼����������ʵĵ��뷽��ʽ��д������Ũ�ȴ�С�Ƚϡ�����ˮ����Խ��Խˮ��Ĺ��ɡ�������ԭ��Ӧ�еĵ����غ㡢������ԭ��Ӧ����ʽ��д�ȵȣ���Ŀ�ѶȽϴ�

��ϰ��ϵ�д�
�����Ŀ
95��ʱ����ˮ��H+�����ʵ���Ũ��Ϊ10-6 mol/L������0.01mol NaOH�����ܽ���95��ˮ�����1L��Һ������Һ����ˮ�������H+��Ũ�ȣ���λ��mol/L����������
| A��10-6 |
| B��10-10 |
| C��10-8 |
| D��10-12 |
���ж��й�֪ʶ��������ȷ���ǣ�������
| A�����ʵ��������������������� |
| B��1Ħ����������������NA��O2���ӵ���Է�������֮�� |
| C������٤�����������ǹ涨������û�е�λ |
| D��H2����Է���������1molH2��������H2��Ħ���������ֵ�λ��ͬ������ֵ����� |
�����йػ�ѧ���������ȷ���ǣ�������
A��Na2S�ĵ���ʽ��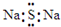 |
B��˳-2-��ϩ�Ľṹ��ʽ�� |
C��O2-�Ľṹʾ��ͼ�� |
| D����ϩ�Ľṹ��ʽ��CH2CH2 |
ij�л����������Է�������Ϊ90��ȡһ�������ĸ�������ȫȼ�����ɶ�����̼��ˮ�����ʵ�����ȣ��������������ɣ�����˵����ȷ���ǣ�������
| A���û�����϶���OԪ�� |
| B����ȷ���Ƿ���OԪ�� |
| C���û������п϶�����̼̼˫�� |
| D���û�����Ľṹֻ��һ�� |
�����������ʵ��л���������ͬ���칹����Ŀ�����ǣ������������칹����������
| A������ʽΪC4H8O�ҿ�������Ʒ�Ӧ�ų��������л������� |
| B������ʽΪC5H10O2����̼�����Ʒ�Ӧ�ų�������̼���л������� |
| C������ʽΪC5H10����ʹ������Ȼ�̼��Һ��ɫ���л������� |
| D��C6H14�зе���ߵ�����������Ӧ��һ�ȴ��л������� |
���������У��������������ǣ�������
| A��ʯī | B������KCl |
| C��ϡ���� | D������NaOH |
 ��1���������������������ҿ������������е��ʷ�Ӧ���磺6Ag��s��+O3��g��=3Ag2O��s����H1
��1���������������������ҿ������������е��ʷ�Ӧ���磺6Ag��s��+O3��g��=3Ag2O��s����H1