��Ŀ����
���ᡢ����������dz����ġ������ᡱ���־������������ͭ��Ӧ��������ش����⣺
(1)ϡ�����Cu ��Ӧ������ϡ�����м���H2O2�����ʹͭ˳���ܽ⡣�÷�Ӧ�Ļ�ѧ����ʽΪ��____________________________��
(2)��һ�������10mol/L��Ũ�����м������ͭƬ������ʹ֮��Ӧ������ԭ������Ϊ0.9mol����Ũ�����ʵ�����__________(����ڡ����� ���ڡ���С�ڡ�)180mL����ʹʣ���ͭƬ�����ܽ⣬�������м�����������Һ(��KNO3��Һ)����÷�Ӧ�����ӷ���ʽΪ_____________________��
(3)þ�������4.8g���ܽ��ڹ�����ijŨ�ȵ�ϡ�����У���ȫ��Ӧ��õ���״����2 .24LNO���塣����Ӧ�����Һ�м����������ռ������ɳ�����������_________�ˡ�
 ÿ�α���ϵ�д�
ÿ�α���ϵ�д� ��ѧ����ϵ�д�
��ѧ����ϵ�д�ﮱ���Ϊ������ζ��������LiCoO2Ϊ�������ϵ�����ӵ���ѱ��㷺������Яʽ��Դ����ҵ�ϳ��Ԧ�-﮻Կ�(��Ҫ�ɷ�ΪLiAlSi2O6��������FeO��MgO��CaO������)Ϊԭ������ȡ����ﮡ�����һ�ֹ���������ͼ��ʾ��
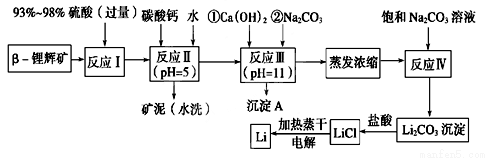
��֪���ٲ��ֽ����������↑ʼ��������ȫ����ʱ��pH���±���
�������� | Fe(OH)3 | Al(OH)3 | Mg(OH)2 |
��ʼ����pH | 2.7 | 3.7 | 9.6 |
��ȫ����pH | 3.7 | 4.7 | 11 |
��Li2CO3�ڲ�ͬ�¶��µ��ܽ�����±���
�¶�/�� | 0 | 10 | 20 | 50 | 75 | 100 |
Li2CO3���ܽ��/g | 1.539 | 1.406 | 1.329 | 1.181 | 0.866 | 0.728 |
��ش��������⣺
��1������������ʽ��ʾLiAlSi2O6����ɣ�________________________��
��2����Ӧ�����̼��Ƶ�������___________________��
��3������������ʹ��̼������Һ����˵��ǰ������ʹ����Ũ�Ȳ�ͬ��ԭ��______________��
��4��ϴ������Li2CO3����Ҫʹ��________(���ˮ������ˮ��)����ѡ���������________________��
��5����������Li2CO3��ȫ��Ӧ�����Һ���������ɵõ����壬�ٽ������ڵ������ﮡ����ʱ�����������л��������������ԭ���ǣ�__________________________��
��6�����й��ڽ���﮼��������˵����ȷ����___________������ţ���
A����Ǽ���������۵������ B�����ݶԽ���ԭ�����������ǿ��
C������﮵���������ʽʮ�ָ��� D����������ܶ���С����ﮣ��������


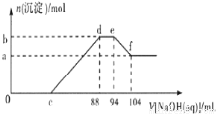

 NaCl��K�����÷�Ӧ����
NaCl��K�����÷�Ӧ����