��Ŀ����
12��N2O����ҽѧ�ϵ�һ��������������һ�ֳ������������壮��ȡN2O�ķ����кܶ࣬�����NH4NO3�TN2O��+2H2O��K2SO3+2NO�TK2SO4+N2O����2NH3+2O2�TN2O��+3H2O����2NaNO3+��NH4��2SO4�T2N2O��+Na2SO4+4H2O�ȣ�����˵����ȷ���ǣ�������| A�� | ��Ӧ��������識������������ǻ�ԭ����H2O���������� | |
| B�� | ��Ӧ����K2SO3�ǻ�ԭ����N2O���������� | |
| C�� | ��Ӧ����ÿ����1 mol N2O��ת��8 mol���� | |
| D�� | ��Ӧ����NaNO3�ǻ�ԭ����N2O���������������ǻ�ԭ���� |
���� A����Ӧ��NH4NO3�TN2O��+2H2O�У�NH4NO3��NԪ�صĻ��ϼۼ������ֽ��ͣ�����N2O���������������ǻ�ԭ���
B����Ӧ��K2SO3+2NO�TK2SO4+N2O���У�NԪ�صĻ��ϼ۽��ͣ�SԪ�صĻ��ϼ����ߣ�
C����Ӧ��2NH3+2O2�TN2O��+3H2O�У�NԪ�صĻ��ϼ���-3���ߵ�+1��
D����Ӧ��2NaNO3+��NH4��2SO4�T2N2O��+Na2SO4+4H2O�У�NaNO3��NԪ�صĻ��ϼ۽��ͣ���NH4��2SO4��NԪ�صĻ��ϼ����ߣ�
��� �⣺A����Ӧ��NH4NO3�TN2O��+2H2O�У�NH4NO3��NԪ�صĻ��ϼۼ������ֽ��ͣ�������識������������ǻ�ԭ��������N2O���������������ǻ�ԭ�������H2O�Ȳ������������ֲ��ǻ�ԭ�����A����
B����Ӧ��K2SO3+2NO�TK2SO4+N2O���У�NԪ�صĻ��ϼ۽��ͣ�SԪ�صĻ��ϼ����ߣ�����K2SO3�ǻ�ԭ��������K2SO4�����������B����
C����Ӧ��2NH3+2O2�TN2O��+3H2O�У�NԪ�صĻ��ϼ���-3���ߵ�+1����ÿ����1 mol N2O��ת��1��2����3+1��=8 mol���ӣ���C��ȷ��
D����Ӧ��2NaNO3+��NH4��2SO4�T2N2O��+Na2SO4+4H2O�У�NaNO3��NԪ�صĻ��ϼ۽��ͣ���NaNO3������������NH4��2SO4��NԪ�صĻ��ϼ����ߣ�����N2O���������������ǻ�ԭ�����D����
��ѡC��
���� ���⿼��������ԭ��Ӧ��Ϊ��Ƶ���㣬���շ�Ӧ��Ԫ�ػ��ϼ۱仯Ϊ���Ĺؼ���ע���Ԫ�ػ��ϼ۽Ƕȷ���������������ԭ��Ӧ��������Ŀ��飬��Ŀ�ѶȲ���

| A�� | ��������Һ | B�� | Fe��OH��3���� | ||
| C�� | ����ֲ���ͺ�ˮ��� | D�� | ��ˮ |
| A�� | X�ж���ͬ�������壬��Y������ͬ�������� | |
| B�� | X��M���������γ��ɼ��Լ����ɵ�XCl4��MCl4���� | |
| C�� | W���������Ӧ��ˮ�������Z������������Ӧ��ˮ���ﷴӦ | |
| D�� | ���Ӱ뾶��С��r��Y2-����r��W3+�� |
| A�� | Һ������ʱҪ���մ������ȣ������������ | |
| B�� | ѹ��ȼú���ϸ�س���������ҵ��������������Ч��ʩ | |
| C�� | �ֻ����������̼��ά��Ĥ��һ�����͵����ǽ������� | |
| D�� | ���������������ͨ������Ҳ����������θ�����IJ�֢ |
| A�� | ����������ʵ���Ũ�ȵ�NaHCO3��Ba��OH��2����Һ��ϣ�HCO3-+Ba2++OH-�TBaCO3��+H2O | |
| B�� | ��NH4Al��SO4��2��Һ�е���Ba��OH��2ǡ��ʹSO42-��ȫ������2Ba2++4OH-+Al3++2SO42-�T2BaBO4��+AlO2-+2H2O | |
| C�� | ��Mg��HCO3��2��Һ�м��������NaOH��Һ��Mg2++2HCO-3+4OH-�TMg��OH��2��+2CO32-+2H2O | |
| D�� | ��Fe��NO3��2��Һ�м���ϡ���3Fe2++4H++NO3-�T3Fe3++NO��+2H2O |
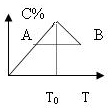 ���ڷ�Ӧ2A+B?2C�ķ�Ӧ������C�İٷֺ������¶ȵı仯��ͼ��ʾ����
���ڷ�Ӧ2A+B?2C�ķ�Ӧ������C�İٷֺ������¶ȵı仯��ͼ��ʾ���� ���в���Ԫ�صĴ��ڣ����ʺ�ԭ�ӽṹ����������
���в���Ԫ�صĴ��ڣ����ʺ�ԭ�ӽṹ����������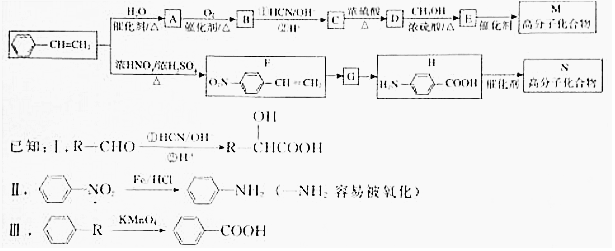
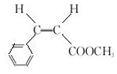 ��
�� 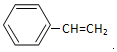 ��F�ķ�Ӧ����Ϊȡ�� ��Ӧ��
��F�ķ�Ӧ����Ϊȡ�� ��Ӧ��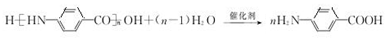 ��
��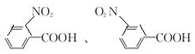 ��
��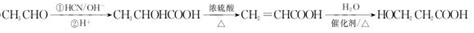 ��
��