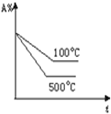
��Ŀ����
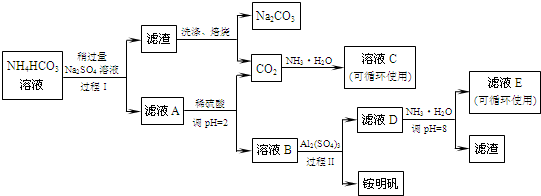
20����A��B��C��D��E��F���ֶ���������Ԫ�أ����ǵ�ԭ������������������AԪ��ԭ�Ӻ�����ӽ���һ��ԭ�ӹ����Ҳ����������ḻ��Ԫ�أ�BԪ��ԭ�Ӻ���P��������s��������1��CԪ��ԭ�ӵ����������Ų��ɱ�ʾΪasaap2a��DΪ����Ԫ����M��P�ܼ�����һ��δ�ɶԵĵ��ӣ�EԪ�ص�ԭ�Ӻ�������p����������ȫ����FԪ��ԭ�ӵļ۵����Ų�ʽΪ3s23p5����1��д��A��C��FԪ�ص�Ԫ�ط��ţ�AH��CO��FCl��
��2��д����̬Dԭ�ӵĵ����Ų�ʽ1s22s22p63s23p1��д����̬Eԭ�ӵĺ�������Ų�ͼ
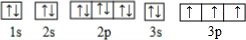 ��
����3��DԪ��ԭ�Ӻ����������4�ֲ�ͬ����չ������13�ֲ�ͬ�˶�״̬�ĵ��ӣ�5�ֲ�ͬ�ܼ��ĵ��ӣ�
���� ��A��B��C��D���ֶ����ڵ�����Ԫ�أ����ǵ�ԭ������������������AԪ��ԭ�Ӻ�����ӽ���һ��ԭ�ӹ����Ҳ����������ḻ��Ԫ�أ���AΪ��Ԫ�أ�BԪ��ԭ�ӵĺ���p�����������s�����������1��Bԭ�Ӻ�������Ų�Ϊ1s22s22p3��ΪNԪ�أ�CԪ��ԭ�ӵ����������Ų��ɱ�ʾΪasaap2a��a=2����CΪOԪ�أ�DΪ����Ԫ����M��P�ܼ�����һ��δ�ɶԵĵ��ӣ���������Ų�Ϊ1s22s22p63s23p1����DΪAlԪ�أ�EԪ�ص�ԭ�Ӻ�������p���ȫ���������ԭ����������Al�����������Ų�Ϊ1s22s22p63s23p3����EΪPԪ�أ�FԪ��ԭ�ӵļ۵����Ų�ʽΪ3s23p5����FΪCl���ݴ˽��
��� �⣺��A��B��C��D���ֶ����ڵ�����Ԫ�أ����ǵ�ԭ������������������AԪ��ԭ�Ӻ�����ӽ���һ��ԭ�ӹ����Ҳ����������ḻ��Ԫ�أ���AΪ��Ԫ�أ�BԪ��ԭ�ӵĺ���p�����������s�����������1��Bԭ�Ӻ�������Ų�Ϊ1s22s22p3��ΪNԪ�أ�CԪ��ԭ�ӵ����������Ų��ɱ�ʾΪasaap2a��a=2����CΪOԪ�أ�DΪ����Ԫ����M��P�ܼ�����һ��δ�ɶԵĵ��ӣ���������Ų�Ϊ1s22s22p63s23p1����DΪAlԪ�أ�EԪ�ص�ԭ�Ӻ�������p���ȫ���������ԭ����������Al�����������Ų�Ϊ1s22s22p63s23p3����EΪPԪ�أ�FԪ��ԭ�ӵļ۵����Ų�ʽΪ3s23p5����FΪCl��
��1��A��C��FԪ�ص�Ԫ�ط��ŷֱ�ΪH��O��Cl���ʴ�Ϊ��H��O��Cl��
��2����̬Dԭ�ӵĵ����Ų�ʽΪ1s22s22p63s23p1��EΪPԪ�أ���̬ԭ�ӵĵ����Ų�ͼΪ��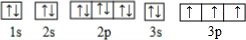 ��
��
�ʴ�Ϊ��1s22s22p63s23p1��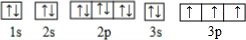
��3��ԭ�ӵĵ����Ų�ʽΪ1s22s22p63s23p1��ԭ�Ӻ����������4�ֲ�ͬ����չ������13�ֲ�ͬ�˶�״̬�ĵ��ӣ�5�ֲ�ͬ�ܼ��ĵ��ӣ�
�ʴ�Ϊ��4��13��5��
���� ���⿼��ṹ����λ�ù�ϵ����������Ų����ɵȣ��ѶȲ������պ�������Ų��������ƶϵĹؼ���

| A�� | C | B�� | HNO3 | C�� | CO2 | D�� | SO2 |
| A�� | O2��O3 | B�� | 35Cl��37Cl | C�� | CH3CHO��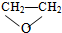 | D�� | CH4��C2H6 |
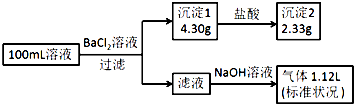 ij100mL��Һ���ܺ���Na+��NH4+��Fe3+��CO32-��SO42-��Cl-�е������֣�ȡ����Һ��������ʵ�飬ʵ�������ͼ���������Լ�������������ȫ���ݳ�������˵������ȷ���ǣ�������
ij100mL��Һ���ܺ���Na+��NH4+��Fe3+��CO32-��SO42-��Cl-�е������֣�ȡ����Һ��������ʵ�飬ʵ�������ͼ���������Լ�������������ȫ���ݳ�������˵������ȷ���ǣ�������| A�� | ԭ��Һһ������CO32-��SO42-��һ��������Fe3+ | |
| B�� | ԭ��Һһ������Cl-�����ܴ���Na+ | |
| C�� | ԭ��Һ��c��Cl-����0.1mol•L-1 | |
| D�� | ��ԭ��Һ�в�����Na+����c��Cl-����0.1mol•L-1 |
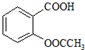 ����������Ӧ����㷺�Ľ��ȡ���ʹ�Ϳ���ҩ������ˮ���������ֽ⣬�ֽ��¶�Ϊ128��135�森ijѧϰС����ʵ������ˮ���ᣨ���ǻ������ᣩ�������[��CH3CO��2O]Ϊ��Ҫԭ�Ϻϳɰ�˾ƥ�֣��Ʊ���������������ͼ1��
����������Ӧ����㷺�Ľ��ȡ���ʹ�Ϳ���ҩ������ˮ���������ֽ⣬�ֽ��¶�Ϊ128��135�森ijѧϰС����ʵ������ˮ���ᣨ���ǻ������ᣩ�������[��CH3CO��2O]Ϊ��Ҫԭ�Ϻϳɰ�˾ƥ�֣��Ʊ���������������ͼ1��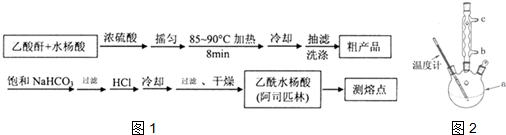
��Ҫ�Լ��Ͳ�Ʒ����������
| ���� | ��Է������� | �۵��е㣨�棩 | ˮ |
| ˮ���� | 138 | 158���۵㣩 | �� |
| ���� | 102 | 139.4���е㣩 | ��Ӧ |
| ����ˮ���� | 180 | 135���۵㣩 | �� |
��1���Ʊ���˾ƥ��ʱ��Ҫʹ�ø����������ԭ���Ƿ�ֹ������ˮ�⣻
��2��д���Ʊ���˾ƥ�ֵĻ�ѧ����ʽ
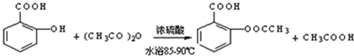 ��
����3���ٺϳɰ�˹ƥ��ʱ������ʵļ��ȷ�����ˮԡ���ȣ�
�ڹ������ôֲ�ƷҪ��������ˮϴ�ӣ���ϴ�ӵľ�������ǹر�ˮ��ͷ����©���мӱ�ˮ����û�����ʹϴ�Ӽ�����ͨ��������ظ���������2-3�Σ�
��4���ᴿ�ֲ����м��뱥��NaHCO3��Һ��û��CO2����Ϊֹ���ٹ��ˣ���ӱ���NaHCO3��Һ��Ŀ����ʹ����ˮ����ת��Ϊ������ˮ������ˮ�����ƣ�������ۺ�����룻
��һ�ָĽ����ᴿ��������Ϊ�ؽᾧ�ᴿ�����������£�
[�ֲ�Ʒ]$��_{��ʯ}^{��������}$ $��_{����}^{����}$ $\stackrel{���ȹ���}{��}$ $��_{����}^{��ȴ}$ $��_{����}^{ϴ��}$[����ˮ����]
��5���Ľ����ᴿ�����м��Ȼ�����װ����2ͼ��ʾ��ʹ���¶ȼƵ�Ŀ���ǿ��Ʒ�Ӧ�¶ȣ���ֹ�¶ȹ��ߣ���������ˮ�������ȷֽ⣻����ˮ������������b���b����c����
��6�������Ʒ���Ƿ���ˮ������Լ���FeCl3��Һ��
��7����ѧϰС����ʵ����ԭ��������2.0gˮ���ᡢ5.0mL����������=1.08g/cm3�������ճƵò�Ʒm=2.2g������������ˮ����IJ���Ϊ84.3%��
| A�� | H+��Na+��HCO3-��Cl- | B�� | Fe3+��SCN-��Cl-��NO3- | ||
| C�� | Mg2+��Cu2+��SO42-��NO3- | D�� | Fe2+��SO42-��H+��NO3- |