��Ŀ����
20���л���M��C25H44O8����һ���������Ƹ�Ѫѹ����ҩ���ҵ���õ��ۡ���AΪ����ԭ�Ϻϳ�M��·����ͼ��ʾ��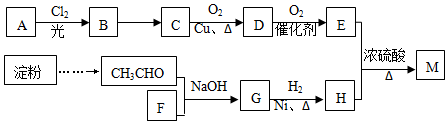
��֪��
I����A������ͼ�е�����ʺɱ�Ϊ72��B�����к˴Ź���������2�����������Ϊ9��2��35%-40%��F��ˮ��Һ����Ϊ�������֣�
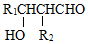
II��R1CHO+R2CH2CHO$\stackrel{NaOH}{��}$
��1��A������Ϊ2��2-�������飨ϵͳ����������B�Ľṹ��ʽΪC��CH3��3CH2Cl��F�ķ���ʽΪCH2O��
��2��B��C�ķ�Ӧ��������������ˮ��Һ�����ȣ�G�Ĺ�����������ȩ�����ǻ���
��3��D��������Һ��Ӧ�ķ���ʽ
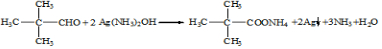 ��
����4��E��H��Ӧ����M�ķ���ʽ
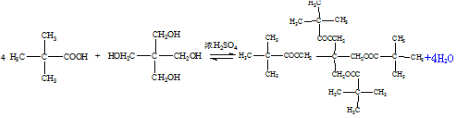 ��
����5��E�ж���ͬ���칹�壬�����ܷ���������Ӧ�����������ͬ���칹�干��4�֣����к˴Ź���������2��������ʽṹ��ʽΪ
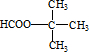 ��
��
���� ��A������ͼ�е�����ʺɱ�Ϊ72������Է�������Ϊ72��������Cԭ�������ĿΪ$\frac{72}{12}$=5��12����A�ķ���ʽΪC5H12��B�����к˴Ź���������2�����������Ϊ9��2����BӦΪC��CH3��3CH2Cl��AӦΪC��CH3��4��35%-40%��F��ˮ��Һ����Ϊ�������֣���FΪHCHO����ת����ϵ��֪CΪC��CH3��3CH2OH��DΪC��CH3��3CHO��EΪC��CH3��3COOH����M�ķ���ʽ��֪HӦ����4���ǻ����Һ���5��Cԭ�ӣ���HΪC��CH2OH��4��GΪC��CH2OH��3CHO��MΪC��CH2OOCC��CH3��3��4���Դ˽����⣮
��� �⣺��1�������Ϸ�����֪AΪC��CH3��4��Ϊ2��2-�������飬BΪC��CH3��3CH2Cl��FΪHCHO������ʽΪCH2O��
�ʴ�Ϊ��2��2-�������飻C��CH3��3CH2Cl��CH2O��
��2��B��C����ˮ�ⷴӦ����Ӧ����Ϊ��������ˮ��Һ�����ȣ�GΪC��CH2OH��3CHO������ȩ�����ǻ���
�ʴ�Ϊ����������ˮ��Һ�����ȣ�ȩ�����ǻ���
��3��DΪC��CH3��3CHO����������Һ��Ӧ�ķ���ʽΪ ��
��
�ʴ�Ϊ�� ��
��
��4��E��H��Ӧ����M�ķ���ʽΪ ��
��
�ʴ�Ϊ�� ��
��
��5��EΪC��CH3��3COOH���ܷ���������Ӧ�����������ͬ���칹�壬ӦΪ�����붡���γɵ�����ͬ���칹����Ŀȡ���ڶ�����������4�֣�����4�֣����к˴Ź���������2��������ʽṹ��ʽΪ ��
��
�ʴ�Ϊ��4�� ��
��
���� ���⿼���л����ƶϣ�Ϊ�߿��������ͣ����ؿ���ѧ���ķ����������漰±����������ȩ�������������ת��������ȷ��A�ķ���ʽ�ǹؼ���ע���������չ����ŵ�������ת�����Ѷ��еȣ�

| A�� | Ӳ�ȱȴ�ͭС | B�� | �ܹ����� | C�� | �ܹ����� | D�� | �۵�ȴ�ͭ�� |
| ʵ�� | �Լ��� | �Լ��� | �Լ��� | ���е����� |
| A | Ũ���� | Na2SO3 | ��ɫʯ����Һ | ��Һ�ȱ�����ɫ |
| B | Ũ���� | KMnO4 | ����-Kl��Һ | ��Һ���� |
| C | ϡ���� | CaCO3 | BaCl2��Һ | �а�ɫ�������� |
| D | Ũ��ˮ | CaO | AlCl3��Һ | �����ɰ�ɫ����Ȼ������ܽ� |
| A�� | A | B�� | B | C�� | C | D�� | D |
ʵ�����ϱ��д�С�����ձ�����ĭ���ϡ���ĭ���ϰ塢��ͷ�ιܡ����β����������0.5mol•L-1���ᡢ0.55mol•L-1NaOH��Һ��ʵ����ȱ�ٵIJ�����Ʒ����Ͳ���¶ȼƣ�
��2����֪����Ӧ����Һ�ı�����cΪ4.18J•��-1•g-1��0.5mol•L-1�����0.55mol•L-1NaOH��Һ���ܶȾ�Ϊ1g•c
m-3��ʵ��ʱ��¼��ʵ�����������
| ʵ�� ���� | ��Ӧ�P���� | �� Һ �� �� | ||
| t1 | t2 | |||
| 1 | 50mL0.55mol•L-1NaOH��Һ | 50mL.0.5mol•L-1HCl��Һ | 20�� | 23.3�� |
| 2 | 50mL0.55mol•L-1NaOH��Һ | 50mL.0.5mol•L-1HCl��Һ | 20�� | 23.5�� |
����֪��CH3COOH��aq��?CH3COO-��aq��+H+��aq����H2��0��
CH3COOH��aq��+OH-��aq��=CH3COO-��aq��+H20��l����H3 ���H3����H1�������������������=����
| A�� | 0.1mol/LNa2SO3��Һ�У�c��Na+���T2c��SO32-��+c��HSO3-��+c��H2SO4��=0.2mol/L | |
| B�� | ������pH=a��ϡH2SO4��pH=b�İ�ˮ�������Ϻ�ǡ����ȫ��Ӧ����a+b��14 | |
| C�� | 0.2mol/LCH3COOH��Һ��0.1mol/LNaOH��Һ�������ϣ�c��H+��-c��OH-���Tc��CH3COO-��-c��CH3COOH�� | |
| D�� | pH��ͬ��������Һ��NH4Cl �ڣ�NH4��2SO4 ��NH4HSO4��c��NH4+����С��ϵΪ���٣��ڣ��� |
��1��ʵ��һ���������Ʊ���Һ�����ƣ�
��Ҫ����0.1000mol/LNaOH����Һ250mL����Ҫ�õ��IJ��������в��������ձ�����ͷ�ιܺ�250��������ƿ���������������û�н��ձ��ȵ�ϴ��Һһ��ת������ƿ����������������ȷ������£����ñ���Һ�����ζ����У�2���е�δ֪Ũ�ȵ�������Һ������ʹ�ඨ�Ľ��ƫ�ߣ���ߡ������͡�������Ӱ�족��
��2��ʵ���������һƿ������Һ�������²ⶨ��������ĵ���ƽ�ⳣ�������ʵ�鷽�����������������Ͷ�Ӧ�IJⶨ������д�ڱ�һ�У�
��һ��
| ���������� | �ⶨ���� |
| ��������Һ�����ʵ���Ũ�� | ��ȡ25.00mL������Һ����ƿ�У��μ�ָʾ������0.1000mol/LNaOH����Һװ���ʽ�ζ��ܣ��ζ����յ㣬��¼���ݣ��ظ��ζ�2�Σ� |
| ��H+�����ʵ���Ũ�� | ȡ����������Һ���ձ��У���pH�ƻ���pH��ֽ�ⶨ��ҺpH�� |
��3��ʵ������̽�����ǿ��������þ����Ӧ���ʵ�Ӱ�죮
�����ʵ�鷽���������������c=2.0g��
������
| ��� | ������� | ���Ũ�ȣ�mol/L�� | ������/mL | þ������/g |
| l | ���� | 0.5 | 17.0 | 2.0 |
| 2 | ���� | 0.5 | 17.0 | c |
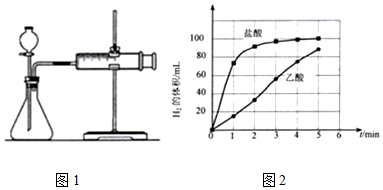
a��ͼ1��װ���У�������ҩƷ֮ǰ������װ�õ������ԣ�
b����Ӧ��ʼ��ÿ��1min��¼һ������H2�������
c��������¼�����ݻ��Ƴ�����ͼ��ͼ2����
��д��þ��������Һ��Ӧ�����ӷ���ʽ��Mg+2CH3COOH=Mg2++2CH3COO-+H2����
������0��5min������þ����Ӧ�ķ�Ӧ���ʱ仯���ɣ�������þ����Ӧ��������ʱ��仯�����ԣ�������þ����Ӧ��ʼ�η�Ӧ���ʺܿ죬һ��ʱ���Ӧ�������Լ�С��
| A�� | ���� | B�� | ���� | C�� | ���� | D�� | ���� |
| A�� | ��1molCl2ͨ��ˮ�У�HClO��Cl-��ClO-������֮��Ϊ2NA | |
| B�� | 12gNaHSO4�����к���������Ϊ0.1NA | |
| C�� | ��״���£�22.4LBr2������������ΪNA | |
| D�� | 7.8gNa2O2��������ˮ��Ӧת�Ƶ�����Ϊ0.2NA |
| A�� | Al��OH��3��Al2O3 | B�� | Al2O3��Al��OH��3 | C�� | Al��AlCl3 | D�� | Al��NaAlO2 |