��Ŀ����
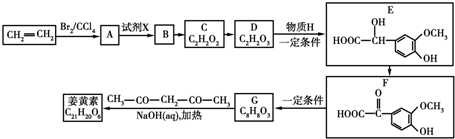
8��ij�л������ʽΪC13H18O2����һ��ʳƷ���ϣ���ϳ�·����ͼ��ʾ��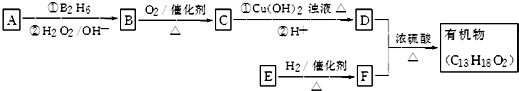
��֪��R-CH=CH2$\underset{\stackrel{��{B}_{2}{H}_{6}}{��}}{��{H}_{2}{O}_{2}/O{H}^{-}}$R-CH2CH2OH
ͨ���������A����Է�������Ϊ56�����ĺ˴Ź���������ʾ��������ҷ����֮��Ϊ1��3��D�����к���֧����F�����к��б���������E�ɷ���������Ӧ���ڴ���������1mol E��2mol H2���Է�����Ӧ����F����ش�
��1��A�����������ŵ�����Ϊ̼̼˫����F�Ľṹ��ʽΪ
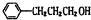 ��
����2��C�����Ƽ���Cu��OH��2����Һ��Ӧ�����ӷ���ʽΪ��CH3��2CHCHO+2Cu��OH��2+NaOH$\stackrel{��}{��}$��CH3��2CHCOONa+Cu2O��+3H2O��
��3��D��F��Ӧ�Ļ�ѧ����ʽΪ
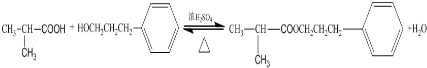 ���䷴Ӧ������ȡ����Ӧ��
���䷴Ӧ������ȡ����Ӧ����4��д�����з�������������F��ͬ���칹��2����F���⣩����FeCl3��Һ����ɫ���ں˴Ź������������ֲ�ͬ��ѧ�������⣬�ҷ������Ϊ1��2��3��6��
��5��������������
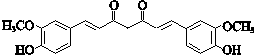
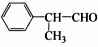
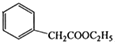 ����һ����Ҫ��ҽҩ�м��壮д���Ա���ͪ��
����һ����Ҫ��ҽҩ�м��壮д���Ա���ͪ��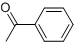 �����Ҵ�Ϊ��Ҫԭ���Ʊ������������ĺϳ�·������ͼ�����Լ���ѡ����
�����Ҵ�Ϊ��Ҫԭ���Ʊ������������ĺϳ�·������ͼ�����Լ���ѡ�����ϳ�·������ͼ��ο�������ʽ��A$��_{��Ӧ����}^{��Ӧ��}$B$��_{��Ӧ����}^{��Ӧ��}$C����H��
���� �л���A�������࣬����Է�������Ϊ56����$\frac{56}{12}$=4��8����֪A�ķ���ʽΪC4H8��A������Ϣ�еķ�Ӧ����B��B������������������Ӧ��A�ĺ˴Ź���������2����ҷ����֮��Ϊ1��3����AΪ��CH3��2C=CH2��B�Ľṹ��ʽΪ��CH3��2CHCH2OH��C�Ľṹ��ʽΪ��CH3��2CHCHO��D�Ľṹ��ʽΪ��CH3��2CHCOOH��D��F��Ӧ�����л���C13H18O2��Ӧ�Ƿ���������Ӧ����FΪ����D�к���4��̼ԭ�ӣ�����F�к���9��̼ԭ�ӣ�F�ķ���ʽΪC9H12O��E���Է���������Ӧ������-CHO���ڴ�������������1mol E��2mol H2��Ӧ��������F��F�����к��б�������������֪EΪ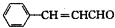 ��FΪ
��FΪ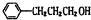 ����GΪ
����GΪ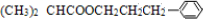 ��
��
��� �⣺�л���A�������࣬����Է�������Ϊ56����$\frac{56}{12}$=4��8����֪A�ķ���ʽΪC4H8��A������Ϣ�еķ�Ӧ����B��B������������������Ӧ��A�ĺ˴Ź���������2����ҷ����֮��Ϊ1��3����AΪ��CH3��2C=CH2��B�Ľṹ��ʽΪ��CH3��2CHCH2OH��C�Ľṹ��ʽΪ��CH3��2CHCHO��D�Ľṹ��ʽΪ��CH3��2CHCOOH��D��F��Ӧ�����л���C13H18O2��Ӧ�Ƿ���������Ӧ����FΪ����D�к���4��̼ԭ�ӣ�����F�к���9��̼ԭ�ӣ�F�ķ���ʽΪC9H12O��E���Է���������Ӧ������-CHO���ڴ�������������1mol E��2mol H2��Ӧ��������F��F�����к��б�������������֪EΪ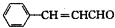 ��FΪ
��FΪ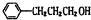 ����GΪ
����GΪ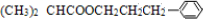 ��
��
��1��AΪ��CH3��2C=CH2�����еĹ�����Ϊ̼̼˫����F�Ľṹ��ʽΪ��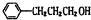 ��
��
�ʴ�Ϊ��̼̼˫����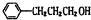 ��
��
��2��CΪ��CH3��2CHCHO��������Cu��OH��2����Һ��Ӧ�Ļ�ѧ����ʽΪ����CH3��2CHCHO+2Cu��OH��2+NaOH$\stackrel{��}{��}$��CH3��2CHCOONa+Cu2O��+3H2O��
�ʴ�Ϊ����CH3��2CHCHO+2Cu��OH��2+NaOH$\stackrel{��}{��}$��CH3��2CHCOONa+Cu2O��+3H2O��
��3��D+F��G�Ļ�ѧ����ʽ�� ������ȡ����Ӧ��
������ȡ����Ӧ��
�ʴ�Ϊ�� ��ȡ����Ӧ��
��ȡ����Ӧ��
��4��F��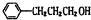 ����ͬ���칹�����㣺����FeCl3��Һ����ɫ��˵�����з��ǻ����ں˴Ź������������ֲ�ͬ��ѧ�������⣬�ҷ������Ϊ1��2��3��6������������ͬ���칹��Ϊ��
����ͬ���칹�����㣺����FeCl3��Һ����ɫ��˵�����з��ǻ����ں˴Ź������������ֲ�ͬ��ѧ�������⣬�ҷ������Ϊ1��2��3��6������������ͬ���칹��Ϊ��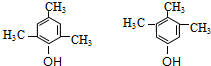 ������2�֣�
������2�֣�
�ʴ�Ϊ��2��
��5�� �����ӳɷ�Ӧ����
�����ӳɷ�Ӧ����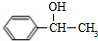 ���ٷ�����ȥ��Ӧ����
���ٷ�����ȥ��Ӧ����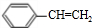 ������B2H6��H2O2/OH-�õ�
������B2H6��H2O2/OH-�õ�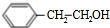 ���������ɱ���ȩ����һ���������ɱ����ᣬ�����Ҵ�����������Ӧ���ɱ������������ϳ�·������ͼΪ����
���������ɱ���ȩ����һ���������ɱ����ᣬ�����Ҵ�����������Ӧ���ɱ������������ϳ�·������ͼΪ����
�ʴ�Ϊ�� $��_{����}^{����}$
$��_{����}^{����}$ $��_{��}^{Ũ����}$
$��_{��}^{Ũ����}$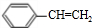 $��_{H_{2}O_{2}/OH-}^{B_{2}H_{6}}$
$��_{H_{2}O_{2}/OH-}^{B_{2}H_{6}}$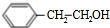 $\stackrel{O_{2}/����}{��}$
$\stackrel{O_{2}/����}{��}$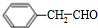 $\stackrel{O_{2}/����}{��}$
$\stackrel{O_{2}/����}{��}$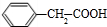 $��_{Ũ����/��}^{�Ҵ�}$
$��_{Ũ����/��}^{�Ҵ�}$ ��
��
�ʴ�Ϊ�� $��_{����}^{����}$
$��_{����}^{����}$ $��_{��}^{Ũ����}$
$��_{��}^{Ũ����}$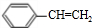 $��_{H_{2}O_{2}/OH-}^{B_{2}H_{6}}$
$��_{H_{2}O_{2}/OH-}^{B_{2}H_{6}}$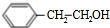 $\stackrel{O_{2}/����}{��}$
$\stackrel{O_{2}/����}{��}$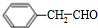 $\stackrel{O_{2}/����}{��}$
$\stackrel{O_{2}/����}{��}$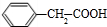 $��_{Ũ����/��}^{�Ҵ�}$
$��_{Ũ����/��}^{�Ҵ�}$ ��
��
���� ���⿼���л����ƶ���ϳɣ���Ҫѧ���Ը������Ϣ�������ã��dz������ͣ����ؿ���ѧ���ķ�������������ע�������Է���������ת����ϵȷ��A�Ľṹ���Ѷ��еȣ�

| A�� | ���� | B�� | ���� | C�� | �����ᾧ | D�� | ��Һ |
| A�� | C2H6��C3H8û��ͬ���칹�壬CH2O2��C2H4O2����ͬϵ�� | |
| B�� | һ�������£���ȫȼ��14g������������Ϊa����ϩ����ȩ������壬������ˮ������Ϊ18��1-a��g | |
| C�� | һ�������£��������������ۡ������ʡ���ϩ������ˮ����ˮ�ⷴӦ | |
| D�� | ȫ�������ϣ�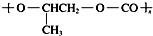 �����ɵ��廷�����飨 �����ɵ��廷�����飨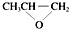 ����CO2�����Ƶ� ����CO2�����Ƶ� |
��1��Ϊ̽�����������I2�������ʵ�Ӱ�죬������������ʵ�飬��д���еĿհ״���
| ��� | ʵ��Ŀ�� | ��Ӧ�� | ��Ӧǰ ��Һ��pH | �¶� |
| 1 | ������ | O3+NaI+H2SO4 | 5.2 | 25�� |
| 2 | ��̽��FeCl2�Է�Ӧ���ʵ�Ӱ�� | O3+NaI+H2SO4+FeCl2 | 5.2 | 25�� |
| 3 | ̽���¶ȶԷ�Ӧ���ʵ�Ӱ�� | O3+NaI+H2SO4 | ��5.2 | 5�� |
��֪����H2S2O3��һ�������2Na2S2O3+I2�TNa2S4O6+2NaI
�ٸõζ�ʵ���г��ձ�����ƿ�⣬����Ҫ�IJ��������Ǽ�ʽ�ζ��ܣ�
�ڸ�ʵ���п�ѡ�õ��ۣ����������ƣ���ָʾ����
�۷�Ӧ����Һ�е�ĺ���Ϊ6.35cVg•L-1��
�ܼ�ͬѧ��Ϊ�ڵζ�ǰӦ����Һ�����ų���Һ���ܽ��O3��O2����Ȼ��ʹ�ζ����ƫ�ߣ���͡��ߡ�����


 ��
�� ��
�� ��
��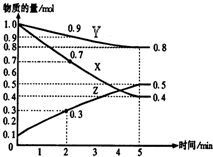 ij�¶�ʱ����2L������X��Y��Z����������ʱ��ı仯��ϵ������ͼ��ʾ��
ij�¶�ʱ����2L������X��Y��Z����������ʱ��ı仯��ϵ������ͼ��ʾ��
 ����ҵ�ϳ�·�����£�
����ҵ�ϳ�·�����£�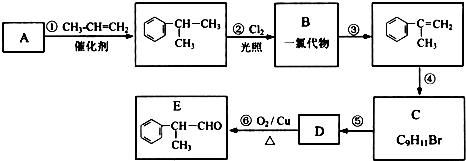
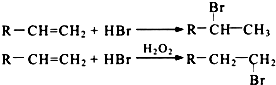 ��R-����������
��R-���������� ��
��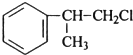 ����дһ�������������Ϊ��ԭ�ӣ�
����дһ�������������Ϊ��ԭ�ӣ�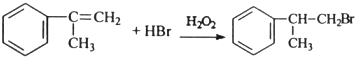 �������з�Ӧ��������
�������з�Ӧ�������� ��
��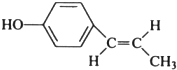 ��ֻд��ʽ�ṹ����
��ֻд��ʽ�ṹ����