��Ŀ����
��9�֣�O2��O3����Ԫ�ص����ֵ��ʣ����������ʽ������и��⣺
��1����������O2��O3�������Ӹ�����Ϊ ��ԭ�Ӹ�����Ϊ �����ӵ����ʵ���֮��Ϊ ��
��2�����µ�ѹ�£�������������ͳ����������Ӹ�����Ϊ ��ԭ�Ӹ�����Ϊ ������֮��Ϊ ��
��3����NAΪ�����ӵ���������ֵ�����a g�����к��еķ�����Ϊb����c g�����ڱ�״���µ����Լ�� ���ú�NA��ʽ�ӱ�ʾ����
��4����8 g SO3���릶g ˮ�У����γɵ���Һ��SO42����ˮ���Ӹ���֮��Ϊ1:5������ ��
��5�����³�ѹ�£�14 g��N2��CO��ɵĻ�������к��е�ԭ����ĿΪ ��
��1��3:2 1:1 3:2 ��2��1:1 2:3 2:3��3�� L
L
��4��10.8 ��5��NA��ԼΪ6.02x1023
��������
�����������1����������Ϊm����������O2��O3�������Ӹ�����=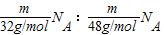 =3:2��һ�����������к���2����ԭ�ӣ�һ�����������к���3����ԭ�ӣ���������ԭ�Ӹ���=��3��2��:��2��3��=1��1�������ʵ���֮��=
=3:2��һ�����������к���2����ԭ�ӣ�һ�����������к���3����ԭ�ӣ���������ԭ�Ӹ���=��3��2��:��2��3��=1��1�������ʵ���֮��=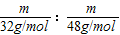 =3:2���ʴ�Ϊ��3:2��1:1��3:2����2����ͬѹ�£�����Ħ�������ͬ������N=
=3:2���ʴ�Ϊ��3:2��1:1��3:2����2����ͬѹ�£�����Ħ�������ͬ������N=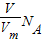 ֪���������O2��O3�������Ӹ�������1:1��һ�����������к���2����ԭ�ӣ�һ�����������к���3����ԭ�ӣ�����ԭ�Ӹ���֮����2:3������m=
֪���������O2��O3�������Ӹ�������1:1��һ�����������к���2����ԭ�ӣ�һ�����������к���3����ԭ�ӣ�����ԭ�Ӹ���֮����2:3������m=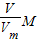 ֪��������֮�ȵ�����Ħ������֮��=32g/mol��48g/mol=2:3���ʴ�Ϊ��1:1��2:3��2:3����3��������Ħ������ΪM=
֪��������֮�ȵ�����Ħ������֮��=32g/mol��48g/mol=2:3���ʴ�Ϊ��1:1��2:3��2:3����3��������Ħ������ΪM=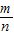 =
=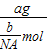 =
=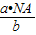 g/mol����c g O2�ڱ�״�������Ϊ��V=
g/mol����c g O2�ڱ�״�������Ϊ��V=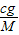 ?Vm=
?Vm=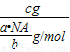 ��22.4 L/mol=
��22.4 L/mol=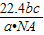 L���ʴ�Ϊ��
L���ʴ�Ϊ��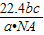 ����4��8g����������0.1mol����ˮ��Ӧ�ķ���ʽ��SO3��H2O=H2SO4��������������0.1mol������0.1molˮ�����Է�Ӧ����Һ��ˮ�����ʵ�����0.5mol��������ˮ��0.6mol��������10.8g����5������N2��CO��ɵĻ�����������Ժ��ֱ�����ϣ�Ħ��������Ϊ28g/mol,���Գ��³�ѹ��,14g��N2��CO��ɵĻ����������ʵ���Ϊ0.5mol�����������߾�Ϊ˫ԭ�ӣ��ʺ��е�ԭ����Ŀ��NA��
����4��8g����������0.1mol����ˮ��Ӧ�ķ���ʽ��SO3��H2O=H2SO4��������������0.1mol������0.1molˮ�����Է�Ӧ����Һ��ˮ�����ʵ�����0.5mol��������ˮ��0.6mol��������10.8g����5������N2��CO��ɵĻ�����������Ժ��ֱ�����ϣ�Ħ��������Ϊ28g/mol,���Գ��³�ѹ��,14g��N2��CO��ɵĻ����������ʵ���Ϊ0.5mol�����������߾�Ϊ˫ԭ�ӣ��ʺ��е�ԭ����Ŀ��NA��
���㣺�������ʵ����ļ��㡣

 ����νӽ̲���ĩ���Ԥϰ�人������ϵ�д�
����νӽ̲���ĩ���Ԥϰ�人������ϵ�д� ������ҵ��ٳɳ����½������������ϵ�д�
������ҵ��ٳɳ����½������������ϵ�д������±��е��ĸ��Ȼ�ѧ����ʽ,�ж������ͱ����ȼ���ȷֱ���
���϶�һ�š�������ȼ�� | Һ��(H2) | ��2H2(g)+O2(g) ��2H2 (l)+O2(l) |
�������ˡ����ơ����ȼ�� | ���� (C3H8) | ��C3H8 (l)+5O2(g) ��C3H8(g)+5O2(g) |
A��571.6 kJ��mol-1,2 221.5 kJ��mol-1
B��241.3 kJ��mol-1,2 013.8 kJ��mol-1
C��285.8 kJ��mol-1,2 013.8 kJ��mol-1
D��285.8 kJ��mol-1,2 221.5 kJ��mol-1
ֻ�������м��к����ж�Ӧ�������������һ�������ʵ����Ĺ�ʽ����
| �� | �� | �� | �� | �� |
�� | �������� | ���������Ħ����� | ������� | ��Һ���������� | �DZ�������ʵ����� |
�� | �����ӵ����� | ������������ | �����ܶ� | ��Һ����� | ���ʵ�Ħ������ |
A���ڢۢ� B���ۢܢ� C���ۢ� D���٢�
 2H2O(l) ��H=-571.6 kJ��mol-1
2H2O(l) ��H=-571.6 kJ��mol-1