��Ŀ����
�������֣���1��30%��H2SO4��50%��H2SO4��������Ϻ�������Һ����������________(����ڡ�����С�ڡ����ڡ�)40%������������ϣ���Ϻ���Һ����������________(����ڡ�����С�ڡ����ڡ�)40%��
��2����֪98%��ŨH2SO4�����ʵ���Ũ��Ϊ18.4 mol��L��1�����ж�49%��H2SO4�����ʵ���Ũ��________(����ڡ�����С�ڡ����ڡ�)9.2 mol��L��1��
��3����һ���¶Ⱥ�ѹǿ�£�1���X2(g)��3���Y2(g)��������2���Z(g)����Z����Ļ�ѧʽ��________ ��
��4���ڱ�״���£�CO��CO2�Ļ����������Ϊ36 g�����Ϊ22.4 L����CO��ռ�������__________ L��������________ g��
��1������ ���� ��2��С�ڣ�3��XY3 ��4��11.2 14
��������
�����������1����ԭ��������Ϊm��30%��H2SO4����Ϊ����0.4m��50%��H2SO4���ʵ�����Ϊ0.5m����Ϻ���������仯���������䣬����w��H2SO4��=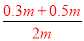 ��100%=40%������������ϣ�������ֱ�ΪVml��30%��H2SO4��Һ�ܶ�Ϊ��1��50%��H2SO4��Һ�ܶ�Ϊ��2�����Ϻ���Һ����������Ϊw��H2SO4��=
��100%=40%������������ϣ�������ֱ�ΪVml��30%��H2SO4��Һ�ܶ�Ϊ��1��50%��H2SO4��Һ�ܶ�Ϊ��2�����Ϻ���Һ����������Ϊw��H2SO4��=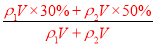 ��100%���������Ũ��Խ���ܶ�Խ�����1����2����w��H2SO4����40%����2��������c=
��100%���������Ũ��Խ���ܶ�Խ�����1����2����w��H2SO4����40%����2��������c=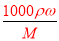 ���㣬��98%��ŨH2SO4���ܶ�Ϊx��49%��H2SO4��Һ���ܶ�Ϊy�����ʵ���Ũ��Ϊc����
���㣬��98%��ŨH2SO4���ܶ�Ϊx��49%��H2SO4��Һ���ܶ�Ϊy�����ʵ���Ũ��Ϊc���� =18.4��
=18.4�� =c�����ԣ�
=c�����ԣ�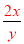 =
=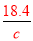 ���������Ũ��Խ���ܶ�Խ����x��y����c��9.2mol/L����3�����ݰ����ӵ����ɿ�֪��1molX2��3molY2��������2mol�����廯������Ը���ԭ���غ㼴�����غ㶨�ɿ�֪���û�����Ļ�ѧʽ��XY3����4����CO�����ʵ���ΪXmol��CO2�����ʵ���ΪYmol����28X+44Y=36��X+Y=1�����X=Y=0.5����CO�����=0.5mol��22.4L/mol=11.2L ,����Ϊ=0.5mol��28g/mol=14g��
���������Ũ��Խ���ܶ�Խ����x��y����c��9.2mol/L����3�����ݰ����ӵ����ɿ�֪��1molX2��3molY2��������2mol�����廯������Ը���ԭ���غ㼴�����غ㶨�ɿ�֪���û�����Ļ�ѧʽ��XY3����4����CO�����ʵ���ΪXmol��CO2�����ʵ���ΪYmol����28X+44Y=36��X+Y=1�����X=Y=0.5����CO�����=0.5mol��22.4L/mol=11.2L ,����Ϊ=0.5mol��28g/mol=14g��
���㣺��ҺŨ�ȵļ���
