��Ŀ����
5������˵����ȷ���ǣ���������ʵ��������Ҫ480mL2.0 mol/L������������Һ��������Һʱ�ȳ����������ƹ���38.4 g��Ȼ���ٰ����ܽ⡢��ȴ��ϴ�ӡ����ݡ�ҡ�ȵIJ�����в���
���ñ�����ζ�NaOH��Һ����Ũ��ʱ����ʽ�ζ���������ˮϴ�Ӻ�û�ñ�������ϴ��ֱ��װ������ζ�������ļ�ҺŨ��ƫ��
���Է���Ӧ��ǡ�������²���ʵ��
�ܸ�����ʴʱ���ܷ�����������Ӧ��2H2O+O2+4e-=4OH-
�ݵ�ⱥ��MgCl2��Һʱ���缫��ӦʽΪ��2H2O+2Cl-=Cl2��+H2��+2OH-
���ں����������п�鱣����Dz��ܸ�ʴ�Dz�����������������������
�߳���ʹ������泥�����������ǿ����ľ�����̬���ʲ��ܻ��ʩ�ã�
| A�� | �٢ڢۢ� | B�� | �ۢܢޢ� | C�� | �٢ۢܢ� | D�� | �ڢۢޢ� |
���� ��ʵ����û��480mL������ƿ����500mL����ƿ��������Ҫ����500mL��Һ���������������Ӧ����500mL��Һ���ܽ��NaOH������
�ڱ�Һ��ϡ�ͣ��������ı�Һ���ƫ�������ƫ�ߣ�
�۷�Ӧ�ܷ��Է�����ȡ���ڡ�H-T��S��ֻ�С�H-T��S��0����Ӧ�����Է����У����Ծ�����Ӧ�ܷ��Է������뷴Ӧ���ر���ʱ䶼�й�ϵ��
�ܸ�������������ʴʱ�������������õ��ӷ�����ԭ��Ӧ��
��þ���������������ӽ������������þ������
�����������п�飬пΪ������
�������ˮ�����Һ�����ԣ���ľ�����̬������ٽ�ˮ�⣮
��� �⣺��ֻ��ѡȡ500mL����ƿ�������ҩƷ����=2mol/L��0.5L��40g/mol=40g���ʢٴ���
��������ñ�Һ��ϴ�ζ��ܻᵼ������Ũ�Ƚ��ͣ���ʹ���������ƫ��������ҺŨ��ƫ�ߣ��ʢڴ���
�۷�Ӧ�ܷ��Է�����ȡ���ڡ�H-T��S�Ƿ�С��0�������H��O����S��0�������ȵġ��ؼ�С�ķ�Ӧ�������·dz���ʱ����H-T��S���ܴ���0�������Է����У������Է���Ӧ��ǡ�������²���ʵ�֣��ʢ���ȷ��
�ܸ�������������ʴʱ�������������õ��ӷ�����ԭ��Ӧ���缫��ӦʽΪO2+2H2O+4e-�T4OH-���ʢ���ȷ��
�ݵ�ⱥ��MgCl2��Һʱ��þ���������������ӽ������������þ����ȷ�ĵ缫��ӦʽΪ��Mg2++2H2O+2Cl-=Cl2��+H2��+Mg��OH��2�����ʢܴ���
�����������п�飬пΪ������������Dz��ܸ�ʴ��Ϊ�����������������������ʢ���ȷ��
�������ˮ�����Һ�����ԣ���ľ�����̬������ٽ�ˮ�⣬���Բ�ľ�����̬���ʲ��ܻ��ʩ�ã��ʢ���ȷ��
���ݷ�����֪����ȷ��Ϊ���ۢܢޢߣ�
��ѡB��
���� ���⿼���Ϊ�ۺϣ��漰�����ĸ�ʴ��������ε�ˮ��ԭ�����к͵ζ�������һ��Ũ�ȵ���Һ������֪ʶ����Ŀ�Ѷ��еȣ���ȷ������ʴԭ�����к͵ζ��������ε�ˮ��ԭ��Ϊ���ؼ�������������ѧ�������Ӧ��������

| A�� | 46g�Ҵ��к��е�C-H������ĿΪ6NA | |
| B�� | 0��lmol��ϩ���к��е�˫������ĿΪ0��lNA | |
| C�� | 1mol����-CH3���к��еĵ�������Ϊ9NA | |
| D�� | ��״���£�22.4 L������ȫȼ�����ɵ�CO2���ӵ���ĿԼΪ6NA |
| A�� | pH=1����Һ�У�Fe2+��NO3-��Cl-��Na+ | |
| B�� | ˮ�������c��H+��=10-12mol•L-1����Һ�У�Ca2+��K+��Cl-��HCO3- | |
| C�� | $\frac{c��{H}^{+}��}{c��O{H}^{-}��}$=1012��ˮ��Һ�У�NH4+��Al3+��NO3-��Cl- | |
| D�� | ������Һ�У�Na+��Cl-��Fe3+��SO42- |
��֪25��ʱ
| ����� | Ka1 | Ka2 |
| H2S | 1.3��10-7 | 7.1��10-15 |
| H2SO3 | 1.3��10-2 | 6.3��10-8 |
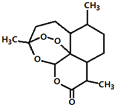 �ҹ���ѧ�������ϻ�2015��ŵ��������ѧ��ҽѧ�������о��õ��������������������ű�����˵��������й�������˵�����������ǣ�������
�ҹ���ѧ�������ϻ�2015��ŵ��������ѧ��ҽѧ�������о��õ��������������������ű�����˵��������й�������˵�����������ǣ�������| A�� | ��ѧʽΪC15H22O5 | |
| B�� | �ܷ���ȡ����Ӧ��������Ӧ | |
| C�� | ��H2SO4�� NaOH��Һ�о������ȶ����� | |
| D�� | �����Ҵ��������ȡҺ����ȡ�������� |
| A�� | N2��NH3 | B�� | SiO2��Na2SiO3 | C�� | Na2O2��Na2CO3 | D�� | SiO2��H2SiO3 |
| A�� | 1��8 | B�� | 5��2 | C�� | 1��5 | D�� | 5��1 |