��Ŀ����
12����ͭ�Ͻ�㷺���ں��չ�ҵ�����и�����л��������Ʊ�ͭ������Ʒ�Ĺ������£�
��ע��Al��OH��3��Cu��OH��2��ʼ�ֽ���¶ȷֱ�Ϊ450���80�棩
��1������A��ϡHNO3��Ӧ�������������ڿ�����Ѹ�ٱ�Ϊ����ɫ���������ɫ����ɫ�����巢���Ļ�ѧ��Ӧ����ʽΪ2NO+O2=2NO2��2NO2?N2O4
��2����������B�����ΪAl��OH��3��CuO�������ɹ���B�Ĺ����У������NaOH�ļ���������NaOH�����������������ķ�Ӧ�����ӷ���ʽΪAl��OH��3+OH-=AlO2-+2H2O
��3��������չ�����һ����Ӧ�Ļ�ѧ����ʽ��
4CuO+2Al2O3$\frac{\underline{\;����\;}}{\;}$4CuAlO2+O2����
��4������ͭ�Ͻ���ͭ����������Ϊ63.5%��������5.0kg�����е�ͭ����ȫת��Ϊ50.0mol CuAlO2��������Ҫ1.0mol•L-1��Al2��SO4��3��Һ25.0L��
��5����ͭ���Ͻ��м�������ϡ���ᣬд��������Ӧ�����ӷ���ʽ3Cu+2NO3-+8H+=2NO��+4H2O+3Cu2+��
���� �ɹ�������ͼ��֪�������ڿ���������ʱ��Cu�������������к���CuO������Ag���������м���������������CuO�����ᷴӦ�����˵õ�����ͭ��Һ���������ᣩ������AΪAg������Һ�м������������������ƣ��õ�����������������ͭ�������л�õ�CuO��Al2O3�����߷�Ӧ�õ�CuAlO2�����߷�Ӧ�õ�CuAlO2��
��1����ɫ��NO�ڳ�����������е�O2Ѹ�ٻ������ɺ���ɫN02��N02�����ת����������������
��2��δ���֮ǰ��Cu��OH��2��Al��OH��3������Al��OH��3��Cu��OH��2��ʼ�ֽ���¶ȷֱ�Ϊ450���80���жϲ������������������������Ӧ��
��3��������������֪��CuO��Al2O3��Ӧ�õ�CuAlO2���ɵ���غ��֪��OԪ�ػ��ϼ����ߣ����������ɣ���ƽ��ɷ���ʽ��
��4������ͭԪ���غ㽨����ϵʽ��Cu��CuAlO2�����㣻������Ԫ���غ㽨����ϵʽ��Al2��SO4��3��2CuAlO2�����㣻
��5����ͭ���Ͻ��м�������ϡ���ᣬͭ��ϡ���ᷴӦ��������ͭ��һ��������ˮ��
��� �⣺��1����ɫ��NO�ڳ�����������е�O2Ѹ�ٻ������ɺ���ɫN02��2NO+O2=2NO2��N02�����ת������������������Ӧ����ʽΪ��2NO2?N2O4��
�ʴ�Ϊ��2NO+O2=2NO2��2NO2?N2O4��
��2��δ���֮ǰ��Cu��OH��2��Al��OH��3��Al��OH��3��Cu��OH��2��ʼ�ֽ���¶ȷֱ�Ϊ450���80�棬��к�Cu��OH��2�ֽ��������ͭ����֪BΪAl��OH��3��CuO��������������������Ӧ��Al��OH��3+NaOH=NaAlO2+2H2O��
�ʴ�Ϊ��Al��OH��3��CuO��Al��OH��3+OH-=AlO2-+2H2O��
��3��������������֪��CuO��Al2O3��Ӧ�õ�CuAlO2���ɵ���غ��֪��OԪ�ػ��ϼ����ߣ����������ɣ���ӦǰCuΪ+2�ۣ���Ӧ��Ϊ+1�����ϼ۽���1����Ӧǰ��Ϊ-2�ۣ���Ӧ��Ϊ0�����ϼ�����2�����ߵ���С��������2���ٸ���ԭ���غ�ã�4CuO+2Al2O3=4CuAlO2+O2����
�ʴ�Ϊ��4��2��4CuAlO2��O2��
��4���ɹ�ϵʽ��Cu��CuAlO2��n��CuAlO2��=$\frac{5000g��63.5%}{64g/mol}$=50.0mol����CuAlO2Ϊ50.0mol��
�ɹ�ϵʽ��Al2��SO4��3��2CuAlO2��Al2��SO4��3=n��CuAlO2����$\frac{1}{2}$=25.0mol��������Ҫ���Ϊ$\frac{25.0mol}{1.0mol•L{\;}^{-1}}$=25.0L��
�ʴ�Ϊ��50.0��25.0��
��5����ͭ���Ͻ��м�������ϡ���ᣬͭ��ϡ���ᷴӦ��������ͭ��һ��������ˮ����Ӧ�����ӷ���ʽΪ3Cu+2NO3-+8H+=2NO��+4H2O+3Cu2+��
�ʴ�Ϊ��3Cu+2NO3-+8H+=2NO��+4H2O+3Cu2+��
���� �����Դӷ����л��������Ʊ�ͭ������ƷΪ�����������˵��ԭ�����������жϡ�������ԭ��Ӧ����ƽ�Լ���ѧ���㣬�ѶȲ���

 ��У����ϵ�д�
��У����ϵ�д�| A�� | NH+4 Na+ NO-3 Cl- | B�� | Fe2+ K+ HS- ClO- | ||
| C�� | Ca2+ K+ SO32- CH3COO- | D�� | K+ Na+ Cl- SO42- |
| A�� | Cu��IO3��2����������Cu��IO3��2�е�ͭ�͵�Ԫ�ر���ԭ | |
| B�� | CuI���������������ǻ�ԭ���� | |
| C�� | ÿ����1 mol CuI����12 mol KI����������Ӧ | |
| D�� | ÿת��1.1 mol���ӣ���0.2 mol IO3-������ |
| A�� | ��ͬ | B�� | �к�CH3COOH�Ķ� | C�� | �к�H2SO4�Ķ� | D�� | ���Ƚ� |
| A�� | Cu2+ H+ SO42- | B�� | Ba2+ NO3- CO32- | ||
| C�� | Al3+ H+ CO32- | D�� | OH- Cu2+ NO3- |
| A�� | 2H2+O2��2H2O+142.9kJ | B�� | 2H2��g��+O2��g����2H2O��l��+142.9kJ | ||
| C�� | 2H2��g��+O2��g����2H2O��l��+571.6kJ | D�� | H2O��l����H2��g��+$\frac{1}{2}$O2��g��-142.9kJ |
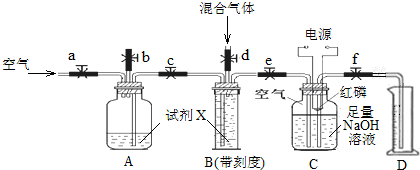
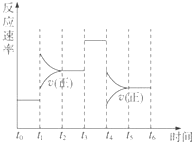 ������̶����ܱ���ϵ�з������з�Ӧ��
������̶����ܱ���ϵ�з������з�Ӧ��