��Ŀ����
20����NAΪ�����ӵ�������ֵ������������ȷ���ǣ�������| A�� | 12.0g���ڵ�NaHSO4�к��е���������Ϊ0.2NA | |
| B�� | Na2O2������CO2��Ӧ����2.24LO2��ת�Ƶ�����Ϊ0.2NA | |
| C�� | 4.6gNO2��N2O4������к��е�ԭ������Ϊ0.3NA | |
| D�� | �ö��Ե缫���1LŨ�Ⱦ�Ϊ2mol/L��AgNO3��Cu��NO3��2�Ļ����Һ������0.2NA������ת��ʱ����������������6.4g���� |
���� A���������������ƻ����ۼ����������Ƶ���������Ӻ�����������ӣ�12.0g�������Ƶ����ʵ���Ϊ0.1mol������0.1mol�����������ӣ�
B����֪���������Ƶ�����
C��NO2��N2O4�����ʽ��ΪNO2��
D��Ag+�����Դ���Cu2+��
��� �⣺A��12.0g���ڵ�NaHSO4�����ʵ���Ϊ0.1mol��0.1mol�������������к���0.1mol�����Ӻ�0.1mol����������ӣ����Ժ��е���������Ϊ0.1NA����A����
B����֪���������Ƶ����������㣬��B����
C��NO2��N2O4�����ʽ��ΪNO2����4.6g������к�NO2�����ʵ���Ϊn=$\frac{4.6g}{46g/mol}$=0.1mol����0.3molԭ�Ӽ�0.3NA����C��ȷ��
D��Ag+�����Դ���Cu2+��1LŨ�Ⱦ�Ϊ2mol/L��AgNO3��Cu��NO3��2�Ļ����Һ������0.2mol����ת��ʱ����������Ag������Ϊ0.2mol��108g/mol=21.6g����D����
��ѡC��
���� ���⿼���˰���٤���������йؼ��㣬���չ�ʽ��ʹ�ú����ʵĽṹ��״̬�ǽ���ؼ����ѶȲ���

��ϰ��ϵ�д�
 Ӧ������ҵ��ϵ�д�
Ӧ������ҵ��ϵ�д�
�����Ŀ
10��NAΪ����٤��������ֵ������˵����ȷ���ǣ�������
| A�� | ����������ˮ��Ӧʱ������0.1mol����ת�Ƶĵ�����Ϊ0.4NA | |
| B�� | 23g Na ������H2O��Ӧ��ȫ�������NA��H2���� | |
| C�� | ��״���£�22.4LN2��H2������к�NA��ԭ�� | |
| D�� | 3mol����Fe��ȫת��ΪFe3O4��ʧȥ8 NA������ |
15��Ԫ�����ڱ�����ʽ���ֶ�������ͼ������Ԫ�����ڱ���һ���֣��Ա���ѧ����Ԫ�����ڱ���������Ԫ�ص������ɣ��ж�����˵����ȷ���ǣ�������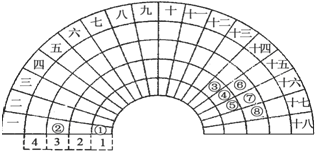

| A�� | ��Ԫ���γɵĵ��ʾ��������ԭ�Ӿ��� | |
| B�� | ��Ԫ�ص���̬�⻯����Һ��������Ϊ����Ӽ������� | |
| C�� | �ڡ��ߡ����Ӧ�ļ����Ӱ뾶���μ�С | |
| D�� | �ڢݢ�����Ԫ���γɵ�����Һ��25��ʱ��pHֵΪ7 |
12�������й���Һ��ɵ������������ǣ�������
| A�� | ������Һ�п��ܴ�������Fe2+��Cl?��NO3?��Na+ | |
| B�� | ��ɫ��Һ�п��ܴ�������Mg2+��NH4+��Cl?��SO42- | |
| C�� | ǿ������Һ�п��ܴ�������NH4+��K+��Cl?��SO42? | |
| D�� | ������Һ�п��ܴ�������Fe3+��K+��I?��SO42- |
9������ʵ��������¼��ȷ���ǣ�������
| A�� | ������ζ�����������Һʱ����ƿ����ȡ25.00mL������������Һ | |
| B�� | ����Ͳ�����ˮ���ռ��Ƶõ��������Ϊ50.28 mL | |
| C�� | ����ֻ250 mL������ƿ����0.1 mol•L-1 500 mL��NaOH��Һ | |
| D�� | ��������ƽ��ȡ2.50 g���������ȳ��ʧˮ������������0.90 g |


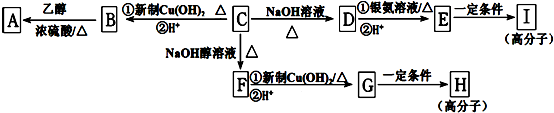
 ����Ӧ����Ϊ�Ӿ۷�Ӧ��
����Ӧ����Ϊ�Ӿ۷�Ӧ�� ��
��
 ��
��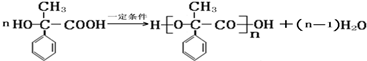 ���÷�Ӧ���������۷�Ӧ��
���÷�Ӧ���������۷�Ӧ�� ���÷�Ӧ�����ǼӾ۷�Ӧ��
���÷�Ӧ�����ǼӾ۷�Ӧ�� ��
��