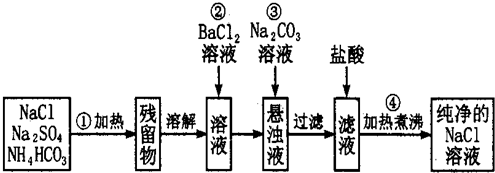
��Ŀ����
10��ijѧ����Ҫ���ռ��������0.5mol•L-1��NaOH��Һ500mL��ʵ�����ṩ������������100mL�ձ�����1000mL����ƿ����500mL����ƿ���ܲ���������������ƽ�������룩��
��ش��������⣺
��1�����㣺��Ҫ��ȡNaOH����10.0g��
��2������ʱ������ʹ�õ������Т٢ۢܢݣ�����ţ�����ȱ�ٵ������ǽ�ͷ�ιܡ�ҩ�ף������������ƣ�
��3��ʵ�������õ��������������÷ֱ��ǣ������ڽ��衢������������
��4������ʱ������ȷ�IJ���˳����BCAFED������ĸ��ʾ��ÿ������ֻ��һ�Σ���
A��������ˮϴ���ձ�2�Ρ�3�Σ�ϴ��Һ��ע������ƿ����
B����ʢ��NaOH������ձ��м�������ˮ�ܽ�
C�����ձ�������ȴ����Һ�ز�����ע������ƿ��
D��������ƿ�ǽ����������µߵ���ҡ��
E�����ý�ͷ�ιܼ�ˮ��ʹ��Һ��Һ��ǡ����̶�����
F������������ƿ��С�ļ�ˮ��ֱ��Һ��ӽ��̶�1cm��2cm��
��5��������������������н�����������ҺŨ��ƫ�ߵ����������ƣ����ţ���
������ƿʵ��ǰ������ˮϴ�ɾ�����δ���
�ڶ��ݹ۲�Һ��ʱ����
�����ƹ�������©�ˣ�3���в���A
�ܼ�����ˮʱ���������˿̶���
��6����ʵ������г��֣�5���Т���������㽫��δ�����
���� ��1������m=nM=cvM���㣻
��2������ʵ������IJ����Լ�ÿ��������Ҫ����ȷ����Ӧ����������
��3�����ݲ�������ʵ���е����ã�
��4�����ݲ������裺�������ܽ⡢��Һ��ϴ�ӡ����ݡ�ҡ�ȵ������
��5������c=$\frac{n}{V}$�������������ʵ����ʵ��������Һ�������Ӱ���жϣ�
��6������ʵ��������ֵĴ���Ӧ�������ƣ�
��� �⣺��1��ʵ��������0.5mol•L-1��NaOH��Һ500mL��ҪNaOH������Ϊ��0.5L��0.5mol•L-1��40g/mol=10.0g��
�ʴ�Ϊ��10.0��
��2�����������г������ܽ⡢��Һ��ϴ�ӡ����ݡ�ҡ�ȵȲ�����һ����������ƽ��������ҩ��ȡ��ҩƷ�����ձ����ܽ⣬��ȴ��ת�Ƶ�500mL����ƿ�У����ò���������������ˮ��Һ�����̶���1��2cmʱ�����ý�ͷ�ιܵμӣ����Ա���ʹ�õ������Т�100mL�ձ�����500mL����ƿ���ܲ���������������ƽ�������룩������Ҫ������Ϊ��ͷ�ιܡ�ҩ�ף�
�ʴ�Ϊ���٢ۢܢݣ���ͷ�ιܣ�ҩ�ף�
��3�����������ܽ�ʱ�������ã�����Һʱ���������ã�
�ʴ�Ϊ�����裻������
��4�����������г������ܽ⡢��Һ��ϴ�ӡ����ݡ�ҡ�ȵȲ�����
�ʴ�Ϊ��BCAFED��
��5��������ƿʵ��ǰ������ˮϴ�ɾ�����δ��ɣ���Ӱ�죬Ũ�Ȳ��䣬�ʢٴ���
�ڶ��ݹ۲�Һ��ʱ���ӣ�Ũ��ƫ�ʢ���ȷ��
�����ƹ�������©�ˣ�4���в���A�����ʵ��������٣�Ũ��ƫ�ͣ��ʢ۴���
�ܼ�����ˮʱ���������˿̶ȣ���Һ���ƫ��Ũ��ƫС���ʢܴ���
�ʴ�Ϊ���ڣ�
��6��ʵ��������ּ�����ˮʱ���������˿̶ȣ�����Һ��Ũ��ƫС��Ӧ�������ƣ�
�ʴ�Ϊ���������ƣ�
���� ���⿼��һ�����ʵ���Ũ����Һ�����ƣ��ѶȲ�����ȷ����ԭ���ǽ���ؼ�����Ŀ�ѶȲ���

 ���ٴ�����ɽ����ϵ�д�
���ٴ�����ɽ����ϵ�д�
�����漰����Ҫ��Ӧ�У�
����Ӧ��4FeO•Cr2O3+8Na2CO3+7O2$\frac{\underline{\;\;��\;\;}}{\;}$8Na2CrO4+2Fe2O3+8CO2
����Ӧ��SiO2+Na2CO3$\frac{\underline{\;\;��\;\;}}{\;}$Na2SiO3+CO2����Al2O3+Na2CO3$\frac{\underline{\;\;��\;\;}}{\;}$2NaAlO2+CO2��
����������������������ʽ��ȫ����ʱ��Һ��pH��
| ������ | Al��OH��3 | Fe��OH��3 | Mg��OH��2 | Cr��OH��3 |
| ��ȫ����ʱ��ҺpH | 4.7 | 3.7 | 11.2 | 5.6 |
��1�����١��з�Ӧ���ڻ�תҤ�н��У���Ӧʱ�費�Ͻ��裬��������ʹ��Ӧ��Ӵ�����֣��ӿ췴Ӧ���ʣ�
��2�����ۡ��е���pH��4.7��Ŀ���dz�ȥ��Һ�е�AlO2-��SiO32-��
��3�����ݡ��м������ữ��Ŀ����ʹCrO42-ת��ΪCr2O72-����д����ƽ��ת�������ӷ���ʽ��2CrO42-+2H+?Cr2O72-+H2O��
��4����ȡ�ظ���������2.5000g���250mL��Һ��ȡ��25.00mL�ڵ���ƿ�У�����10mL 2mol•L-1H2SO4�������⻯�ƣ����Ļ�ԭ����ΪCr3+�������ڰ���5min��Ȼ�����100mLˮ������3mL����ָʾ������0.1200mol•L-1Na2S2O3����Һ�ζ���I2+2S2O32-�T2I-+S4O62-����
���жϴﵽ�ζ��յ�������ǣ����μ����һ�������������Һʱ����Һ��ɫ��ȥ��������ڲ���ɫ��
����ʵ���й���ȥNa2S2O3����Һ40.00ml�����ò�Ʒ�����ظ����ƵĴ��ȣ��������������������ʲ����뷴Ӧ��83.84%��
| A�� | Ư�۵���Ч�ɷ�������� | B�� | ����������ˮ�γɵ��������ǿ�� | ||
| C�� | �⻯��ˮ��Һ��ʹ���۱��� | D�� | ����ͨ��ˮ������������ |
| A�� | CO2 | B�� | H2O | C�� | NaOH | D�� | H2SO4 |
| A�� | 7 | B�� | 6 | C�� | 5 | D�� | 4 |
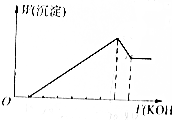 ��þ���Ͻ���ϡH2SO4��Һ��ȫ�ܽ����������Һ�м���KOH��Һ����������ɳ�����������ϵ��ͼ��ʾ����Ͻ���þ�������������ǣ�������
��þ���Ͻ���ϡH2SO4��Һ��ȫ�ܽ����������Һ�м���KOH��Һ����������ɳ�����������ϵ��ͼ��ʾ����Ͻ���þ�������������ǣ�������| A�� | 1��1 | B�� | 8��9 | C�� | 4��3 | D�� | 4��9 |