��Ŀ����
��ͼ����Ԫ�����ڱ���һ���֣���Ա��еĢ١�����Ԫ�أ���ջش�
��1������ЩԪ���У���ѧ��������õ�ԭ�ӵ�ԭ�ӽṹʾ��ͼΪ ��
��2���ۢݢ�Ԫ��ԭ���γɼ����ӵ����Ӱ뾶�ɴ�С��˳���� �������ӷ��ţ���
��3���ޢ���Ԫ���γɵĻ������侧�������� ���û��������ʽΪ ���ɢ�Ԫ���γɵĵ�����ṹʽΪ ��
��4���ܵĵ�����ˮ��Ӧ�����ӷ���ʽ�� ��
��5����֪ijЩ��ͬ��Ԫ�ص�����Ҳ��һ���������ԣ���Ԫ�آ����Ԫ�ص��������������Ƶ����ʣ�д��Ԫ�آٵ�����������NaOH��Һ��Ӧ�Ļ�ѧ����ʽ ��
| ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 | |
| 2 | �� | �� | �� | |||||
| 3 | �� | �� | �� | �� | �� |
��2���ۢݢ�Ԫ��ԭ���γɼ����ӵ����Ӱ뾶�ɴ�С��˳����
��3���ޢ���Ԫ���γɵĻ������侧��������
��4���ܵĵ�����ˮ��Ӧ�����ӷ���ʽ��
��5����֪ijЩ��ͬ��Ԫ�ص�����Ҳ��һ���������ԣ���Ԫ�آ����Ԫ�ص��������������Ƶ����ʣ�д��Ԫ�آٵ�����������NaOH��Һ��Ӧ�Ļ�ѧ����ʽ
���㣺Ԫ�������ɺ�Ԫ�����ڱ����ۺ�Ӧ��
ר�⣺Ԫ����������Ԫ�����ڱ�ר��
��������Ԫ�������ڱ���λ�ã���֪��ΪBe����ΪN����ΪF����ΪNa����ΪAl����ΪSi����ΪCl����ΪAr��
��1��Arԭ�ӻ�ѧ��������ã�ԭ�Ӻ�����18�����ӣ���3�����Ӳ㣬���������Ϊ2��8��8��
��2�����Ӳ�ṹ��ͬ���˵����Խ�����Ӱ뾶ԽС�����Ӳ�Խ�����Ӱ뾶Խ��
��3���ޢ���Ԫ���γɵĻ�����ΪSiCl4�����ڷ��Ӿ��壬������Si��Clԭ��֮���γ�1�Թ��õ��Ӷԣ��ɢ�Ԫ���γɵĵ���ΪN2��������Nԭ��֮���γ�3�Թ��õ��Ӷԣ�
��4������ˮ��Ӧ��������������������
��5��Be��OH��2�������������������ƣ�Be��OH��2���������Ʒ�Ӧ����Na2BeO2��ˮ��
��1��Arԭ�ӻ�ѧ��������ã�ԭ�Ӻ�����18�����ӣ���3�����Ӳ㣬���������Ϊ2��8��8��
��2�����Ӳ�ṹ��ͬ���˵����Խ�����Ӱ뾶ԽС�����Ӳ�Խ�����Ӱ뾶Խ��
��3���ޢ���Ԫ���γɵĻ�����ΪSiCl4�����ڷ��Ӿ��壬������Si��Clԭ��֮���γ�1�Թ��õ��Ӷԣ��ɢ�Ԫ���γɵĵ���ΪN2��������Nԭ��֮���γ�3�Թ��õ��Ӷԣ�
��4������ˮ��Ӧ��������������������
��5��Be��OH��2�������������������ƣ�Be��OH��2���������Ʒ�Ӧ����Na2BeO2��ˮ��
���
�⣺��Ԫ�������ڱ���λ�ã���֪��ΪBe����ΪN����ΪF����ΪNa����ΪAl����ΪSi����ΪCl����ΪAr��
��1��Arԭ�ӻ�ѧ��������ã�ԭ�Ӻ�����18�����ӣ���3�����Ӳ㣬���������Ϊ2��8��8��ԭ�ӽṹʾ��ͼΪ��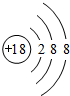 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��2�����Ӳ�ṹ��ͬ���˵����Խ�����Ӱ뾶ԽС�����Ӳ�Խ�����Ӱ뾶Խ�����Ӱ뾶Ϊ��Cl-��F-��Al3+���ʴ�Ϊ��Cl-��F-��Al3+��
��3���ޢ���Ԫ���γɵĻ�����ΪSiCl4�����ڷ��Ӿ��壬������Si��Clԭ��֮���γ�1�Թ��õ��Ӷԣ������ʽΪ ���ɢ�Ԫ���γɵĵ���ΪN2��������Nԭ��֮���γ�3�Թ��õ��Ӷԣ��ṹʽΪN��N���ʴ�Ϊ�����Ӿ��壻
���ɢ�Ԫ���γɵĵ���ΪN2��������Nԭ��֮���γ�3�Թ��õ��Ӷԣ��ṹʽΪN��N���ʴ�Ϊ�����Ӿ��壻 ��N��N��
��N��N��
��4������ˮ��Ӧ�����������������������ӷ���ʽΪ��2Na+2H2O=2Na++2OH-+H2�����ʴ�Ϊ��2Na+2H2O=2Na++2OH-+H2����
��5��Be��OH��2�������������������ƣ�Be��OH��2���������Ʒ�Ӧ����Na2BeO2��ˮ����Ӧ����ʽΪ��Be��OH��2+2 NaOH=Na2BeO2+2H2O���ʴ�Ϊ��Be��OH��2+2 NaOH=Na2BeO2+2H2O��
��1��Arԭ�ӻ�ѧ��������ã�ԭ�Ӻ�����18�����ӣ���3�����Ӳ㣬���������Ϊ2��8��8��ԭ�ӽṹʾ��ͼΪ��
 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
����2�����Ӳ�ṹ��ͬ���˵����Խ�����Ӱ뾶ԽС�����Ӳ�Խ�����Ӱ뾶Խ�����Ӱ뾶Ϊ��Cl-��F-��Al3+���ʴ�Ϊ��Cl-��F-��Al3+��
��3���ޢ���Ԫ���γɵĻ�����ΪSiCl4�����ڷ��Ӿ��壬������Si��Clԭ��֮���γ�1�Թ��õ��Ӷԣ������ʽΪ
 ���ɢ�Ԫ���γɵĵ���ΪN2��������Nԭ��֮���γ�3�Թ��õ��Ӷԣ��ṹʽΪN��N���ʴ�Ϊ�����Ӿ��壻
���ɢ�Ԫ���γɵĵ���ΪN2��������Nԭ��֮���γ�3�Թ��õ��Ӷԣ��ṹʽΪN��N���ʴ�Ϊ�����Ӿ��壻 ��N��N��
��N��N����4������ˮ��Ӧ�����������������������ӷ���ʽΪ��2Na+2H2O=2Na++2OH-+H2�����ʴ�Ϊ��2Na+2H2O=2Na++2OH-+H2����
��5��Be��OH��2�������������������ƣ�Be��OH��2���������Ʒ�Ӧ����Na2BeO2��ˮ����Ӧ����ʽΪ��Be��OH��2+2 NaOH=Na2BeO2+2H2O���ʴ�Ϊ��Be��OH��2+2 NaOH=Na2BeO2+2H2O��
���������⿼��Ԫ�����ڱ���Ԫ��������Ӧ�ã����ضԻ�ѧ����Ŀ��飬�ѶȲ���ע���Ԫ�������ɵ��������գ�

��ϰ��ϵ�д�
 ���б�ˢ��ϵ�д�
���б�ˢ��ϵ�д�
�����Ŀ
��֪BrCl�ܷ������з�Ӧ��nBrCl+2M=MCln+MBrn��MΪ+n�۵Ľ���Ԫ�أ���BrCI+H20=HCl+HBrO������������ȷ���ǣ�������
| A��BrCl���к�±�ص������ƵĻ�ѧ���� |
| B��BrCl��M��Ӧʱ��BrCl��Ϊ��������Ϊ��ԭ�� |
| C��BrCl��NaOH��Һ��Ӧ����NaCI��NaBrO������ |
| D����BrCl��FeS04��Һ��Ӧ����Һ������ɫ |
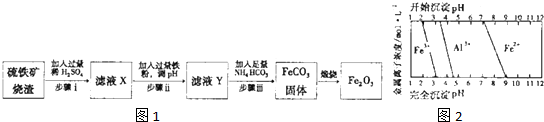
 ���ڵؿ��еĺ���Լռ 5%���ң�����ʯ������ܶ࣬��Ҫ���д�����ʯ����Ҫ�ɷ���Fe3O4����������ʯ����Ҫ�ɷ��� Fe2O3���ȣ�
���ڵؿ��еĺ���Լռ 5%���ң�����ʯ������ܶ࣬��Ҫ���д�����ʯ����Ҫ�ɷ���Fe3O4����������ʯ����Ҫ�ɷ��� Fe2O3���ȣ�