��Ŀ����
2��������Һ����Ũ�ȹ�ϵ��ȷ���ǣ�������| A�� | �����½�pH=12�İ�ˮ��ˮϡ�ͣ�ϡ��������Һ��$\frac{c��O{H}^{-}��}{c��N{H}_{3}•{H}_{2}O��}$��С | |
| B�� | ͬŨ�ȵĴ����ƺʹ����������Һ�У�c��Na+����c��CH3COO-����c��H+����c��OH-�� | |
| C�� | 25����ͬpH�Ģ�CH3COONa��NaHCO3��NaAlO2������Һ�е�c��Na+�����٣��ڣ��� | |
| D�� | Ũ�Ⱦ�Ϊ0.1mol•L-1��KHS��HCl��������Һ�У�c��HS-��+c��H2S��=0.1mol•L-1 |
���� A����ˮ��ˮϡ�ͣ��ٽ�һˮ�ϰ��ĵ��룬��n��OH-������n��NH3��H2O����С��
B��ͬŨ�ȵĴ����ƺʹ����������Һ����ĵ�������ε�ˮ�⣬�����ԣ�
C�������Դ��̼�������������֪��ͬpH�Ģ�CH3COONa��NaHCO3��NaAlO2������Һ��Ũ��Ϊ�٣��ڣ��ۣ�
D����������Һ����Һ���Ϊԭ����2������������غ������
��� �⣺A����ˮ��ˮϡ�ͣ��ٽ�һˮ�ϰ��ĵ��룬��n��OH-������n��NH3��H2O����С����ϡ��������Һ��$\frac{c��O{H}^{-}��}{c��N{H}_{3}•{H}_{2}O��}$����A����
B��ͬŨ�ȵĴ����ƺʹ����������Һ����ĵ�������ε�ˮ�⣬�����ԣ�������Ũ��Ϊc��CH3COO-����c��Na+����c��H+����c��OH-��������ѭ����غ㣬��B����
C�������Դ��̼�������������֪��ͬpH�Ģ�CH3COONa��NaHCO3��NaAlO2������Һ��Ũ��Ϊ�٣��ڣ��ۣ���������Һ�е�c��Na+�����٣��ڣ��ۣ���C��ȷ��
D����������Һ����Һ���Ϊԭ����2�����������غ��֪c��S2-��+c��HS-��+c��H2S��=$\frac{0.1mol/L}{2}$=0.05mol•L-1����D����
��ѡC��
���� ���⿼��������ʵĵ��뼰����Ũ�ȵıȽϣ�Ϊ��Ƶ���㣬���յ��롢ˮ�⼰����غ㡢�����غ�Ϊ���Ĺؼ������ط�����Ӧ�������Ŀ��飬�ۺ��Խ�ǿ����Ŀ�ѶȲ���

| A�� | �մ�--NaHCO3 | B�� | ����--C2H5OH | C�� | ����-KAl��SO4��2 | D�� | ��ʯ��-CaO |
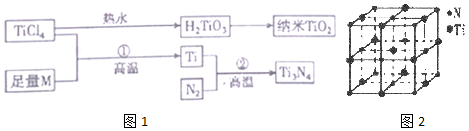
ͼ�е�M�Ƕ����ڽ���Ԫ�أ�M�IJ��ֵ����������
| I1 | I2 | I3 | I4 | I5 | |
| ������/��kJ•mol-1�� | 738 | 1451 | 7733 | 10 540 | 13 630 |
��1��TiO2Ϊ���Ӿ��壬��֪�����������ӵ���λ��Ϊ6�������ӵ���λ��Ϊ3���������ӵĵ����Ų�ʽΪ1s22s22p63s23p6
��2������Ti�����M�ľ���ԭ�Ӷѻ�ģʽ��ͬ����ѻ�ģ��Ϊ�������ܶѻ� ����д�ѻ�ģ�����ƣ���������ԭ���ڶ�άƽ�������λ��Ϊ6
��3��������TiCl4Ϊ��ɫҺ�壬�е�Ϊ136.4�棬�ɴ˿�֪�侧������Ϊ���Ӿ��壬���ɸþ�������ӵĿռ乹��Ϊ��������
��4����һ�ֵ����Ѿ���ľ�����NaCl�������ƣ���2ͼ��ʾ����֪�õ����ѵ��ܶ�Ϊp g•cm-3����þ�����N��Ti֮����������Ϊ$\frac{1}{2}\root{3}{\frac{4��62}{��{N}_{A}}}��1{0}^{10}$ pm����NAΪ�����ӵ³�������ֵ��ֻ����ʽ�����þ�������Tiԭ�Ӿ�������������Tiԭ����12 ����
��5����ѧ��ͨ��X����̽��KC1��MgO��CaO��TiN�ľ�����NaCl�ľ���ṹ���ƣ���֪�������Ӿ���ľ��������������
| ���Ӿ��� | NaCl | KC1 | CaO |
| ������/��kJ•mol-1�� | 786 | 715 | 3401 |
| A�� | ���³�ѹ�£�46 gN2O4�к���ԭ����Ϊ2NA | |
| B�� | ���³�ѹ�£�11.2 LNH3�к����ۼ�����ĿΪ1.5NA | |
| C�� | ��⾫��ͭ������������32 gʱ��·��ת�Ƶ�������ΪNA | |
| D�� | 1 L 0.2 mol/LNa2SO4��Һ�к���������������Ϊ0.6NA |
| A�� | һ�������£���1molN2��3molH2��Ϸ�����Ӧ��ת�Ƶĵ�������Ϊ6NA | |
| B�� | 1L0.1mol•L-1��Na2CO3��Һ�������ӵ���������0.1NA | |
| C�� | ��FeI2��Һ��ͨ������Cl2������2molFe2+������ʱ������Cl2�ķ�����ΪNA | |
| D�� | 1mol-CH3�������ĵ�������Ϊ10NA |
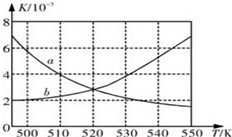 ��Ϊ�˼���CO���ŷţ�ij�����о�С����CO��H2 Ϊԭ�Ϻϳ������Դ�����ѣ�DME������Ӧ���£�4H2��g��+2CO��g��
��Ϊ�˼���CO���ŷţ�ij�����о�С����CO��H2 Ϊԭ�Ϻϳ������Դ�����ѣ�DME������Ӧ���£�4H2��g��+2CO��g��CH3OCH3��g��+H2O��g����H=-198kJ/mol��
��1����ͼ��ʾ����ȷ��Ӧƽ�ⳣ��K���¶ȱ仯��ϵ��������a�������߱����ĸ�������ж�����������Ӧ���ȣ��¶����ߣ�ƽ�ⳣ����С��
��2����һ���¶��£���2.0L�̶����ܱ������г���2molH2��1molCO������һ��ʱ���Ӧ4H2��g��+2CO��g��
CH3OCH3��g��+H2O��g�� �ﵽƽ�⣮��Ӧ�����в�õIJ������ݼ��±���
| ʱ��/min | 0 | 20 | 40 | 80 | 100 |
| n��H2��/mol | 2.0 | 1.4 | 0.85 | 0.4 | - |
| n��CO��/mol | 1.0 | - | 0.425 | 0.2 | 0.2 |
| n��CH3OCH3��/mol | 0 | 0.15 | - | - | 0.4 |
| n��H2O��/mol | 0 | 0.15 | 0.2875 | 0.4 | 0.4 |
�ڴ�ƽ��ʱ��H2 ��ת����Ϊ��80%��
���������¶��£���ƽ����2L�������ٳ���0.4molH2 ��0.4molCH3OCH3 ��g������ѧƽ�����������������������ƶ���
��3����Li2CO3 ��Na2CO3 �������λ������Ϊ����ʣ�COΪ����ȼ����������CO2 �Ļ��������Ϊ������ȼ�����Ƶ���650�湤����ȼ�ϵ�أ�����ӦʽΪ��2CO+2CO32--4e-=4CO2��ȼ�ϵ�صĵ�·����0.2NA�����ӷ���ת�ƣ�����������2.24��CO2��
����֪��25��ʱ��Ksp[Mg��OH��2]=1.8��10-11��Ksp[Cu��OH��2]=2.2��10-20����25���£���Ũ�Ⱦ�Ϊ0.1mol/L��MgCl2 ��CuCl2 �����Һ����μ��백ˮ��������Cu��OH��2�������ѧʽ�����ɸó��������ӷ���ʽΪCu2++2NH3•H2O?Cu��OH��2��+2NH4+��
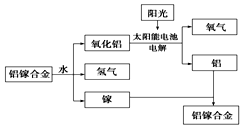 �����նȴ�ѧ�о�������һ���������غϽ��Ʊ��������¹��գ���ͼ��ʾ���������йظù��յ�˵��������ǣ�������
�����նȴ�ѧ�о�������һ���������غϽ��Ʊ��������¹��գ���ͼ��ʾ���������йظù��յ�˵��������ǣ�������| A�� | ���غϽ���ˮ��Ӧ�Ļ�ѧ����ʽΪ2Al+3H2O$\frac{\underline{\;һ������\;}}{\;}$Al2O3+3H2�� | |
| B�� | �ܷ�ӦʽΪ2H2O$\frac{\underline{\;һ������\;}}{\;}$2H2��+O2�� | |
| C�� | �ù����У�������ת����ʽֻ������ | |
| D�� | ���غϽ����ѭ��ʹ�� |
| A�� | 2Al2O3+3C$\frac{\underline{\;����\;}}{\;}$4Al+3CO2�� | B�� | CuCl2$\frac{\underline{\;���\;}}{\;}$Cu+Cl2�� | ||
| C�� | Fe3O4+4CO$\frac{\underline{\;����\;}}{\;}$3Fe+4CO2 | D�� | 2HgO$\frac{\underline{\;���\;}}{\;}$2Hg+O2�� |