��Ŀ����
��ͼ��ʾ�й����ʣ����ɶ�����Ԫ���γɣ�֮���ת����ϵ������AΪ�����Ľ������ʣ�BΪ�ǽ������ʣ�һ���Ǻ�ɫ��ĩ����C�dz�������ɫ��ζҺ�壬D�ǵ���ɫ�Ĺ��廯�������Ӧ����ͼ����ʡ�ԣ���

��1��A�Ļ�ѧʽΪ ��K�Ļ�ѧʽΪ ��D�ĵ���ʽΪ ��
��2����Ӧ���е�C��D���������÷�Ӧ���ܻ�ѧ����ʽ�� ��
��3����Ӧ���У���B��F���ʵ���֮��Ϊ4��3��G��H������֮��Ϊ ��
��4����Ӧ�ܵ����ӷ���ʽΪ ��
��5����A���������Fe2O3�Ļ����a g���ܽ��ڹ�����200mL c��H+��=0.1mol?L-1��������Һ�У�Ȼ�������м���NaOH��Һ�պ�ʹ�����������ʱ����ȥNaOH ��Һ100mL����NaOH ��Һ��Ũ��Ϊ ��

��1��A�Ļ�ѧʽΪ
��2����Ӧ���е�C��D���������÷�Ӧ���ܻ�ѧ����ʽ��
��3����Ӧ���У���B��F���ʵ���֮��Ϊ4��3��G��H������֮��Ϊ
��4����Ӧ�ܵ����ӷ���ʽΪ
��5����A���������Fe2O3�Ļ����a g���ܽ��ڹ�����200mL c��H+��=0.1mol?L-1��������Һ�У�Ȼ�������м���NaOH��Һ�պ�ʹ�����������ʱ����ȥNaOH ��Һ100mL����NaOH ��Һ��Ũ��Ϊ
���㣺������ƶ�
ר�⣺�ƶ���
������C�dz�������ɫ��ζҺ��˵��C��ˮ��D�ǵ���ɫ�Ĺ��廯�������ˮ��CO2��Ӧ������֪DΪNa2O2����Ϸ�Ӧ�ٽ���A�������Һ��Ӧ�����������ɵ�AΪAl����ΪNa2AlO2��E��F�ֱ�ΪO2��H2�е�һ�֣�BΪ�ǽ������ʣ�һ���Ǻ�ɫ��ĩ��������F��Ӧ����G��H����G����������Ʒ�Ӧ������֪BΪ̼��FΪO2��EΪH2��GΪCO2��HΪCO�����ݷ�Ӧ�ۿ���֪KΪNa2CO3����Һ��ΪNa2CO3��Һ���ɷ�Ӧ�ܿ�֪LΪAl��OH��3���ݴ˽��
���
�⣺C�dz�������ɫ��ζҺ��˵��C��ˮ��D�ǵ���ɫ�Ĺ��廯�������ˮ��CO2��Ӧ������֪DΪNa2O2����Ϸ�Ӧ�ٽ���A�������Һ��Ӧ�����������ɵ�AΪAl����ΪNa2AlO2��E��F�ֱ�ΪO2��H2�е�һ�֣�BΪ�ǽ������ʣ�һ���Ǻ�ɫ��ĩ��������F��Ӧ����G��H����G����������Ʒ�Ӧ������֪BΪ̼��FΪO2��EΪH2��GΪCO2��HΪCO�����ݷ�Ӧ�ۿ���֪KΪNa2CO3����Һ��ΪNa2CO3��Һ���ɷ�Ӧ�ܿ�֪LΪAl��OH��3��
��1��������������֪��AΪAl��KΪNa2CO3��DΪNa2O2�������ʽΪ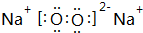 ��
��
�ʴ�Ϊ��Al��Na2CO3�� ��
��
��2����Ӧ���е�C��D���������÷�Ӧ���ܻ�ѧ����ʽ��4Al+2Na2O2+6H2O=4NaAlO2+6H2��+O2����
�ʴ�Ϊ��4Al+2Na2O2+6H2O=4NaAlO2+6H2��+O2����
��3����Ӧ���У���C��O2���ʵ���֮��Ϊ4��3����n��C����n��O��=2��3������ԭ���غ��֪[n��CO2��+n��CO��]��[2n��CO2��+n��CO��]=4��6��������n��CO2����n��CO��=1��1��
�ʴ�Ϊ��1��1��
��4����Ӧ�ܵ����ӷ���ʽΪ��2AlO2-+CO2+3H2O=2Al��OH��3��+CO32-��
�ʴ�Ϊ��2AlO2-+CO2+3H2O=2Al��OH��3��+CO32-��
��5����Al2O3��Fe2O3�Ļ����a g���ܽ��ڹ�����200mL c��H+��=0.1mol?L-1��������Һ�У�Ȼ�������м���NaOH��Һ�պ�ʹ�����������ʱ����ȥNaOH��Һ100mL�����ʱ��Һ������ΪNa2SO4����n��NaOH��=2n��Na2SO4��=2n��H2SO4��=n��H+��=0.2L��0.1mol?L-1=0.02mol����NaOH ��Һ��Ũ��Ϊ
=0.2mol/L��
�ʴ�Ϊ��0.2mol/L��
��1��������������֪��AΪAl��KΪNa2CO3��DΪNa2O2�������ʽΪ
 ��
���ʴ�Ϊ��Al��Na2CO3��
 ��
����2����Ӧ���е�C��D���������÷�Ӧ���ܻ�ѧ����ʽ��4Al+2Na2O2+6H2O=4NaAlO2+6H2��+O2����
�ʴ�Ϊ��4Al+2Na2O2+6H2O=4NaAlO2+6H2��+O2����
��3����Ӧ���У���C��O2���ʵ���֮��Ϊ4��3����n��C����n��O��=2��3������ԭ���غ��֪[n��CO2��+n��CO��]��[2n��CO2��+n��CO��]=4��6��������n��CO2����n��CO��=1��1��
�ʴ�Ϊ��1��1��
��4����Ӧ�ܵ����ӷ���ʽΪ��2AlO2-+CO2+3H2O=2Al��OH��3��+CO32-��
�ʴ�Ϊ��2AlO2-+CO2+3H2O=2Al��OH��3��+CO32-��
��5����Al2O3��Fe2O3�Ļ����a g���ܽ��ڹ�����200mL c��H+��=0.1mol?L-1��������Һ�У�Ȼ�������м���NaOH��Һ�պ�ʹ�����������ʱ����ȥNaOH��Һ100mL�����ʱ��Һ������ΪNa2SO4����n��NaOH��=2n��Na2SO4��=2n��H2SO4��=n��H+��=0.2L��0.1mol?L-1=0.02mol����NaOH ��Һ��Ũ��Ϊ
| 0.02mol |
| 0.1L |
�ʴ�Ϊ��0.2mol/L��
���������⿼��������ƶϣ���Ŀ�Ѷ��еȣ���ɫ��ζҺ��C�뵭��ɫ�Ĺ���D�ܷ�Ӧʽ�ƶϵ�ͻ�ƿڣ��ٽ��ת����ϵ��Ϣ�����ƶϣ�ע����Ŀ�л�ѧ������д�������غ㷨���еļ��㣮

��ϰ��ϵ�д�
�����Ŀ
����0.4mol?L-1HA��Һ��0.2mol?L-1NaOH��Һ�������������Һ�������й����۲���ȷ���ǣ�������
| A�����۸���Һ�����Ի��Ǽ��ԣ�����c��Na+��+c��H+��=c��A-��+c��OH-�� |
| B�����۸���Һ�����Ի��Ǽ��ԣ�����c��Na+��=c��A-��+c��HA�� |
| C��������Һ��HA������������A-ˮ������������c��A'��c��Na+����c��HA����c��H����c��OH- |
| D��������Һ��A-ˮ����������HA��������������c��A-����c��Na+����c��HA����c��OH-����c��H+����c��OH-�� |
��ʯī���缫�����1mol?L-1�������ʵ���Һ����Һ��pH���ֲ�����ǣ�������
| A��Na2SO4 |
| B��NaOH |
| C��HCl |
| D��NaCl |
�����£����и���������ָ����Һ��һ���ܴ���������ǣ�������
| A���μӼ��ȳʺ�ɫ����Һ��K+��Fe2+��NO3-��Cl- |
| B����ˮ�������c��H+��=10-12mol?L-1����Һ��Na+��K+��ClO-��SO32- |
| C��0.1mol?L-1NH3?H2O��Һ��Cu2+��NH4+��NO3-��SO42- |
| D����ɫ����Һ�壺K+��Na+��C6H5O-��HCO3- |
��NA��ʾ�����ӵ�����������������ȷ���ǣ�������
| A�����³�ѹ�£�36g C5H10������һ������7NA�����õ��Ӷ� |
| B��6g�����������ǵĻ������������������ȫȼ������0.2NA��ˮ���� |
| C��10mL5mol/L ��NH4��2SO4��Һ�к���NH4+��ĿΪ0.1NA�� |
| D��1mol FeCl3���ת��ΪFe��OH��3������γ�NA������ |
ʳ������ʳ�Σ��������к��е��������ƣ���ʹѪ�쵰����Fe2+ ת��Ϊ����Ѫ�쵰��ʹ�˷��������ж�������ά����C�ɽ��NaNO2 ���ж�������������ȷ���ǣ�������
| A��NaNO2�ǻ�ԭ�� |
| B��ά����C�ǻ�ԭ�� |
| C��ά����C�ܽ� NaNO2��ԭ |
| D��NaNO2������ |

