��Ŀ����
1��ij��ȤС��ͬѧ��ʵ�����ü���l-������ŨH2SO4���廯�ƻ����ķ������Ʊ�1-�嶡�飬���������ͼ��ʾ��ʵ��װ�ã����еļг�����û�л�������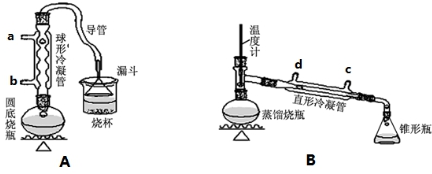
��ش��������⣺
��1��Aװ���У����ձ����Һ�浹��һ��©������Ŀ���Ǽȿ������ճ�֣��ֿ��Է�ֹ����������װ���ж��õ��������ܣ�Aװ������ˮ��b������ĸ���ţ����룬Bװ������ˮ��c������ĸ���ţ����룮
��2���Ʊ������У������Ũ�������ȱ������ϡ�ͣ���Ŀ����ab��������ĸ��
a�����ٸ�����ϩ���ѵ����� b������Br2������ c��ˮ�Ƿ�Ӧ�Ĵ���
��3����ͬѧ��ͨ����������Ǽ������ò������Ƿ��С�-CH2CH2CH2CH3������ȷ�����������Ƿ���ڶ��ѣ�CH3CH2CH2CH2OCH2CH2CH2CH3���������۸�ͬѧ��Ƶļ��������Ƿ������Ϊʲô���𣺲�����������1-�嶡��Ҳ����-CH2CH2CH2CH3��
��4��Ϊ�˽�һ���ᴿ1-�嶡�飬��С��ͬѧ�������л�����й��������±���
| ���� | �۵�/�� | �е�/�� |
| 1-���� | -89.5 | 117.3 |
| 1-�嶡�� | -112.4 | 101.6 |
| ���� | -95.3 | 142.4 |
| 1-��ϩ | -185.3 | -6.5 |
���� ��1�����ݷ�������ԭ������������в�������ͨˮʱ������Ч���Ϻã�
��2��l-������Ũ����������£��ɷ������Ӽ�ͷ�������ˮ��
��3�����ݺ�������ǵ����ã����з��ӽṹ�ͻ�ѧ��ɷ�����
��4������1-�嶡��ķе�Ϊ101.6����
��� �⣺��1����������©���л������ã������ܳ�ֱ������ҷ�ֹ�����������ܲ�������ͨˮ������Ч���Ϻã�����Aװ������ˮ��bͨ�룬Bװ������ˮ��cͨ�룬
�ʴ�Ϊ���ȿ������ճ�֣��ֿ��Է�ֹ������b��c��
��2��l-������Ũ����Ĵ������·�����������ˮ��ȡ��ϩ�����Ӽ���ˮ�õ��ѣ��������Ӽ���ˮ�γ���CH3CH2CH2CH2OCH2CH2CH2CH3����������ˮ��������ϩCH2=CHCH2CH3��ͬʱ�������ӱ�Ũ�����������嵥�ʣ������Ʊ������У������Ũ�������ȱ������ϡ�͵�Ŀ���ǣ����ٸ�����ϩ���ѵ����ɡ�����Br2�����ɣ���ab��ȷ��
�ʴ�Ϊ��ab��
��3������������������ʶԲ�ͬ�����ĺ��������������ԣ����з��ӽṹ�ͻ�ѧ��ɷ���������CH3CH2CH2CH2BrҲ����-CH2CH2CH2CH3�����Բ���ͨ�������������ȷ�����������Ƿ���ڶ��ѣ�CH3CH2CH2CH2OCH2CH2CH2CH3����
�ʴ�Ϊ��������������1-�嶡��Ҳ����-CH2CH2CH2CH3��
��4���ᴿ1-�嶡�飬�ռ��������Ϊ1-�嶡�飬�����뽫1-�嶡������������Һ�����������¶ȴ���е㣬1-�嶡��ķе�Ϊ101.6�棬��ʵ����ҪѸ�������¶���101.6���ռ��������
�ʴ�Ϊ��101.6�森
���� ���⿼�����Ʊ���������ƣ���Ŀ�Ѷ��еȣ���ȷʵ��Ŀ�ġ�ʵ��ԭ��Ϊ���ؼ���ע�������Ʊ�������������۵�ԭ������������ѧ���ķ�����������ѧʵ��������

| ʵ���� | NaOH��Һ�����mL�� | ��������������mL�� |
| 1 | 22.62 | 20.00 |
| 2 | 22.72 | 20.00 |
| 3 | 22.80 | 20.00 |
��2�������������ݣ��ɼ�����������Ũ��ԼΪ0.11mol/L��������λ��Ч���֣���
��3��������ʵ���У����в���������������ȷ������ɲⶨ���ƫ�ߵ���DF������ѡ�۷֣�
A���ζ��յ����ʱ���Ӷ���
B����ʽ�ζ���ʹ��ǰ��ˮϴ��δ�ô���������Һ��ϴ
C����ƿˮϴ��δ����
D������ǰNaOH�����л���Na2CO3����
E��NaOH����Һ����������е�CO2��Ӧ������Na2CO3
F����ʽ�ζ��ܼ��첿�������ݣ��ζ�����ʧ��

| A�� | ͭ�缫Ӧ��X������ | |
| B�� | H+�����ӽ���Ĥ���������ƶ� | |
| C�� | M�缫��ӦʽΪCO��NH2��2+H2O-6e-�TCO2��+N2��+6H+ | |
| D�� | ��N�缫����0.25 mol����ʱ�������������缫����32 g |
��Ӧԭ��������ͼ4��
��Ӧ�Լ������������������
| ���� | ��Է� ������ | ��״ | �۵� | �е� | �ܶ� | �ܽ�� | ||
| ˮ | �Ҵ� | ���� | ||||||
| �ױ� | 92 | ��ɫҺ����ȼ�ӷ� | -95 | 110.6 | 0.8669 | ���� | ���� | ���� |
| ������ | 122 | ��ɫƬ״����״���� | 122.4 | 248 | 1.2659 | �� | ���� | ���� |
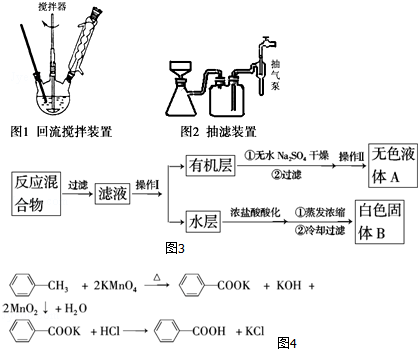
ʵ�鷽����һ�����ļױ���KMnO4��Һ����ͼ1װ���У���90��ʱ����Ӧһ��ʱ�䣬��ֹͣ��Ӧ������ͼ3���̷����������ͻ���δ��Ӧ�ļױ���
��1������������IJ�������Ϊ��Һ©�����ձ���������Ϊ����
��2�������Һ����ɫ��Ҫ�ȼ���������أ�Ȼ���ټ���Ũ�����ữ������������ص�Ŀ���dz�ȥδ��Ӧ�ĸ�����أ������������ữʱ�ᷢ�����ᱻ�������������������������
��3�����й�����������װ����ʹ����ȷ����ABD��
A�����˿��Լӿ�����ٶȣ��õ��ϸ���ij���
B����װ�綯������ʱ���������¶˲�����������ƿ�ס��¶ȼƵȽӴ�
C��ͼ1��������װ��Ӧ����ֱ�Ӽ��ȵķ���
D����������ˮ���������½��ϳ�
��4����ȥ�����ڱ������еļױ�Ӧ�ȼ���NaOH��Һ����Һ��ˮ���ټ���Ũ�����ữ��Ȼ����ˣ����T�ɵõ������ᣮ
��5�����Ȳⶨ����ȡ1.220g��Ʒ�����100mL��Һ��ȡ����25.00mL��Һ�����еζ�������KOH���ʵ���Ϊ2.4��10-3mol����Ʒ�б�������������Ϊ96%��
| A�� | ij�¶���ˮ�����ӻ�����ΪKw���ڸ��¶��£�a mol•L-1һԪ��HA��b mol•L-1һԪ��BOH�������ϣ���Һ�����ԣ�����Һ��c��H+��=$\sqrt{Kw}$ | |
| B�� | ��֪MgCO3��Ksp=6.82��10-6mol2•L-2�����ں��й���MgCO3��Na2CO3��Һ�У���c��Mg2+��=c��CO32-������c��Mg2+��•c��CO32-��=6.82��10-6mol2•L-2 | |
| C�� | Ksp��AB2��С��Ksp��CD����˵��AB2���ܽ��С��CD���ܽ�� | |
| D�� | NH4Cl��NH3•H2O�Ļ����Һ�У��ֱ����NH4+��ˮ��ƽ���NH3•H2O�ĵ���ƽ�⣬���߶ԶԷ���ƽ�ⶼ���˴ٽ����� |
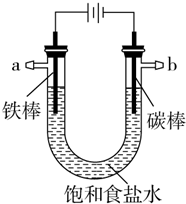 ��̼�����������������������NaCl��Һ����ʼԼ20���������������ɫ���ǣ�Ȼ��ʼ���ֻ���ɫ���ǣ�������ɽ϶�ĺ��ɫ����������˵����ȷ���ǣ�������
��̼�����������������������NaCl��Һ����ʼԼ20���������������ɫ���ǣ�Ȼ��ʼ���ֻ���ɫ���ǣ�������ɽ϶�ĺ��ɫ����������˵����ȷ���ǣ�������| A�� | �����缫��Ӧʽ��Fe-3e-�TFe3+ | |
| B�� | ˮ������������ɫ������ΪNaCl���� | |
| C�� | ����ɫ������Fe��OH��2 | |
| D�� | ���ŵ��Ľ��У���Һ�������Ա�ɼ��� |
I����ʵ������ģ�ҵ�������̿���Ҫ�ɷ�ΪMnO2��������SiO2��Fe2O3��A12O3�ȣ��Ʊ������̵����ʣ����������ͼ��
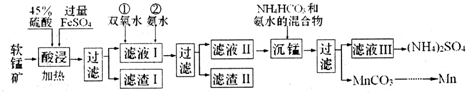
��֪��̼�����ڿ����и��¼��ȹ������ΪMn2O3���������������Ksp������ֵ�������
| ���� | Mn��OH��2 | Fe��OH��2 | Fe��OH��3 | Al��OH��3 |
| Ksp | 10-13 | 10-17 | 10-39 | 10-33 |
��2������ҺI �еμ�˫��ˮ��Ŀ���ǽ�Fe2+����ΪFe3+������ת��ΪFe��OH��3��ȥ�������ҺI ��c��Mn2+��=0.1mol•L-1��Ϊ��֤�μӰ�ˮ������Mn��OH��2��Ӧ������ҺpH���Ϊ8��
��3�������̡������е����ӷ���ʽΪMn2++HCO3-+NH3•H2O=MnCO3��+NH4++H2O��
��4����̼���̺ͽ�����Ϊԭ�Ͽ����Ʊ����̣�д���Ʊ����������һ����Ӧ�Ļ�ѧ����ʽMn2O3+2Al$\frac{\underline{\;����\;}}{\;}$Al2O3+2Mn��
��ѧ�ҷ�����NH3ȼ�ϵ�أ���KOHΪ����ʣ�ԭ����ͼ��ʾ��
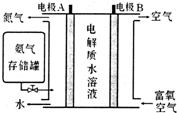
��5����ȼ�ϵ�صĸ�����ӦʽΪ2NH3+6OH--6e-=N2+6H2O��
��6���Ըõ��Ϊ��Դ����ʯī�缫�������أ�K2MnO4����Һ��ȡ������أ�ͨ��10���ӣ�����ǿ��Ϊ0.5A�������Ƶø������0.49g����֪1mol������������Ϊ96500C����
| A�� | 1mol����1molCl2��ַ�Ӧ��ת�Ƶ�����Ϊ3NA | |
| B�� | ���³�ѹ�£�2gD216O �к������������������������������ | |
| C�� | 14g ����ʽΪCnH2n �ģ�n��2�������к��е�C=C ����Ŀһ��Ϊ$\frac{{N}_{A}}{n}$ | |
| D�� | �ں�Al3+����ΪNA��AlCl3��Һ�У�Cl-����Ϊ3NA |
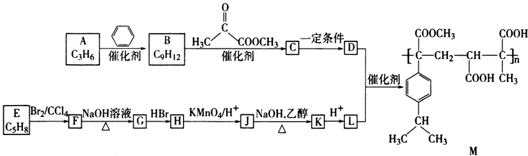
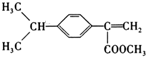 ��
��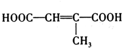 ��д�ṹ��ʽ����
��д�ṹ��ʽ����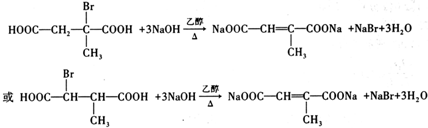 ��
�� ��д�ṹ��ʽ����
��д�ṹ��ʽ����