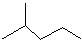
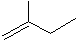
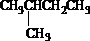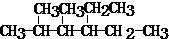��Ŀ����
��1��12.4g Na2X����0.4mol Na+��Na2X��Ħ������Ϊ ������Է�������Ϊ ��X�����ԭ������Ϊ �������ʵĻ�ѧʽΪ ��
��2�� mol H2O�������е���ԭ������2mol NH3�к��е���ԭ������ͬ��
��3��a��Xԭ�ӵ�������Ϊb g����X�����ԭ�������ɱ�ʾΪ ��
��4��20g A���ʺ�14g B������ȫ��Ӧ������8.8g C���ʡ�3.6g D���ʺ�0.2mol E���ʣ���E���ʵ�Ħ������Ϊ ��
��2��
��3��a��Xԭ�ӵ�������Ϊb g����X�����ԭ�������ɱ�ʾΪ
��4��20g A���ʺ�14g B������ȫ��Ӧ������8.8g C���ʡ�3.6g D���ʺ�0.2mol E���ʣ���E���ʵ�Ħ������Ϊ
���㣺�����ӵ����ɼ�����
ר�⣺������
����������n=
=
=
������ʵĹ��ɼ��㣮
| m |
| M |
| V |
| Vm |
| N |
| NA |
���
�⣺��1��12.4��Na2X�к���0.4molNa+��Na2X�����ʵ���Ϊ��n��Na2X��=
n��Na+��=0.4mol��
=0.2mol��
Na2X��Ħ������Ϊ��M��Na2X��=
=62g/mol��
Ħ����������ֵ�ϵ�������Է�����������Na2X�����ԭ����Ϊ62��
��ԭ�ӵ����ԭ��������23������X�����ԭ��������62-23��2=16��
XΪ��ԭ�ӣ������ʵĻ�ѧʽΪNa2O��
�ʴ�Ϊ��62g/mol��62��16��Na2O��
��2��2mol NH3�к��е���ԭ��Ϊ6mol����ˮ�����ʵ���ӦΪ3mol���ʴ�Ϊ��3��
��3��a��Xԭ�ӵ�������Ϊb g����NA��ԭ�ӵ�����Ϊ
NAg���������ԭ������Ϊ���ʴ�Ϊ��
NA��
��4����ѧ��Ӧ��ѭ�����غ㶨�ɣ���Ӧ��m��A��+m��B��=m��C��+m��D��+m��E����
��m��E��=20g+14g-8.8g-3.6g=21.6g��
M��E��=
=108g/mol��
�ʴ�Ϊ��108g/mol��
| 1 |
| 2 |
| 1 |
| 2 |
Na2X��Ħ������Ϊ��M��Na2X��=
| 12.4g |
| 0.2mol |
Ħ����������ֵ�ϵ�������Է�����������Na2X�����ԭ����Ϊ62��
��ԭ�ӵ����ԭ��������23������X�����ԭ��������62-23��2=16��
XΪ��ԭ�ӣ������ʵĻ�ѧʽΪNa2O��
�ʴ�Ϊ��62g/mol��62��16��Na2O��
��2��2mol NH3�к��е���ԭ��Ϊ6mol����ˮ�����ʵ���ӦΪ3mol���ʴ�Ϊ��3��
��3��a��Xԭ�ӵ�������Ϊb g����NA��ԭ�ӵ�����Ϊ
| b |
| a |
| b |
| a |
��4����ѧ��Ӧ��ѭ�����غ㶨�ɣ���Ӧ��m��A��+m��B��=m��C��+m��D��+m��E����
��m��E��=20g+14g-8.8g-3.6g=21.6g��
M��E��=
| 21.6g |
| 0.2mol |
�ʴ�Ϊ��108g/mol��
������������Ҫ�������ʵ����ļ��㣬��Ŀ�ѶȲ���ע�������غ㶨�ɡ�Ħ����������ֵ�ϵ�������Է��������ȸ�������ã�������ض�ѧ�������������ͽ��ⷽ����ָ����ѵ����

��ϰ��ϵ�д�
�����Ŀ
��NA���������ӵ�����������˵����ȷ���ǣ�������
| A��23g�����Ʊ�Ϊ������ʱ�õ��ĵ�����ΪNA |
| B����״���£�18gˮ�����ĵ�����ĿΪ18NA |
| C����״���£�22.4 L����������ԭ����ĿΪ2NA |
| D�����³�ѹ�£�2.8g N2��CO�Ļ����������ԭ����Ϊ0.2NA |
����˵������ȷ���ǣ�������
| A��2 mol CH4 ��������O2��Ħ����������32 g |
| B��1 mol�κ������ж�������ͬ��ԭ���� |
| C��0.5 mol NaClԼ����6.02��1023������ |
| D��1 mol?L-1 KCl��Һ�к�������1 mol |
��NA��ʾ�����ӵ�������ֵ��������������ȷ���ǣ�������
| A�����³�ѹ��42g ��ϩ�Ͷ�ϩ��������У����Լ���Ϊ6NA |
| B��100 mL 0.1 mol?L-1 Na2CO3��Һ�У�����������0.03NA |
| C����״���£�11.2L��O2��NO�Ļ���ﺬ�еķ�����ԼΪ0.5��6.02��1023 |
| D��6.4g SO2������������Ӧ����SO3��ת�Ƶ�����Ϊ0.2NA |