��Ŀ����
2���о�̼���仯������ת������Դ�ij�����á���̼����������Ҫ�����ã���1����֪����CH4��g��+H2O��g��?CO��g��+3H2��g����H1=+206.1kJ•mol-1
��2H2��g��+CO��g��?CH3OH��l����H2=-128.3kJ•mol-1
��2H2��g��+O2��g��?2H2O ��g����H3=-483.6kJ•mol-1
25��ʱ���ں��ʵĴ��������£����ü��������һ���ϳ�Һ̬�״����Ȼ�ѧ����ʽΪCH4��g��+$\frac{1}{2}$O2��g��?CH3OH��l����H1=-164.0kJ•mol-1��
��2�����÷�Ӧ�����Ʊ�������Ϊ��̽���¶ȡ�ѹǿ�Է�Ӧ�����ʡ�ת���ʵ�Ӱ�죬ijͬѧ�������������Ա�ʵ�飨�¶�Ϊ400���500�棬ѹǿΪ101kPa��404kPa����
| ʵ����� | �¶ȡ� | ѹǿ/kPa | CH4��ʼŨ��/mol•L-1 | H2O��ʼŨ��/mol•L-1 |
| 1 | 400 | p | 3.0 | 7.0 |
| 2 | t | 101 | 3.0 | 7.0 |
| 3 | 400 | 101 | 3.0 | 7.0 |
�ڽ������ʵ�����CH4��ˮ��������1L�����ܱ������У�����������Ӧ����400���´ﵽƽ�⣬ƽ�ⳣ��K=27����ʱ������CO���ʵ���Ϊ0.10mol����CH4��ת����Ϊ90.9%��
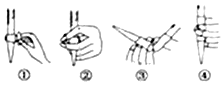
��3����ѧ�������CO2�� ȡ C��̫���ܹ�����ͼ1��ʾ��
2������ϵͳ�������ķ�Ӧ��n��FeO����n��CO2��=6��1����Fe xOy�Ļ�ѧʽΪFe3O4��
�ڡ��ȷֽ�ϵͳ����ÿ�ֽ�l mol Fe xOy��ת�Ƶ��ӵ����ʵ���Ϊ2mol��
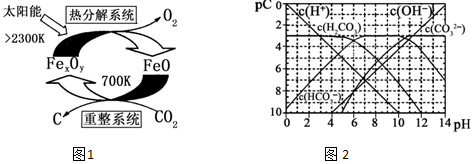
��4��pC����pH����ָ��ϡ��Һ�е�����Ũ�ȵij��ø�����ֵ����ij��Һ�����ʵ�Ũ��Ϊ1��10-3mol•L-1�������Һ�����ʵ�pC=-lg��1��10-3��=3����ͼ2Ϊ25��ʱH2CO3��Һ��pC-pHͼ����ش��������� ��������Ũ��С��10-5mol/L������Ϊ�����Ӳ����ڣ���
����ͬһ��Һ�У�H2CO3��HCO3-��CO32-���ܣ���ܡ����ܡ����������森
����H2CO3һ������ƽ�ⳣ������ֵKa1=1��10-6��
������ѪҺ����Ҫͨ��̼�����λ�����ϵ$\frac{c��{H}_{2}C{O}_{3}��}{c��HC{O}_{3}^{-}��}$���Ե����������ά��pH=7.4���������������ѪҺ��ʱ��ѪҺ������ϵ�е�$\frac{c��{H}^{+}��}{c��{H}_{2}C{O}_{3}��}$���ս�A��
A�����B����СC����������D�����жϣ�
���� ��1�����ݸ�˹���ɵâ�+��+$\frac{1}{2}$���ۼ��������һ���ϳ�Һ̬�״����Ȼ�ѧ����ʽ��
��2���ٷ�Ӧ�����ȷ�Ӧ���¶����ߣ�������У�k���ڸ��ݻ�ѧƽ�ⳣ��k��������ʽ��ʾƽ��ʱ��Ũ�ȣ�����ת���ʣ�
��3������ʾ��ͼ��֪������ϵͳ��CO2��FeO��Ӧ����FexOy��C������ԭ���غ�ȷ��FexOy�Ļ�ѧʽ��
���ȷֽ�ϵͳ����FexOy�ֽ�ΪFeO������������FeԪ�ػ��ϼ۱�ۼ���ת�Ƶ��ӣ�
��4���ٸ������Ӵ�����Һ��������жϣ�
����ͼ���֪��pH=6ʱ��pC��H2CO3��=pC��HCO3-�������Ka1��H2CO3��=$\frac{c��{H}^{+}��•c��HC{{O}_{3}}^{-}��}{c��{H}_{2}C{O}_{3}��}$���㣻
�ۡ�������Һ���м���������ǿ���ǿ���Һ��pH�仯���������ʱ��̼��������ӷ�Ӧ��
��� �⣺��1�����Ȼ�ѧ����ʽ��
��CH4��g��+H2O��g��?CO��g��+3H2��g����H1=+206.1kJ•mol-1
��2H2��g��+CO��g��?CH3OH��l����H2=-128.3kJ•mol-1
��2H2��g��+O2��g��?2H2O ��g����H3=-483.6kJ•mol-1
���ݸ�˹���ɣ���+��+$\frac{1}{2}$���۵�CH4��g��+$\frac{1}{2}$O2 ��g��?CH3OH��l����H1=-164.0kJ•mol-1��
�ʴ�Ϊ��CH4��g��+$\frac{1}{2}$O2 ��g��?CH3OH��l����H1=-164.0kJ•mol-1
��2���ٷ�Ӧһ�����ȷ�Ӧ���¶����ߣ���ѧƽ�������ƶ���kֵ���ʵ��2���¶ȸ���ʵ��3������k2��k3
�ʴ�Ϊ����
����CH4��H2O����ʼ��Ϊx����������ʽ��
CH4��g��+H2O��g��?CO��g��+3H2��g��
��ʼ����x x 0 0
ת������0.1 0.1 0.1 0.3
ƽ������x-0.1 x-0.1 0.1 0.3
��ƽ�ⳣ������ʽk=$\frac{c��CO��•c^3����{H}_{2}��}{c��C{H}_{4}��•c��{H}_{2}O��}$=$\frac{0.1mol/L•��0.3mol/L��^3}{��x-0.1��mol/L•��x-0.1��mol/L}$=27�����x=0.11����ƽ��ʱ�����ת����Ϊ��$\frac{0.1mol}{0.11mol}��100%$=90.9%��
�ʴ�Ϊ��$\frac{10}{11}$��90.9%��90.91%��$\frac{10}{11}$��100%��
��3��������ʾ��ͼ��֪������ϵͳ��CO2��FeO��Ӧ����FexOy��C�������ķ�Ӧ��n��FeO����n��CO2��=6��1������Feԭ�ӡ�Oԭ���غ��֪x��y=6����6+2��=3��4����FexOy�Ļ�ѧʽΪFe3O4��
�ʴ�Ϊ��Fe 3O4
���ȷֽ�ϵͳ����Fe3O4�ֽ�ΪFeO���������ֽ�l mol Fe3O4ת�Ƶ��ӵ����ʵ���Ϊ$1mol��3����\frac{8}{3}-2��$=2mol��
�ʴ�Ϊ��2mol
��4����̼����ڵ���Һ�����Խ�ǿ��̼������Ӵ��ڵ���Һ�м��Խ�ǿ������̼������Ӻ�̼��ܴ������森
�ʴ�Ϊ�����ܣ�
����ͼ���֪��pH=6ʱ��pC��H2CO3��=pC��HCO3-�������Ka1��H2CO3��=$\frac{c��{H}^{+}��•c��HC{{O}_{3}}^{-}��}{c��{H}_{2}C{O}_{3}��}$=$\frac{1{0}^{-6}•c��HC{{O}_{3}}^{-��}}{c��{H}_{2}C{O}_{3}��}$=1��10-6��
�ʴ�Ϊ��1��10-6
��������Ũ������ƽ�������ƶ��ų�CO2��̼��Ũ�Ȼ������䣬��c��H+��/c��H2CO3�����ս����
�ʴ�Ϊ��A
���� ���⿼���˸�˹���ɵ�Ӧ�ã����������ˮ��Һ�еĵ���ƽ�⣬���ʵ�������ؼ��㣬��ѧƽ��ļ��㼰Ӱ�����أ��ۺϿ��飬�ѶȽϴ�

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�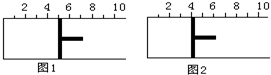 ��ͼ��ʾ��һ�������½�lmolA�����3molB�������һ������ɱ���ܱ������У��ɻ���������λ��ͼ1��ʾ���ں��º�ѹ�·���A��g��+3B��g��?2C��g��������Ӧ�ﵽƽ��ʱ������λ����ͼ2��ʾ����ƽ��ʱA��ת����Ϊ��������
��ͼ��ʾ��һ�������½�lmolA�����3molB�������һ������ɱ���ܱ������У��ɻ���������λ��ͼ1��ʾ���ں��º�ѹ�·���A��g��+3B��g��?2C��g��������Ӧ�ﵽƽ��ʱ������λ����ͼ2��ʾ����ƽ��ʱA��ת����Ϊ��������| A�� | 20% | B�� | 40% | C�� | 50% | D�� | ��ȷ�� |
| A�� | 2���SO2������O2��Ӧ��һ����������2���SO3 | |
| B�� | �����������䣬����ѹǿ������Ӧ���������淴Ӧ����Ҳ���� | |
| C�� | ����2molSO2��1molO2�ų��������Ǽ���1molSO2��0.5molO2�ų�������2�� | |
| D�� | ƽ����ټ���1molSO3��SO2��ת�������� |
| A�� | ���ᡢ���ʯ�� | B�� | ���ᡢ�ռС�մ� | ||
| C�� | ̼�ᡢ�Ҵ��������� | D�� | ���ᡢ��ʯ�ҡ������� |
| A�� | ������Һ���У�pN=7 | |
| B�� | ������Һ���У�c��NH4+��•c��NH2-����1.0��10-14 | |
| C�� | 1 L�ܽ���0.001 mol NH4C1��Һ����Һ����pN=3 | |
| D�� | 1 L�ܽ���0.001 mol NaNH2��Һ����Һ����pN=11 |
��2FeCl2+Cl2�T2FeCl3
��2FeCl3+2HI�T2FeCl2+I2+2HCl
��I2+Na2S�T2NaI+S
�����й����ʵ������Ե�ǿ������˳������ȷ���ǣ�������
| A�� | Cl2��Fe3+��I2��S | B�� | Cl2��I2��Fe3+��S | C�� | Cl2��Fe3+��S��I2 | D�� | Fe3+��I2��C12��S |