��Ŀ����
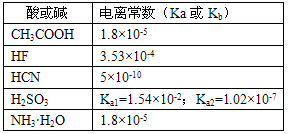
���볣�����Ա�ʾ������ʵĵ���������±��dz����¼�������ĵ���ƽ�ⳣ����Ka��������ĵ���ƽ�ⳣ����Kb��
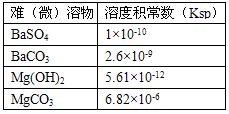
�ܶȻ��������Ա�ʾ�ѣ���������ܽ��������±��dz����¼����ѣ���������ܶȻ�������Ksp��
����˵����ȷ����
[ ]
A�����ԣ�HF >CH3COOH >H2SO3>HCN
B�������ܶȻ�������Ksp������ͬѧ��Ϊ��NaCl��Һ��������MgCl2������Ҫ���Թ�����NaOH��Һ��������Na2CO3��Һ����ͬѧ��Ϊ��ʹ��BaSO4����Һ�мӹ����Ĵ�����Һ��BaSO4Ҳ����ת��ΪBaCO3
C��Ũ����ͬ��NaCN��Һ��HCN����Һ�������ϣ���pH>7����Һ�и�����Ũ�ȴ�СΪ��c(OH��) > c(H+)> c(Na+)> c(CN��)
D��CH3COONH4ˮ��Һ�����ԣ�����Ϊ���ݵ��볣����CH3COO�����H+��NH4+���OH������������ʵij̶�һ��������ˮ��Һ�е�H+��OH��Ũ�����
B�������ܶȻ�������Ksp������ͬѧ��Ϊ��NaCl��Һ��������MgCl2������Ҫ���Թ�����NaOH��Һ��������Na2CO3��Һ����ͬѧ��Ϊ��ʹ��BaSO4����Һ�мӹ����Ĵ�����Һ��BaSO4Ҳ����ת��ΪBaCO3
C��Ũ����ͬ��NaCN��Һ��HCN����Һ�������ϣ���pH>7����Һ�и�����Ũ�ȴ�СΪ��c(OH��) > c(H+)> c(Na+)> c(CN��)
D��CH3COONH4ˮ��Һ�����ԣ�����Ϊ���ݵ��볣����CH3COO�����H+��NH4+���OH������������ʵij̶�һ��������ˮ��Һ�е�H+��OH��Ũ�����
D

��ϰ��ϵ�д�
�����Ŀ
