��Ŀ����
10�����������[��NH4��2SO4•FeSO4•6H2O]�׳�Ī���Σ�������ˮ����100�桫110��ʱ�ֽ⣮�ڶ��������г������궨�ظ���ء�������ص���Һ�ı����ʣ�������ұ�𡢵�Ƶȣ���ѧ����С���о�Ī���ξ���ǿ��ʱ�ķֽ���
��1������ͬѧ������ͼ1��ʾ��װ�ý����о���װ��C�пɹ۲쵽����������Һ��죬�ɴ˿�֪�ֽ��������NH3��
��2������ͬѧ��ΪĪ���ξ���ֽ�IJ����л�����SO3��g����SO2��g����N2��Ϊ������֤��ѡ�ü���ʵ���е�װ��A����ͼ2��ʾ�IJ���װ�ý���ʵ�飮
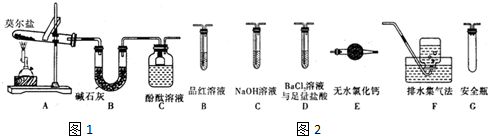
������ͬѧ��ʵ���У�װ���������ӵĺ���˳��ΪA��G��D��B��C��F��
��װ��D��������������������հ���������Һ�ữ���ų�SO2�ĸ��ţ�����SO3��ʵ��������D�г��ְ�ɫ������
��Ϊ����������林��ȣ���ȡm gĪ������Ʒ�����500ml��Һ���ס��ҡ�����λͬѧ�������������ʵ�鷽������ش�
������ȡ25.00mL�����������Һ��0.1000mol•L-1������KMnO4��Һ�����ν��еζ���
�ҷ�����ȡ25.00mL�����������Һ��������ʵ�飮
����Һ$\stackrel{����BaCl_{2}��Һ}{��}$ $\stackrel{����}{��}$ $\stackrel{ϴ��}{��}$ $\stackrel{����}{��}$ $\stackrel{����}{��}$wg����
����������ͨ��NH${\;}_{4}^{+}$�ⶨ��ʵ�����ͼ��ͼ3��ʾ��ȡ25.00mL�����������Һ���и�ʵ�飮
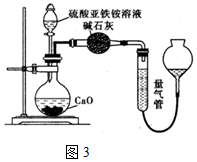
��1�������е����ӷ���ʽΪMnO4-+5Fe2++8H+=Mn2++5Fe3++4H2O����ʵ���������ȷ������ⶨ�������С���ҷ���������ԭ��ΪFe2+�ѱ�����������������֤�Ʋ�ķ���Ϊȡ���������������Һ����������KSCN��Һ������Һ��ΪѪ��ɫ��˵��Fe2+�ѱ���������������
��2���ҷ����г����Ƿ�ϴ�Ӹɾ��ļ��鷽����ȡ���һ��ϴ�ӵ���Һ����AgNO3��Һ����û�г�����˵����ϴ�ɾ���
��3����������������������Լ���c
a��ˮ b������NaHCO3��Һ c��CCl4
��4�������NH3��������Ϊ��״���£�ΪV L������������林���Ϊ$\frac{392V��20}{44.8m}��100%$��
���� ��1��Ī����[��NH4��2Fe��SO4��2•6H2O]���ȷֽ⣬�ֽ�ʱ����������Ͷ�����������������������壬����װ��ͼ��֪����ʯ�ҿ��������������壬����������̪��Һ����ɫ��
��2����Ҫ��������SO3��g����SO2��g����N2���ڼ���ʵ���е�װ��A�������������ȫƿ��ͨ���Ȼ�����Һ����SO3����ͨ��Ʒ����Һ����SO2����Ũ�������Ƴ�ȥ������������ˮ�������ռ��������ݴ˴��⣻
�����ڲ������������а����������Ȼ�����Һ�м������������ᣬ�������հ�������ֹ���������ᱵ�������ų�SO2�ĸ��ţ�SO3ͨ���Ȼ�����Һ�п��Բ������ᱵ����������������ʹƷ����ɫ��
��1�������еķ�Ӧ������KMnO4��Һ�������������������������������Ӿ��л�ԭ�ԣ��ױ������������������ӵ��������������ӣ�
��2���ҷ����г����Ƿ�ϴ�Ӹɾ����������ʵ�����ϴ��Һ���Ƿ��������Ӽ������ϴ���Ƿ�ɾ���
��3�����ݰ������ܽ��Ժ�װ���ص������
��4�����ݰ�����������㰱�������ʵ���������N�غ�������������淋�������������������������
��� �⣺��1��Ī����[��NH4��2Fe��SO4��2•6H2O]���ȷֽ⣬�ֽ�ʱ����������Ͷ�����������������������壬����װ��ͼ��֪����ʯ�ҿ��������������壬����������̪��Һ����ɫ������װ��C �пɹ۲쵽����������Һ��죬�ɴ˿�֪Ī���ξ���ֽ�IJ������� NH3��װ��B����Ҫ������ ���շֽ�������������壬
�ʴ�Ϊ����Һ��죻NH3��
��2����Ҫ��������SO3��g����SO2��g����N2���ڼ���ʵ���е�װ��A�������������ȫƿ��ͨ���Ȼ�����Һ����SO3����ͨ��Ʒ����Һ����SO2����Ũ�������Ƴ�ȥ������������ˮ�������ռ�����������װ���������ӵĺ���˳��ΪA��G��D��B��C��F��
�ʴ�Ϊ��G��D��B��C��F��
�����ڲ������������а����������Ȼ�����Һ�м������������ᣬ�������հ�������ֹ���������ᱵ�������ų�SO2�ĸ��ţ�SO3ͨ���Ȼ�����Һ�п��Բ������ᱵ����������������ʹƷ����ɫ������װ��D����������������� ���հ���������Һ�ữ���ų�SO2�ĸ��ţ�����SO3��ʵ�������� D���а�ɫ������
�ʴ�Ϊ�����հ���������Һ�ữ���ų�SO2�ĸ��ţ�D���а�ɫ������
��1�����������Һ���������ԣ���������������Ϊ�����ӣ���Ӧ�����ӷ���ʽΪ��MnO4-+5Fe2++8H+=Mn2++5Fe3++4H2O���������Ӿ��л�ԭ�ԣ��ױ������������ӣ��ʲ�����������Ũ��ƫС���ɼ����������ӵ��������������ӣ��������Ϊ��ȡ���������������Һ����������KSCN��Һ������Һ��ΪѪ��ɫ��˵��Fe2+�ѱ���������������
�ʴ�Ϊ��MnO4-+5Fe2++8H+=Mn2++5Fe3++4H2O��Fe2+�ѱ���������������ȡ���������������Һ����������KSCN��Һ������Һ��ΪѪ��ɫ��˵��Fe2+�ѱ���������������
��2���ҷ����г����Ƿ�ϴ�Ӹɾ����������ʵ�����ϴ��Һ���Ƿ��������Ӽ������ϴ���Ƿ�ɾ������岽��Ϊ��ȡ���һ��ϴ�ӵ���Һ����AgNO3��Һ����û�г�����˵����ϴ�ɾ���
�ʴ�Ϊ��ȡ���һ��ϴ�ӵ���Һ����AgNO3��Һ����û�г�����˵����ϴ�ɾ���
��3����װ���е�����Һ�����ϣ�������Һ���ռ�����Ҫ����������Һ��Ӧ�����ܽⰱ��������������ˮ�ͱ���̼������Һ�����������Ȼ�̼�����������Ȼ�̼���ռ����ʴ�Ϊ��c��
��4��VL���������ʵ���Ϊ��$\frac{VL}{22.4L/mol}$=$\frac{V}{22.4}$mol��
m g�����������Ʒ�к�N�����ʵ���Ϊ$\frac{500mL}{20mL}$��$\frac{V}{22.4}$mol=$\frac{25V}{22.4}$mol��
��������淋Ĵ���Ϊ��$\frac{\frac{25V}{22.4}��\frac{1}{2}mol��392g/mol}{mg}$��100%=$\frac{392V��20}{44.8m}��100%$��
�ʴ�Ϊ��$\frac{392V��20}{44.8m}��100%$��
���� ���⿼������ʵ�鷽����ƣ����ؿ���ѧ��֪ʶ�ۺ�Ӧ�á�ʵ���������������ʵ�鷽������������ۺ��Խ�ǿ���ѵ�������ʵ���Ⱥ�˳����ʵ��Ŀ�ļ����ʵ����ʽ�������˳��ע��Ҫ�ų��������ظ��ţ���Ŀ�Ѷ��еȣ�

 ��ʦ����ָ���ο�ʱϵ�д�
��ʦ����ָ���ο�ʱϵ�д�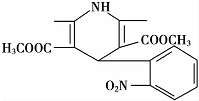
| A�� | �û�����ķ���ʽΪC17H17N2O6 | |
| B�� | �û�����ķ����к������ֲ�ͬ�Ĺ����� | |
| C�� | �û������������������ӳɷ�Ӧ����������������Ȼ�̼��Һ��Ӧ | |
| D�� | �û������ܷ���ˮ�ⷴӦ���ۺϷ�Ӧ��ȡ����Ӧ |
 ��ͼ��һ��﮷��������ȵ��װ�ã�����ܷ�ӦΪxLi+LiV3O8=Li1+xV3O8������ʱ������������������ط�Ӧʹ�������ڻ�����֪��Li�۵�181�棬LiCl-KCl�������۵�352�森����˵����ȷ���ǣ�������
��ͼ��һ��﮷��������ȵ��װ�ã�����ܷ�ӦΪxLi+LiV3O8=Li1+xV3O8������ʱ������������������ط�Ӧʹ�������ڻ�����֪��Li�۵�181�棬LiCl-KCl�������۵�352�森����˵����ȷ���ǣ�������| A�� | �������̵�����ת��ֻ�漰��ѧ��ת��Ϊ���� | |
| B�� | �ŵ�ʱ������ӦΪ��Li1+xV3O8-xe?�TLiV3O8+xLi+ | |
| C�� | �ŵ�ʱCl?����LiV3O8�缫 | |
| D�� | Li-Si�Ͻ��۵����352�� |
| A�� | Y����̬�⻯��ķе���ͬ��������ߣ���������̬�⻯�����ȶ� | |
| B�� | W����������X��Z�γɵĻ�������������ѧ����ͬ | |
| C�� | �����Ӱ뾶��W��Z��Y | |
| D�� | ���ʵ������ԣ�Y��W |
| A�� | ������м������������������Һ�����ӷ���ʽΪAl3++2SO42-+2Ba2++4OH-�T2BaSO4��+AlO2-+2H2O | |
| B�� | 2Cu��IO3��2+24KI+12H2SO4�T2CuI��+13I2+12K2SO4+12H2O�У���1mol�������μӷ�Ӧʱ�������������ʵ����ʵ���Ϊ11mol | |
| C�� | ǿ����Һ�У��ô���������Fe��OH��2��Ӧ��ʪ���Ʊ��������Ƶ�����Ҫ���̣��ɱ�ʾΪ��3ClO-+2Fe��OH��2�T2FeO42-+3Cl-+H2O+4H+ | |
| D�� | ������SO2ͨ��������ˮ�е����ӷ���ʽΪ��SO2+2NH3•H2O�T2NH4++SO32- |
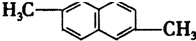 �����ϵ�����ȡ����ͬ���칹���У�������
�����ϵ�����ȡ����ͬ���칹���У�������| A�� | 7�� | B�� | 8�� | C�� | 9�� | D�� | 10�� |
| A�� | 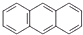 ���죩�� ���죩��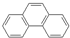 ���ƣ�����Ϊͬ���칹�壬��1H-NMR�ף��˴Ź������ף���ʾ���߾������ֲ�ͬ����ԭ�ӣ��ұ�����Ϊ2��2��1���ʲ�����1H-NMR�������� ���ƣ�����Ϊͬ���칹�壬��1H-NMR�ף��˴Ź������ף���ʾ���߾������ֲ�ͬ����ԭ�ӣ��ұ�����Ϊ2��2��1���ʲ�����1H-NMR�������� | |
| B�� | һ�㲻��ʹ�ù����ϴ�Ӽ�ϴ����֯Ʒ��˿֯Ʒ������ʹ�ù����ϴ�Ӽ�ϴ�Ӱ��ڣ��۰�����������ά�������ڣ��۱�ϩ�棩�Ⱥϳ���ά��Ʒ | |
| C�� | ���ͪ������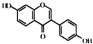 �ͷ������� �ͷ������� 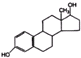 �Ǿ����������ԵĴƼ��أ����ǵĹ�ͬ���Ǿ������Ƶ��ʺ�̼���Ʒ�Ӧ���Ҿ������־���������ԵĹ����ţ������д��ǻ��ͷ��ǻ���Ϊ���ʲ�ͬ�Ĺ����ţ� �Ǿ����������ԵĴƼ��أ����ǵĹ�ͬ���Ǿ������Ƶ��ʺ�̼���Ʒ�Ӧ���Ҿ������־���������ԵĹ����ţ������д��ǻ��ͷ��ǻ���Ϊ���ʲ�ͬ�Ĺ����ţ� | |
| D�� | һ�������½���������ȵ�CH3OH��CH3COOH������HCOOH��HOCOOCH2OH���������������ȫȼ�գ�ȼ�պ��ȫ������ͨ�������������Ʋ㣬�������Ʋ����ӵ�����֮��Ϊ23��15 |
����һ������100mL 0.1000mol/LNaOH����Һ����
�������ȡ20.00mL����ϡ���������ƿ�У����μ�2��3�η�̪��Һ��ָʾ�������Լ����Ƶı�NaOH��Һ���еζ����ظ������ζ�����2��3�Σ���¼�������£�
| �ζ����� | ���������mL�� | ���ռ������mL�� | |
| �ζ�ǰ���� | �ζ������ | ||
| ��һ�� | 20.00 | 0.50 | 20.40 |
| �ڶ��� | 20.00 | 1.00 | 24.10 |
| ������ | 20.00 | 4.00 | 24.10 |
A����Һ�� B����Ͳ C����ʽ�ζ��� D����ʽ�ζ���
��2���ζ��ﵽ�յ���������һ������������Һ���룬��Һ����ɫǡ�ñ��dz��ɫ���Ұ�����ڲ���ɫ��
��3�������������ݣ��ɼ�����������Ũ��Ϊ0.1000mol/L��
��4����ȥ��ʽ�ζ��������ݵķ���Ӧ������ͼ�ı�������Ȼ�����ἷѹ������ʹ���첿�ֳ�����Һ��
��5��������ʵ���У����в���������������ȷ������ɲⶨ���ƫ�ߵ���ADEF����ѡ�۷֣���
A�����Ʊ���Һ����ʱ����ˮ�����̶�
B����ƿˮϴ��ֱ��װ����Һ
C����ʽ�ζ���ˮϴ��δ�ô���ϡ������Һ��ϴ
D���ζ������յ�ʱ�����Ӷ����ζ��ܶ���
E����ʽ�ζ��ܼ��첿�������ݣ��ζ�����ʧ
F������ǰNaOH�����л���Na2CO3���壮
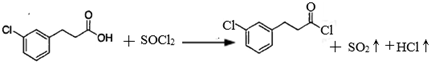 ��
��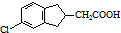 �ĺϳ�·������ͼ��ע����Ӧ���������ϳ�·������ͼʾ�����£�
�ĺϳ�·������ͼ��ע����Ӧ���������ϳ�·������ͼʾ�����£�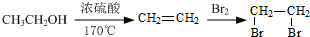
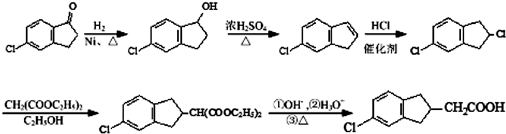 ��
��