��Ŀ����
10��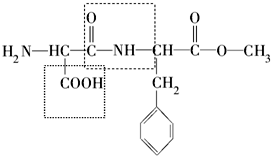 Ϊ���ѧѧ����ȡ�õijɾ��Լ������������Ĺ��ף����Ϲ���2011�궨Ϊ�����ʻ�ѧ�ꡱ��������Ϊ����ѧ�������ǵ�������ǵ�δ��������ѧ��עʳƷ��ȫ�����ཡ�������Ŀɳ�����չ��Ϊ�����������������������������ˮƽ��������Ҫ���ã���ش��������⣺
Ϊ���ѧѧ����ȡ�õijɾ��Լ������������Ĺ��ף����Ϲ���2011�궨Ϊ�����ʻ�ѧ�ꡱ��������Ϊ����ѧ�������ǵ�������ǵ�δ��������ѧ��עʳƷ��ȫ�����ཡ�������Ŀɳ�����չ��Ϊ�����������������������������ˮƽ��������Ҫ���ã���ش��������⣺��1��ά���صĹ�������ӽṹ������أ�����ѡ�����ܴٽ�Ca2+���գ��������Ͳ������Dz����Ǣܣ��ܷ���ҹä֢���������˵��Ǣ٣�����ţ���
������A ������B2������C ������D
��2����˾������һ���˹��ϳɵ���ζ����������Լ��200��������û��Ӫ�����ʺ���������֢���ߣ�д����˾������������߿��ڹ����ŵ����ƣ��ļ����Ȼ���
��3�����������ϰ�����Ӫ��ȱ��֢������Ҫ��������ȡ�����ʣ�������ʡ�����֬���������ࡱ����������ģ�����ʳ���и��������ʵ���C������ĸ����
A������ B�������� C������ D������
��4��ʳƷ���Ӽ���Ϊ����ʳƷɫ���㡢ζ��Ʒ�ʣ��Լ�Ϊ�����ͼӹ����յ���Ҫ������ʳƷ�е��˹��ϳɻ�����Ȼ���ʣ�ΪԤ����ȱ������������ʳ��������KIO3����ѧʽ����ΪӪ��ǿ������̼�����ơ�̼������dz��������ɼ���д��̼��������Ϊ���ɼ�������ԭ������ѧ����ʽ����2NaHCO3 $\frac{\underline{\;\;��\;\;}}{\;}$Na2CO3+H2O+CO2����
��5�����ݰ���ƥ������������һ������ʽ����������������ʡ�˻�ͽϸ�ˮƽ���������Լ�Ⱥ�ڽ����ȹ�����Ҫ���ڽ���ʱ��ʹ���˴����ֽ�����������ĺͲ����Ȳ��ϣ���ش𣺸ֽ���������� ���ϲ��ϣ�ѡ��������ϡ��������ǽ������ϡ������л��߷��Ӳ��ϡ����ϲ��ϡ����������������ǽ������ϣ�ͬ�ϣ���������ͨ��������Ҫԭ���Ǵ��ʯ��ʯ��ʯӢ���������Ļ�Ա���ǿ�������ڿ�����ȴ�������ֳ����õĿ���ʴ�ԣ���ԭ������������е�����������Ӧ�����������һ�����ܵ�����Ĥ���������ĵ���Ϣ����ϵͳ��ʹ���˹��ά�����ά����Ҫ�ɷ���SiO2����ѧʽ����
���� ��1��ά���ؼȲ����빹������ϸ����Ҳ��Ϊ�����ṩ���������������������Ҫ����С������������ĸ������������Ҫ�����ã��������ȱ��ά����A�Ỽҹä֢��ȱά����C�Ỽ��Ѫ�����ֿ����½���ȱά����D�Ỽ���Ͳ����������ɣ��ݴ˽��
��2�����ݰ�˾����Ľṹ��ʽ�����жϰ�˾������������߿��ڹ����ŵ�����Ϊ�ļ����Ȼ���
��3��ȱ�������ʻ�Ӫ��ȱ��֢������ʳ���к��е����ʣ�
��4��ΪԤ����ȱ������������ʳ��������KIO3��ΪӪ��ǿ���������⣻̼���������ȷֽ����ɶ�����̼���������Ϊ���ɼ���
��5���ֽ���������ڸ��ϲ��ϣ������������ǽ������ϣ����첣������Ҫԭ��Ϊ���ʯ��ʯ��ʯӢ����������е�����������Ӧ�����������һ�����ܵ�����Ĥ��������һ������ʴ�����ά����Ҫ�ɷ���SiO2��
��� �⣺��1���ܷ���ҹä֢����ά����A����Ԥ�����Ͳ�����ά����D���ܷ��λ�Ѫ������ά����C���ʴ�Ϊ���ܣ��٣�
��2���ɰ�˾����Ľṹ��ʽ����˾������������߿��ڹ����ŵ�����Ϊ�ļ����Ȼ����ʴ�Ϊ���ļ����Ȼ���
��3��ȱ�������ʻᵼ���������������ϰ�����Ӫ��ȱ��֢������ʳ���и��������ʣ����������к��зḻ�ĵ����ʣ�
�ʴ�Ϊ�������ʣ�C��
��4��ΪԤ����ȱ������������ʳ��������KIO3��ΪӪ��ǿ���������⣻̼���������ȷֽ����ɶ�����̼���������Ϊ���ɼ�������ʽΪ��2NaHCO3 $\frac{\underline{\;\;��\;\;}}{\;}$Na2CO3+H2O+CO2�����ʴ�Ϊ��KIO3��2NaHCO3 $\frac{\underline{\;\;��\;\;}}{\;}$Na2CO3+H2O+CO2����
��5���ֽ���������ڸ��ϲ��ϣ������������ǽ������ϣ����첣������Ҫԭ��Ϊ���ʯ��ʯ��ʯӢ����Ϊ��������е�����������Ӧ�����������һ�����ܵ�����Ĥ��������һ������ʴ���������ڿ�����ȴ�������ֳ����õĿ���ʴ�ԣ����ά����Ҫ�ɷ���SiO2��
�ʴ�Ϊ�����ϲ��ϣ����ǽ����������������е�����������Ӧ�����������һ�����ܵ�����Ĥ��SiO2��
���� ���⿼���Ϊ�ۺϣ�������Ӫ�����ʡ������Լ�ҽҩ�ȽǶȵĿ��飬Ϊ�߿��������ͣ�����������ѧ�������õĿ�ѧ���������ѧϰ�Ļ����ԣ��ѶȲ���ע�⻯ѧ�������������е�ʵ��Ӧ�����ص㣮

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | SO2��SiO2��CO��Ϊ���������� | B�� | ϡ���������ᡢ�Ȼ�����Һ��Ϊ���� | ||
| C�� | ���ᡢˮ��������ˮ��Ϊ����� | D�� | �ռ�����ᡢ���Ȼ�̼��Ϊ����� |
��1����֪һ�������·�����Ӧ��2SO2��g��+O2��g��?2SO3��g����H��0���ڷ�Ӧ�����У�����Ӧ���ʵı仯��ͼ��ʾ����������ʵı仯�ش��ȡ�Ĵ�ʩ���ı����������

��2��̽����Ӧ������0��l0mol/L��Na2S2O3��Һ��ϡH2SO4��Ӧ���ʵ�Ӱ�죮�������ⶨ��������
| ��� | ��Ӧ�¶�/�� | Na2S2O3ŨҺ/mL | �� | 0.1mol/LH2SO4��Һ/mL | �� |
| �� | 25�� | 10.0 | 0 | 10.0 | |
| �� | 25�� | 5.0 | a | 10.0 | |
| �� | 45�� | 10.0 | 0 | 10.0 |
��3����֪2KMnO4+5H2C2O4+3H2SO4=K2SO4+2MnSO4+8H2O+10CO2�����ڿ�ʼһ��ʱ���ڣ���Ӧ���ʽ�С����Һ��ɫ�����ԣ�������ͻȻ��ɫ����Ӧ������������
�������������ijͬѧ��Ϊ�÷�Ӧ���ȣ�������Һ�¶���������Ӧ��������Ӱ�컯ѧ��Ӧ���ʵ����ؿ�������뻹�����Ǵ������������ӵĴ����ã���Ӱ�죮
������ʵ��֤����IJ��룬�����Ը��������Һ��������Һ�⣬�����ڷ�Ӧһ��ʼʱ����C������ĸ��ţ���
A�������B���Ȼ���c��������D��ˮ��
��1����ˮ��T��ʱ��pH=6�����¶���1mol•L-1��NaOH��Һ�У���ˮ�������c��OH-��=10-12 mol•L-1��
��2��ijһԪ������Һ��A�����Ԫǿ�ᣨB����pH��ȣ���������Һϡ����ͬ�ı�����pH��A����pH��B�� �����������=������������������ϡ����Һ�к͵�Ũ�ȵ������NaOH��Һ������ϡ����Һ�����V��A����V��B�������������=����������
��3����֪����Ԫ��H2R �ĵ��뷽��ʽ�ǣ�H2R=H++HR-��HR-?R2-+H+����0.1mol•L-1NaHR��Һ��c��H+��=a mol•L-1����0.1mol•L-1H2R��Һ��c��H+���� ��0.1+a�� mol•L-1�����������������=������������H2R�е�һ���������H+��HR-�ĵ���������������ã�
��4������ƽ�ⳣ���Ǻ���������ʵ���̶ȵ�����������֪��
| ��ѧʽ | ���볣����25�棩 |
| HCN | K=4.9��10-10 |
| CH3COOH | K=1.8��10-5 |
| H2CO3 | K1=4.3��10-7��K2=5.6��10-11 |
����NaCN��Һ��ͨ��������CO2��������Ӧ�Ļ�ѧ����ʽΪNaCN+H2O+CO2=HCN+NaHCO3��
| A�� | �����������O2-+2H+�TH2O | |
| B�� | �����ʯ��ˮ��ͨ�������CO2��Ca2++2OH-+CO2�TCaCO3��+H2O | |
| C�� | ����ʯ��ˮ��Ca��HCO3��2��Һ��Ӧ��Ca2++HCO3-+OH-�TCaCO3��+H2O | |
| D�� | ����������Һ������þ��Һ��Ӧ��Ba2++SO42-�TBaSO4�� |
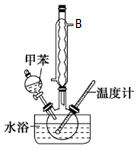 �����ױ����졢�䡢�����֣����ж������ױ���ҽҩ��Ⱦ�ϵȹ�ҵ��һ����Ҫ�л��м��壬������Ũ����Ϊ��������Ũ����Ϊ������ͨ���ױ���������Ӧ�Ʊ���һ���µ��Ʊ��������ױ���ʵ�鷽���ǣ��Է�������Ϊ������������NaHSO4Ϊ��������ѭ��ʹ�ã�����CCl4��Һ�У�����������������ˮ���ã���45�淴Ӧ1h����Ӧ�������ˣ���Һ�ֱ���5%NaHCO3����Һ��ˮϴ�����ԣ��پ������ᴿ�õ��������ױ���
�����ױ����졢�䡢�����֣����ж������ױ���ҽҩ��Ⱦ�ϵȹ�ҵ��һ����Ҫ�л��м��壬������Ũ����Ϊ��������Ũ����Ϊ������ͨ���ױ���������Ӧ�Ʊ���һ���µ��Ʊ��������ױ���ʵ�鷽���ǣ��Է�������Ϊ������������NaHSO4Ϊ��������ѭ��ʹ�ã�����CCl4��Һ�У�����������������ˮ���ã���45�淴Ӧ1h����Ӧ�������ˣ���Һ�ֱ���5%NaHCO3����Һ��ˮϴ�����ԣ��پ������ᴿ�õ��������ױ�����ش��������⣺
��1������B�����������������ܣ���������������������
��2�������ڷ�Һ©����ϴ�Ӿ����л��㴦���²㣨��ϡ����¡�����Һʱ��������Һ�����������������ԭ�����Һ©�����������⣬���з�Һ©���Ͽ�����δ��
��3�������µ����鷽���й��˵�Ŀ���ǻ���NaHSO4����Һ��5%NaHCO3��Һϴ�ӵ�ԭ���dz�ȥ���ᡢ���ᡢ���ᣮ
��4�����и����˴������༰�����Լױ�������ӦӰ���ʵ������
| ���� | n�������� | ���������и����칹������������%�� | �ܲ��� ��%�� | ||
| n���ױ��� | �������ױ� | �������ױ� | �������ױ� | ||
| ŨH2SO4 | 1.0 | 35.6 | 60.2 | 4.2 | 82.0 |
| 1.2 | 36.5 | 59.2 | 4.0 | 83.8 | |
NaHSO4 | 0.25 | 46.3 | 52.8 | 0.9 | 92.8 |
| 0.32 | 48.0 | 51.7 | 0.3 | 95.0 | |
| 0.36 | 45.0 | 54.4 | 0.6 | 93.9 | |
���ɱ������ݿ�֪�ױ�������Ҫ�õ��������ױ����������ױ���ԭ���DZ����ϵļ�ʹ���������Ӽ���̼ԭ�ӵ��ڶ�λ������Ӧ�������У�
����Ũ������ױ�������ȣ�NaHSO4���ױ����������������������ж������ױ�������ߣ����������������ѭ��ʹ�ã����ش�һ����
�ܶ������ױ����������ױ��IJ����������������
| �ܶ�/g•cm-3 | �е�/�� | �ܽ��� | |
| �ױ� | 0.866 | 110.6 | ������ˮ�������������ױ� |
| �������ױ� | 1.286 | 237.7 | ������ˮ��������Һ̬�� |
| �������ױ� | 1.162 | 222 | ������ˮ��������Һ̬�� |

���У�����1������Ϊ��Һ������2����Ҫʹ�����������е�ABC������ţ���
A�������� B���ƾ��� C���¶ȼ� D����Һ©�� E��������
| A�� | δ��ȴ��ת�ơ����� | B�� | ����ƿ��ԭ����������ˮ | ||
| C�� | ����ʱ���ӿ̶��߹۲�Һ�� | D�� | ����ʱ�Ӷ���ˮ������������ |
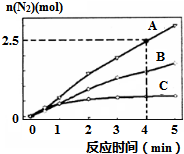 ��������������������й㷺��;��
��������������������й㷺��;��